Transcriber’s Notes
Obvious typographical errors have been silently corrected. Variations
in hyphenation have been standardised but all other spelling and
punctuation remains unchanged.
The original makes extensive use of „. This has been replaced by the
original text in some cases where this improved clarity or layout.
The mathematical and chemical formulae accurately represent the original
but have not been error checked.
HYGIENE:
A MANUAL
OF
Personal and Public Health
BY
ARTHUR NEWSHOLME, M.D., F.R.C.P., Lond.,
UNIVERSITY SCHOLAR IN MEDICINE; DIPLOMATE IN PUBLIC HEALTH, UNIV. LOND.; MEDICAL
OFFICER OF HEALTH OF BRIGHTON; MEMBER OF THE COUNCIL AND EXAMINER TO THE
SANITARY INSTITUTE; EXAMINER IN STATE MEDICINE TO THE UNIVERSITY OF
LONDON; LATE EXAMINER IN PREVENTIVE MEDICINE TO THE UNIVERSITY OF
OXFORD, AND PRESIDENT OF THE INCORPORATED SOCIETY OF MEDICAL
OFFICERS OF HEALTH.
NEW EDITION, 1902. ILLUSTRATED.
LONDON:
Geo. Gill & Sons, Ld., Minerva House, Warwick Lane.
v
The writing of a preface is perhaps superfluous for a book which has
had a large and steady sale for nearly twenty years, and which has
evidently met with the approval of a large constituency. A few words
of introduction appear, however, desirable in view of the facts that the
present edition has been almost entirely re-written; that a large amount
of new matter has been introduced; and that, so far as is known, the
comments on each subject represent the most recent and authoritative
knowledge upon it.
An attempt has been made to meet the requirements of medical
students, as well as of science students and general readers, for whom
former editions were chiefly intended. A large class of medical students
and practitioners do not require the detailed statement of the subject
contained in the larger text-books. For them, and, it is hoped, also for
a large number of candidates for diplomas in public health and in sanitary
science, the present edition will prove to be useful. At the same time,
the subject has been treated as non-technically as is consistent with
accuracy, in order to retain its suitability for non-medical readers. A
large number of new illustrations have been introduced.
The new chapters dealing with Dietetics, Trade Nuisances,
Meteorological Observations, Tuberculosis, Disinfection, and Vital
Statistics will, it is believed, enhance the value of the book.
Attention is also drawn to the solutions of mathematical problems
in the different branches of hygiene, of which a table of contents is given
on page viii.
In its new form, it is hoped that this work will be found to have
retained its value as a plain and straightforward account of its subject
for the general public and for science students; and to have become a
practical guide to sanitary inspectors and to medical students, whether
preparing for a diploma in public health, or studying hygiene as an
important branch of medicine. The use of smaller type for specially
technical matter of less general interest will facilitate discriminative
reading.
ARTHUR NEWSHOLME.
Brighton,
February 28th, 1902.
vii
TABLE OF CONTENTS.
| Chapter. |
|
PAGE |
| I. |
—Introductory |
1 |
| II. |
—Food |
4 |
| III. |
—The Varieties of Food |
9 |
| IV. |
—Diseases due to Food |
23 |
| V. |
—Diet |
29 |
| VI. |
—The Preparation and Preservation of Food |
38 |
| VII. |
—Condiments and Beverages |
45 |
| VIII. |
—Fermented Drinks |
55 |
| IX. |
—Water |
65 |
| X. |
—The Storage and Delivery of Water |
74 |
| XI. |
—Impurities of Water |
78 |
| XII. |
—Origin and Effects of the Impurities of Water |
89 |
| XIII. |
—The Purification of Water |
94 |
| XIV. |
—Composition and Properties of Air |
100 |
| XV. |
—Suspended Impurities of Air |
105 |
| XVI. |
—Gaseous and Other Impurities of Air |
111 |
| XVII. |
—Trade Nuisances |
120 |
| XVIII. |
—The Examination of Air |
125 |
| XIX. |
—The Purification of Air |
129 |
| XX. |
—General Principles of Ventilation |
132 |
| XXI. |
—Problems as to Ventilation |
137 |
| XXII. |
—Methods of Ventilation |
146 |
| XXIII. |
—Ventilation by the Introduction of Warmed Air |
155 |
| XXIV. |
—The Warming of Houses |
158 |
| XXV. |
—House Drainage |
165 |
| XXVI. |
—Cesspools and Main Sewers |
183 |
| XXVII. |
—Problems as to Flow in Sewers |
187 |
| XXVIII. |
—The Disposal of Sewage |
190 |
| XXIX. |
—Conservancy Methods |
194 |
| XXX. |
—Position of the House |
201 |
| XXXI. |
—The Materials used in the Construction of a House |
205 |
| XXXII. |
—Construction of the House |
209 |
| XXXIII. |
—The Soil |
219 |
| XXXIV. |
—Climate and Weather |
227
viii |
| XXXV. |
—Meteorological Observations |
237 |
| XXXVI. |
—Personal Hygiene |
245 |
| XXXVII. |
—Personal Hygiene—Exercise |
249 |
| XXXVIII. |
—Personal Hygiene—Rest and Sleep |
257 |
| XXXIX. |
—Personal Hygiene—Cleanliness |
260 |
| XL. |
—Clothing |
265 |
| XLI. |
—Parasites |
273 |
| XLII. |
—The Rôle of Insects in Spreading Disease |
281 |
| XLIII. |
—Infective Diseases |
284 |
| XLIV. |
—Acute Infective Diseases |
291 |
| XLV. |
—Tuberculosis |
309 |
| XLVI. |
—Notification and Isolation |
317 |
| XLVII. |
—Disinfection |
324 |
| XLVIII. |
—Vital Statistics |
335 |
SPECIAL TABLE OF CONTENTS FOR ARITHMETICAL
PROBLEMS IN HYGIENE.
| PAGE |
| Problems in Milk Analysis |
12 |
| Problems in Dietetics |
35 |
| Problems in Water Analysis |
86 |
| Problems in Air Analysis |
126 |
| Problems as to Ventilation |
137 |
| Problems as to Flow in Sewers |
187 |
| Problems in Meteorology |
242 |
| Problems as to Work |
254 |
| Problems in Vital Statistics |
336 |
1
HYGIENE.
In classical mythology, Æsculapius was worshipped as the god of
Medicine, while his daughter Hygeia had homage done to her as
the sweet and smiling goddess of Health. The temples of these
two deities were always placed in close contiguity; and statues
representing Hygeia were often placed in the temple of Æsculapius.
In these statues she is represented as a beautiful maid, holding in
her hand a bowl, from which a serpent is drinking—the serpent
typifying the art of medicine, then merely an art, now establishing
its right more and more to the dignity of a science.
That considerable attention was paid in very early times to
matters relating to health, is also shewn by the elaborate directions
contained in the Mosaic law as to extreme care in the choice of
wholesome foods and drinks, in isolation of the sick, and attention
to personal and public cleanliness. It is not surprising, therefore,
to find that the Jews, throughout the whole of their history, have
apparently enjoyed a high standard of health.
In this country great ignorance of the laws of Health has prior
to the last fifty years prevailed, and consequently preventible
diseases have been rampant, and have claimed innumerable victims.
Each century has been marked by great epidemics, which have
swept through the country, scattering disease and death in their
course. In the fourteenth century, for instance, there was the Black
Death, a disease so fatal that it left scarcely one-fourth part of the
people alive; while Europe altogether is supposed to have lost
about 40 millions of its inhabitants, and China alone 13 millions.
A century and a half later came the Sweating Sickness (though
there were a score of minor epidemics in between). This was
carried by Henry the Seventh’s army throughout the country, and
so great was the mortality, that “if half the population in any town
escaped, it was thought great favour.” Considerable light is thrown
on the rapid spread of this disease after its importation, when we
remember that there were no means of ventilation in the houses;
that the floors were covered with rushes which were constantly put
on fresh without removing the old, thus concealing a mass of filth2
and exhaling a noisome vapour; while clothing was immoderately
warm and seldom changed; baths were very seldom indulged in,
and soap hardly used.
In the sixteenth and seventeenth centuries there were five or six
epidemics of The Plague, and it was only eradicated from London,
when all the houses from Temple Bar to the Tower were burned
down in the Great Fire of September 2nd, 1666, which destroyed
the insanitary and necessitated the building of new and larger
houses.
Scurvy, jail-fever, and small-pox, are other diseases which were
formerly frightfully prevalent. Jail-fever, the same disease as the
modern typhus-fever, has now become practically extinct in its
former habitat, owing largely to the noble work of John Howard,
“whose life was finally brought to an end by the fever, against the
ravages of which his life had been expended.” This disease was
fostered by overcrowding, ill-ventilation, and filth.
Scurvy formerly produced a very great mortality, especially
among sea-faring men. In Admiral Anson’s fleet in 1742, out of
961 men, 626 died in nine months, or nearly two out of every three,
and this was no solitary case. Captain Cook, on the other hand,
conducted an expedition round the world, consisting of 118 men;
and although absent over three years, only lost one life. He was
practically the first to demonstrate the potency of fresh vegetables
in preventing scurvy.
The striking facts respecting small-pox will be found on page
293. The general death-rate has also greatly declined. Thus while
the annual death-rate in London 200 years ago was 80 per 1,000, it
only averaged 18.8 in the four years 1896-99; and the death-rate of
England and Wales has declined from 22.4 in 1841-50 to 18.7 per
1,000 in 1891-95 and 17.6 in 1896-99.
That much still remains to be done is evident on every hand.
There is little doubt that the general death-rate might be reduced
to 15 per 1,000 per annum, instead of the present 18, were the laws
of health applied in every household and community. It has been
estimated that on the average at least 20 cases of sickness occur for
every death; therefore nearly half of the population is ill at least
once a year. A simple calculation will show how much loss the
community annually suffers from this vast mass of preventible
sickness. It amounts to many millions of pounds, leaving out
of the reckoning the suffering and distress which are always
associated with sickness. For details relating to special diseases,
see page 297.
In the prevention of this mass of sickness, the knowledge of its
causation is half the battle; when once a disease is traced to its
source, as a rule, the agency which produces it can be avoided.
The reason why even more progress has not been made in the
prevention of disease is not far to seek. In order to prevent a
disease it is necessary to remove its causes. The causes of disease
can only be ascertained by a careful investigation of its phenomena;
and it is only within the last century that these have been studied
to any large extent scientifically. Such knowledge of morbid3
processes not only results in improved measures of treatment, but
in more rational and complete measures of prevention. Thus, not
only is the number of diseases which are curable becoming gradually
augmented, but the number preventible is even more rapidly on the
increase.
Inasmuch as the preservation of health involves the prevention
of disease, Hygiene, the science of health, is sometimes called
Preventive Medicine.
The subject of Hygiene naturally divides itself into two parts,
the health of the individual, and that of the community, or
Personal and Public Health.
The former treats of the influence of habits, cleanliness,
exercise, clothing, and food on health; while the latter is concerned
with the interests of the community at large, as affected by a pure
supply of air and water, the removal of all excreta, the condition of
the soil, and with the administrative measures required to secure
the removal of evil conditions. It is obvious, however, that these
two divisions are not mutually exclusive. What is important to the
health of the community, is equally so to each individual member
of it. The purity of air and water, for instance, is of immense
importance both personally and collectively.
It will be convenient to study first the three main factors in
relation to health—food, water, and air—subsequently considering
other matters of importance to health (see pages 4-157).
4
Physiological Considerations.—All substances are foods
which, after undergoing preparatory changes in the digestive organs
(rendering them capable of absorption into the circulation), serve to
renew the organs of the body, and maintain their functions. Foods
have been classified as tissue producers or energy producers, the first
class renewing the composition of the organs of the body, and the
second class supplying the combustible material, the oxidation (or
more correctly the metabolism) of which is the source of the energy
manifested in the body. The two main manifestations of energy in
the body are heat and mechanical motion, which are to a large
extent interchangeable.
All foods come under one of these heads; they are either tissue
or energy producers. They may be both, and in many cases are
so. Thus, all nitrogenous foods (as meat, legumens, etc.) not only
help to form the nitrogenous tissues of the body, but their largest
share becomes split up into fats and urea, and so forms a source of
heat to the body. Similarly fats may possibly, after assimilation,
enter into the composition of the various tissues containing fat (of
which the brain is the most important), though they usually
supply an immediate source of heat. Proteid foods are, however,
the tissue producers par excellence, other foods serving as the immediate
sources of energy when metabolised in the body.
Certain foods do not directly serve either as tissue or energy
producers, but are useful in aiding the assimilation of food. Such
are the various condiments which may be classed as adjuncts to
food. Salt is so necessary to the assimilation of food and to the
composition of the various tissues, that it may be ranked as an
important food. Water, again, though already oxidised, and so not
an immediate source of energy, is absolutely necessary to the assimilation
of food, to the interchange between the various tissues and
the blood, and to the elimination of effete products.
Classification of Foods.—Inasmuch as milk supplies all the
food necessary for health and growth during the first year of life, it
may reasonably be expected to afford some guidance as to the
necessary constituents of a diet for the adult; although the conditions
of life being altered in the latter, we can hardly expect the
same proportions of the different materials to hold good. In the
infant rapid growth and building up of new tissues and organs are
going on, involving the necessity for a larger proportional amount
of nitrogenous food than in the adult.
5
The following is the average composition of 100 parts of
|
HUMAN MILK. |
COW’S MILK. |
| Casein |
2.4 |
4.0 |
| Albumin |
.6 |
.9 |
| Fat |
2.9 |
3.5 |
| Sugar |
5.9 |
4.0 |
| Salts |
.16 |
.7 |
|
───── |
───── |
| Total Solids |
11.96 |
13.1 |
| Water |
88.04 |
86.9 |
It is evident from this analysis of milk that our food must
contain (at least) representatives of all the above divisions. We
have, therefore:—
- 1. Nitrogenous Foods.
- 2. Hydrocarbons or Fats.
- 3. Carbohydrates or Amyloids.
- 4. Salts.
- 5. Water.
Condiments and stimulants (tea, coffee, alcohol) are not foods
in the strict sense of the word, and will be discussed in a later
chapter.
Nitrogenous Foods include albumin, casein, gluten, legumen,
fibrin, and gelatin. They all agree in consisting of a complex
molecule containing many atoms of carbon, hydrogen, oxygen, and
nitrogen, with the addition of smaller quantities of sulphur, and
in some cases phosphorus. The nitrogenous substances used as
food may be divided into two groups, (a) those containing gelatin, and
(b) numerous bodies which receive the common name of proteids
or albuminoids.
The percentage composition of gelatin is:—
| CARBON. |
HYDROGEN. |
NITROGEN. |
OXYGEN. |
| 50.0 |
6.6 |
18.3 |
25.1 |
The percentage composition of all proteids lies within the
following limits:—
| CARBON. |
HYDROGEN. |
NITROGEN. |
OXYGEN. |
SULPHUR. |
| 52.7 to |
6.9 to |
15.4 to |
20.9 to |
0.8 to |
| 54.5 |
7.3 |
16.5 |
23.5 |
1.6 |
Proteids also contain a small amount of phosphorus, chiefly as
phosphate of lime, but also in minute quantity in their essential
structure. Various proteids are used in food, e.g. serum-albumin in6
the blood and tissues of animals; egg-albumin in the white of
eggs; myosin in flesh; casein in milk; legumin, or plant-casein, in
the seeds of leguminous plants; gluten in wheat-flour, etc.
Proteid foods are pre-eminently important, as they construct
and keep in repair the tissues of the body. They are not used solely
for this purpose. A large share of the energy of the body is derived
from the metabolism of proteids. The amount required for these
purposes will be discussed on page 32. Meanwhile, it may be said
that it is not found to be compatible with efficient health simply to
supply an amount of proteid food which will suffice to replace the
wear and tear of the tissues, leaving fats and carbohydrates to
supply the energy of the body. Deficiency of proteid food always
leads to ill-health; and it would appear that in all cases proteid food
determines, to a large extent, the metabolism of non-nitrogenous
food, and so is favourable to all vital action. The action of nitrogenous
food in thus increasing metabolism may make it, when in
relative excess, a tissue waster. Banting’s cure for corpulence is
founded on this principle, lean meat alone being taken, all starchy
and saccharine foods being carefully avoided.
By metabolism is meant the changes undergone by food before
it reaches the state in which it is finally eliminated from the
body. It is commonly spoken of as oxidation, but this word less
exactly represents the facts. The complexity of the changes
undergone by food in the body may be better appreciated by a
glance at the following schematic statement, which only gives an
approximation to the truth:-
HYPOTHETICAL
ALBUMINOID. |
TYPICAL FAT
(STEARIN). |
STARCH. |
GRAPE-
SUGAR. |
| C₇₂H₁₁₂N₁₈SO₂₂. |
C₃H₅(C₁₈H₃₅O₂)₃. |
x(C₆H₁₀O₅). |
(C₆H₁₂O₆). |
| ─────────────────────┬──────────────────── |
Various intermediate products, which are finally broken down
into and eliminated as |
| ┌──────────┬────────────┐ |
| Urea, CH₄N₂O. |
Carbonic acid, CO₂. |
Water, H₂O.> |
Hydrocarbons, or fats, consist of three elements, carbon,
hydrogen, and oxygen, the amount of oxygen present not being
sufficient to oxidise completely either the hydrogen or the carbon.
Thus the molecule of stearin, which may be taken as a typical fat,
has the formula C₃H₅ (C18H35O₂)₈.
In respect to their comparatively unoxidised condition fats
compare favourably with starch and sugar, C₆H10O₅ and C₆H12O₆
respectively. It is evident that in starch the H10O₅ = 5H₂O, and
that in sugar H12O₆ = 6H₂O, so that in both cases only carbon
remains uncombined with oxygen. Dried fats produce by their
oxidation 2¼ times as much heat as a corresponding amount of
sugar or starch; but the relative advantage of fat is not quite so
great as would appear from this comparison, inasmuch as
metabolism within the body is not identical with oxidation.
The fat obtained from food is not simply deposited in the body
as such, to form a store of combustible matter, and to fill up the7
interstices between the different tissues. If this were so, the kind
of fat deposited would vary with the food, which is not the case.
The fat of the body is probably not formed directly from fatty food,
but as the result of the metabolism of nitrogenous foods when this
metabolism is incomplete. In the formation of milk this can be
distinctly proved: the fat cells are formed from the protoplasm of
the cells of the mammary gland.
Possibly carbohydrate food may be a source of fat, as well as
nitrogenous and fatty food. This appears to be the case in the
Strasburg goose, which is kept penned up in a warm room, and fed
entirely on barley-meal, in order to produce an enormous fatty liver
for the delicacy termed pâté de foie gras. But it may be that the
large accumulation of fat in the liver is due to the warmth and
inaction of the goose diminishing metabolism, and producing a fatty
degeneration of the nitrogenous material of the liver.
Fats and carbohydrates, unlike proteids, do not excite metabolism
in the system, and so, if in excess of the requirements of the
system, can be stored up with comparative ease. Quiet and warmth,
diminishing metabolism, conduce to the accumulation of fat in
animals being fed for the market; and the same applies to human
beings.
Carbohydrates or amyloids include the various starchy
and saccharine foods. They are inferior to fats in nutritive power,
but, being very digestible, are in much greater favour. In the
process of digestion, starch is converted into grape sugar, and starch
and sugar are practically equal in nutritive power.
Even when carbohydrates are entirely absent from the food,
they may be produced in the organism by the breaking up of nitrogenous
matter. This certainly happens in diabetes, in which the
nitrogenous food rapidly becomes converted into sugar and urea.
The deprivation of carbohydrate food is much better borne than
that of fats, because in the latter the hydrogen is not completely
oxidized, and because fats aid the assimilation of other food.
Salts, and especially common salt (chloride of sodium), are
essential to health. An average adult human body contains about
seven pounds of mineral matter, of which about five-sixths is in the
bones. On analysis the whole body yields about five per cent. of ash.
Chloride of sodium is necessary for the production of the acid
(hydrochloric) of gastric juice, and of the salts of bile; half the
weight of the ash of blood consists of it. An adult requires 150 to
200 grains of salt per day; a large part of this is taken in meat,
bread, etc.; and but little need be taken as a condiment. Potassium
salts form an important part of milk, muscle juice, and the blood
corpuscles. They are obtained from bread and fresh vegetables and
fruits. It has been maintained that deficiency of potassium salts
causes scurvy (see page 28); but this is now discredited, and
probably potash is chiefly useful because of the vegetable acids
with which it is associated in fruits and vegetables, which when
oxidised, help to maintain the alkalinity of the blood, e.g., tartrates,
citrates, and malates, which become carbonates in the circulation.
Calcium phosphate (bone earth) is essential for the growth of bones,8
and is very important for the young. The best source for it is milk.
There is more lime in a pint of milk than in a pint of lime water.
Next to milk, come eggs, and then cereals, especially rice as a
source of calcium. Lime salts and phosphates as drugs do not
benefit like the same substances taken in natural food, and rickets
is not curable by taking such drugs.
Oxide of iron is always present in the ash of blood and muscles,
and in smaller quantities in milk. Fish and veal are usually deficient
in it, while beef and yolk of egg are foods richest in iron. The
amount of iron required in food is minute, and it is amply supplied
by ordinary diet.
Phosphorus is an essential building material for the body. It
is contained in foods chiefly in organic combination. The foods
richest in it are yolk of egg, sweetbread (thymus), fish-roe, calves’
brains, and the germ of wheat. Milk and cheese are very rich in
phosphates.
Water forms an important article of diet. This is evident from
the fact that 80 per cent. of the blood consists of it, and 75 per cent.
of the solid tissues; and from the fact that the daily loss of water
from the system averages 50 ounces (2½ pints) by the kidneys, and
about 40 ounces by the skin and lungs. Water is not simply
received into the system as a liquid. It forms a large proportion of
the solid food taken. Thus, 87 per cent. of milk, 78 per cent. of
fish, 72 per cent. of lean meat, 38 per cent. of bread, 13 per cent. of
peas, and 92 per cent. of cabbage, consist of water.
Solid food is dissolved in the alimentary canal by the watery
secretions derived from the blood. Water swallowed as food,
begins to pass on into the intestine at once. The statement that
free consumption of water at meals delays digestion by diluting the
gastric juice is therefore not well grounded. In the blood, water
serves to carry nutrient materials to all the tissues; and, at the same
time being circulated all over the system, equalises the temperature,
favours chemical changes, and washes all the tissues. By water
again, the effete matters which have been separated by the kidneys
are washed out of its tubes.
The Oxygen of the air, in a broad sense, forms one of the foods
of the system. This will be considered later.
Besides the above classification, foods have also been classified
as follows:—
| 1. Inorganic food—Oxygen, salts. |
| 2. Organic foods |
 |
Animal |
 |
Nitrogenous. |
| Non-nitrogenous. |
| Vegetable |
 |
Nitrogenous. |
| Non-nitrogenous. |
| Or, as— |
| 1. Solid foods |
 |
Animal |
 |
Nitrogenous. |
| Non-nitrogenous. |
| Vegetable |
 |
Nitrogenous. |
| Non-nitrogenous. |
| 2. Liquid foods |
 |
Water. |
| Milk and its products. |
| Tea and similar beverages. |
| Alcoholic beverages. |
| 3. Gaseous foods—Air. |
9
Nitrogenous Animal Foods.—These are divided into two
groups, the one containing gelatin, and the other all the proteid or
albuminoid substances, which are taken in the flesh of various
animals, and in milk and eggs.
Gelatin is obtainable from bones, and from connective tissue
wherever found. Being easily digested, and absorbed, it has been
very popular as an invalid’s food; but the fact that animals cannot
sustain life on it without the addition of proteids proves that its
value is limited. It is incapable of building tissues, but is a valuable
proteid-saver, being able to save from metabolism half its weight of
proteid, or twice as much as is spared by an equal weight of
carbohydrate. Its utility in this direction is, however, limited,
because of the dilute form in which it is taken in ordinary foods. It
is useful for invalids, partly because it forms a bulk, and prevents
the evil tendency to give their food in too concentrated a form;
partly because it forms a source of easily metabolised material, and
so prevents tissue-waste; and partly because it commonly contains
phosphate of lime, derived from the bones forming the source of
gelatin.
Gelatin as prepared for the table contains a considerable proportion
of water; as little as one per cent. of gelatin in water will
cause it to gelatinise on cooling. Isinglass obtained from the
floating bladder of the sturgeon is an example of the purest kind of
gelatin; glue is an inferior sort, made from bones, etc.
Gelatin is only a cheap food when obtained, for instance, from
bones which cannot otherwise be utilised. When made from veal
it is costly out of proportion to its dietetic value.
The Flesh of various animals is one of the main sources of our
nitrogenous and fatty food. Meats may be divided into two kinds,
viz., red meat and white meat. These gradually merge into one
another. As common examples of red meats, we have beef, mutton,
pork, game, wild fowl, and salmon.
The common fowl and turkey, most fishes, rabbits, crustaceans,
and molluscs, are examples of white meat. As a rule white meats
are more digestible than red, having more delicate fibres, and containing
a smaller proportion of nitrogenous matter.
Flesh consists almost entirely of muscular tissue, of which there
are two kinds, striped and unstriped.
The striped is the variety most commonly used as food.
Unstriped muscle has a softer texture, but is not so easily masticated10
as striped, and for this reason may be indigestible. Tripe is
composed of the unstriped muscle and connective tissue of the
stomach of the cow, and if well cooked forms a cheap and easily
digested dish.
The influence of feeding on the quality of the meat is great. In
ill-fed or old animals, connective tissue is more abundant, and the
meat is tougher. Well-fed and fattened meat contains for equal
weights much more nutritious matter than non-fattened meat, the
fat which is deposited in the muscle replacing water and not
proteid. Hence the gain in nutritive value is an absolute one, and
is not attained at the expense of the proteid part of the meat. Young
animals, again, contain more water and fat and a larger proportion
of connective tissue than the full-grown, and are consequently not
so nourishing.
Meat ought to be eaten either before the onset of rigor mortis,
or near its end, before putrefaction has commenced. During rigor
mortis it is denser, tougher, and more difficult to digest than after it.
The proportion of fat in meat varies greatly in different individuals
of the same species, in different animals, and in different
parts of the same animal. According to Dr. Ed. Smith, the proportion
of fat in fat oxen is ⅓, in fat sheep ½, in calves ⅙, lambs ⅓,
and fat pigs ½.
Good meat, whether beef or mutton, ought to have a marbled
appearance, a medium colour, neither pale pink nor deep purple; its
texture should be firm, and not leave the impress of the finger; its
odour slight and pleasant, the juice reddish and acid, the bundles
of fibres not coarse, and free from foreign particles imbedded
in them; and lastly, it should not be taken from an animal
killed near the time of parturition, nor in consequence of any
accident or disease.
Beef is, as a rule, more lean than mutton or pork; it has a
closer texture, and more nutritive material in a given bulk. It is
also fullest of the red-blood juices, and possesses a richer flavour
than the two others.
Liebig’s beef extract contains little if any albumin or gelatin.
It is a useful stimulant to the gastric secretion, as in soups at the
beginning of a meal, but is not a food. Its chief constituents are
the various extractives of meat, the most important of which are
inosinic acid, kreatin (C₄H₉N₃O₂,H₂O), and inosite, or muscle sugar
(C₆H12O₆, 2H₂O). Even in substances like Bovril, containing
powdered meat fibre mixed with Liebig’s extract, the amount of
nutritive material is very small. The white of one egg contains as
much nutritive matter as three teaspoonsful of bovril. None of these
substances can be trusted like eggs or milk to keep a patient alive
for several weeks.
Mutton is regarded as being more suitable for people of sedentary
occupation than beef. Lamb is more watery than mutton, and
less nutritious.
Veal, as ordinarily prepared in this country, is difficult of
digestion; its shreddy, juiceless fibres eluding the teeth, and consequently
not undergoing proper mastication.
11
Pork is not so digestible as beef or mutton, partly because of
the large proportion of fat, and partly because its fibres are hard and
difficult to masticate. Its digestibility varies greatly with its age,
breeding, and proportion of fat.
The Flesh of Birds contains very little fat, and that found
separate from the meat is rarely nice. Most birds are edible, but
fish-eating birds are apt to be nasty. As a rule, the flavour of the
male bird is richer than that of the female. The chief virtues in
poultry are their tenderness, and the large proportion of phosphates
they contain. They are deficient in fat and in iron. To compensate
for the former, one commonly takes with them melted butter
and fat bacon or pork sausages; to compensate for the latter, the
addition of Liebig’s extract to the gravy is useful. Young, and
consequently tender, birds are known by their large feet and leg-joints.
When a bird appears at table with violet-tinged thighs and
a thin neck, if possible avoid being helped to the leg. Wild fowls
are harder and less digestible than tame. In ducks and geese fat is
more abundant, and of a stronger flavour; they are, consequently,
not so digestible as fowls.
Fish forms an important article of diet. It is easily cooked,
and usually very digestible; it possesses a larger bulk in proportion
to its nutritive quality, and hence is very valuable for those who
habitually take an excess of meat food, which is commonly the case
with those leading sedentary lives, and in declining years. There
appears to be no foundation for the statement that fish is rich in
phosphorus, and is thus a good brain food. Generally, white-fleshed
fish is more digestible than red-fleshed (such as salmon), the latter
usually containing more fat than the former. When the fat is distributed
throughout the flesh, as in the salmon, fish is more satisfying
than when it is mainly stored up in the liver, as in the cod-fish.
According to Payen, the percentage proportion of fat in soles is
only 0.248, in whiting 0.383, conger eel 5.021, mackerel 5.758, eels
23.861. The addition of some fatty food, as melted butter, is very
advisable to such meats as poultry, rabbits, soles, whiting, plaice,
haddock, cod, turbot, and other fishes; whereas sprats, eels,.
herrings, pilchards, salmon, etc., are more or less rich in fat.
A Hen’s Egg usually weighs a little under two ounces. It
consists of 74 per cent. of water and 26 per cent. of solid matter.
The white of the egg is chiefly albumin, the yolk consists of a very
digestible oil, rich in phosphorus and iron, each particle of the oil
being enveloped in a form of albumin called vitellin. The salts are
chiefly contained in the shell. There is no sugar in the egg, the
necessity for such oxidisable material for the chick being obviated
by the heat produced by incubation. Eggs, when kept for some
time, lose weight, owing to evaporation through the porous shell;
similarly, air entering from without sets up decomposition. In
a solution of brine containing an ounce of common salt to half a
pint of water, fresh eggs sink, stale ones float; rotten eggs may
even float in fresh water. Eggs may be preserved by keeping them
in brine, or, better still in lime water, or by smearing them over
with lard or butter, as soon as possible after they are laid.
12
Cow’s Milk has a specific gravity of 1028-34, and on allowing
it to stand in a long narrow vessel ought to form ten or twelve per
cent. of its volume of cream. The percentage composition of
human and cow’s milk has been given on page 5. The legal
minimum standard for dairy milk, which is presumably derived
from a number of cows, is now 3 per cent. of fat, and 8.5 per cent.
of “solids not fat.” This standard is unfortunately very low, and
allows a considerable margin of adulteration, which cannot be
prevented by legal means. Thus ordinary milk derived from a herd
of cows would probably contain 4.5 per cent. of fat; and it is, therefore,
practicable to mix pure new milk with a large proportion of
separated milk, and yet keep within the legal standard. This is
largely done in towns, and infants suffer much from the deficiency
of cream in their sole food (see page 28). The lactometer determines
the specific gravity, which should be taken at a temperature of
60° F. In skimmed or separated milk it will be over 1034;
watering on the contrary lowers the specific gravity. If the
milk has been both watered and skimmed the specific gravity will
give an uncertain indication. Measurement of the cream in a tall
narrow glass will enable one to detect the second possible source of
fallacy; but the composition of milk can only be certainly determined
by analysis. This is done (a) by evaporating a weighed amount
of milk to dryness and then re-weighing. (b) From a separate
amount of dried milk the fat is extracted by ether, the ether
then evaporated, the remaining fat weighed, and its percentage
calculated. The weight of fat deducted from the total solids i.e.
(b) from (a), gives the “solids not fat.” The following example will
make the method then followed clear. The sample gives 7.9 per
cent. of “solids not fat.” Genuine milk contains at least 8.5 per
cent. of “solids not fat.”
Then the sample contains—
100 × 7.9 ∕ 8.5 = 92.9 per cent. of genuine milk,
i.e. 7.1 per cent. of water has been added to it.
Half a pint of milk supplies as much nitrogenous nutriment as
two good-sized eggs, and as three and a half ounces of beef. Milk
may be deteriorated (1) by skimming or “separating” by machinery,
or (2) by the addition of water—the first diminishing the proportion
of fats, and the second the total amount of solids.
Skim Milk still contains nearly 1 per cent. of fat, but
Separated Milk, in which the cream has been removed by
centrifugal apparatus, contains less than 1 ∕ 8 per cent.
Skim or separated milk forms a cheap source of nitrogenous
food; but when it is sold mixed with new or alone as new milk,
the public is defrauded, and infants fed on it are robbed of the fat
which is so essential for their growth.
Condensed Milk is milk deprived of a large part of its water.
It represents three times its volume of fresh milk. There are in the
market (a) unsweetened and condensed whole milk, (b) sweetened
and condensed whole milk, and (c) sweetened and condensed skim13
or separated milk. Unfortunately the latter is most largely sold
because cheapest; and infants are thus often robbed of fat, a most
important element in their food. Always examine the label of each
tin carefully, to ascertain whether the milk has been deprived of its
cream. The law requires that this fact should be stated on the
label. Tins which have bulged should be rejected. Condensed
milk is more easily digested by infants than new cow’s milk,
but it lacks the anti-scorbutic properties of new milk (see page
28). Even the condensed whole milk if diluted beyond 1 part of
milk to 3 of water is deficient in fat. Sweetened condensed milk
has one-third its weight of extraneous sugar added to it, and on
this account it tends in children to produce fatness, and a distaste
for simple food; in children fed on it alone ossification (formation
of bone) is retarded, and resistance to illness is diminished. The
only dietetic advantages it possesses over fresh cow’s milk are
its freedom from possible disease germs and easier digestibility.
The digestion of milk is preceded by its clotting in the
stomach. The same thing happens when junket is formed by the
addition of rennet to milk. This is a different process from the
curdling of milk, which occurs when milk turns sour. The latter is
caused by the splitting up of milk sugar and the formation of lactic
acid by certain micro-organisms in the milk. When milk is heated,
a skin is formed, consisting of coagulated albumin, in which is also a
little casein, fat, and salts of lime. Boiled milk becomes sterilized.
Cow’s milk should always be boiled, unless it is quite certain that
the cows from which it is derived are perfectly healthy, and that
the milk has not been exposed to infection before reaching the
house. The disadvantages of boiling which are outweighed by
its advantages, are that the taste of the milk is altered, some
nutritive matter is lost by the formation of the “skin,” and the
casein is not quite so easily digested. Pasteurization of milk, i.e.
keeping it at a temperature of 70° C. (158° F.) for 20 to 30 minutes
has been proposed as an alternative to boiling. This appears to
destroy the bacilli causing tuberculosis (see page 312). The typhoid
bacilli are killed at 60° C. in five minutes when suspended in
emulsion. Pasteurization is not, however, so certainly efficacious
for other disease-germs as is boiling, and is not so easily carried
out in domestic life as boiling. By boiling milk in a double
saucepan, i.e. in a water-bath, very little change occurs in the taste
of the milk, especially if it be cooled rapidly and strained.
Cheese is prepared by coagulating milk by “rennet,” the
mucous membrane of the fourth stomach of the calf, salted and
dried before using. By this means the casein is precipitated,
carrying down with it the cream, and a large proportion of the salts
of milk. The whey, containing the sugar, soluble albumin, and
remaining salts, is separated by straining, while the mixed curd and
fat are pressed in moulds. Cheese thus consists of casein, fat in
varying proportions, water and salts, especially phosphate of lime.
It is coloured with annatto, a vegetable colouring matter. When
new, cheese is tough; when old, its oils tend to become rancid;
the best age is from nine to twenty months. It is probable that14
cheese in small amount helps the digestion of other foods, though it
is itself a highly concentrated and comparatively indigestible food.
When toasted it is proverbially indigestible.
There are many different kinds of cheese. The following
classification gives the more important varieties:—
(1) Cream cheese is the new curd only slightly pressed, and is more
digestible than ordinary cheese.
(2) Next to these are cheeses made with whole milk rich in cream, such as
Stilton, Gorgonzola, Cheshire, and Cheddar.
(3) Cheeses made of poor or partially skimmed milk, such as Shropshire,
Single Gloucester, and Gruyère.
(4) Cheeses made of skimmed milk, such as Suffolk, Parmesan, and Dutch.
American cheeses may belong to any of these classes; they are
generally pure, but occasionally are made from separated milk,
margarine being added to take the place of cream. The sale of
such cheeses, except under the name of “margarine cheese,” is now
illegal.
Non-Nitrogenous Animal Foods.—These are all fats, and
the most important are the various meat fats and butter. They
possess a higher food value than carbohydrates in the proportion of
2¼; to 1. The composition of the various fats differs somewhat;
they usually contain varying proportions of olein, palmitin, and
stearin, which are compounds of glycerine with the radicle of a fatty
acid (stearin = C₃H₅ (C18H35O₂)₃). Thus mutton suet consists of
stearin, olein, and palmitin, with a preponderance of stearin. Beef
suet contains less stearin and more olein than mutton suet. The
more olein a fat contains the less solid it is. Olive oil is composed
almost entirely of olein. Palmitin, which melts sooner than stearin,
is the chief solid constituent of butter, while olein is its chief liquid
constituent. Butter is specially distinguished by containing 7 to 8
per cent. of “volatile fatty acids,” such as butyric, caproic, etc.,
combined with glycerine. The presence and amount of these
compounds is an important test for the freedom of butter from
adulterating fats.
Cod-liver oil is next to butter the most digestible animal fat
known. The best cod-liver oil is frozen at a low temperature, by
which means the stearin is frozen out, and nearly pure olein is left.
Traces of iodine have been found in it, and more commonly a small
amount of bile, which probably increases its digestibility.
The temperature at which a fat becomes hard is a fair guide to
its digestibility. Thus we know that beef, and still more, mutton
fat, would become solid, under conditions in which bacon dripping
is still soft. Where digestion is weak, there may be an instinctive
loathing of fat meat; for such persons, especially for children,
some other fat should always be substituted. Thus the addition
of butter to the potatoes makes up the deficiency.
Butter forms 3½ to 4½ per cent. of cow’s milk. It is separated
from milk by churning, the oil particles being deprived by this15
means of their albuminous coats. The more completely the butter-milk
is separated the longer the butter keeps. It can be kept longer
if salt is added, or in hot weather by keeping it under frequently-changed
water. Rancidity indicates the decomposition of traces of
the fat of butter into its fatty acid and glycerine.
Cream contains about 30 per cent. of butter fat, Cheshire
cheese 25 per cent., and skim milk cheese 7 per cent.
Butter milk differs from skim milk in the presence of lactic
acid. It is more digestible than skim milk, the casein being in a
more flocculent condition.
The odour and flavour of butter are not due to olein and
palmitin, the two chief constituents, but to a smaller quantity of
butyrin, caproin, and caprylin fats of a much lower series. Ordinary
butter contains a considerable proportion of water, and the presence
of about 8 per cent. renders it more palatable; if it is over 15 per
cent., the butter is considered adulterated. An excessive amount of
salt is sometimes present. The most frequent adulteration is the
substitution of foreign fats for butter fat, e.g. lard, palm oil, rape seed
oil, or cocoa-nut oil. Margarine is most frequently used for this
purpose.
Margarine is prepared from beef-fat by melting, the stearin
becoming solid again at a temperature at which olein and margarine
still remain liquid. It forms a wholesome and cheap food, being
nearly as digestible as butter, for which more expensive food it is
often fraudulently sold. When mixed with a small proportion of
butter its recognition by smell, etc., is almost impossible, but on
careful chemical analysis, it is found to have a higher melting point
and a lower specific gravity than butter, and a much smaller percentage
of soluble fatty acids than the latter. Thus:—
|
MELTING
POINT. |
SPECIFIC
GRAVITY. |
SAPONIFICATION AND FORMATION
OF INSOLUBLE FATTY ACID. |
| Butter |
32° C. |
.913 |
88 per cent. insoluble fatty acid |
| Margarine |
35° C.1 |
.904-.907 |
952 per cent. insoluble fatty acid |
Cereal Foods.—Gluten is peculiar to plants, and is chiefly
found in plants belonging to the great family of grasses. Gluten is
to bread what casein is to milk, and myosin to flesh. If one takes
a piece of dough made from wheat flour, and holds it under a stream
of water from the tap, a large part of it is washed away, while a
sticky adherent mass is left behind. This is gluten, and it is its
tenacity which enables bread to be made. If the fluid with which
the dough was washed is collected, it will be found to contain
a large quantity of starch, a small amount of sugar, of albumin, and16
certain salts. All cereals possess these constituents in various
proportions, as may be seen from the following table:—
|
WATER. |
PROTEID. |
FAT. |
CARBO-
HYDRATES. |
CELLULOSE. |
MINERAL MATTER. |
| Wheatmeal |
12.1 |
12.9 |
1.9 |
70.3 |
1.6 |
1.2 |
| Fine wheat flour |
13.0 |
9.5 |
0.8 |
75.3 |
0.7 |
0.7 |
| Oatmeal |
7.2 |
14.2 |
7.3 |
65.9 |
3.5 |
1.9 |
| Barley meal |
11.9 |
10.0 |
2.2 |
71.5 |
1.8 |
2.6 |
| Maize meal |
11.4 |
8.5 |
4.6 |
72.8 |
1.4 |
1.3 |
| Rice (husk removed) |
12.0 |
7.2 |
2.0 |
76.8 |
1.0 |
1.0 |
The proteid varies in character in the different cereals; wheat
flour has the largest proportion of gluten (8 to 12 per cent.) and
therefore makes the best bread.
Good wheat flour ought to be white, not gritty or lumpy, not
acid or musty, forming a coherent stringy dough. Examined
microscopically, it should show the absence of any fungi, or acarus
farinæ, or of foreign starches, such as barley, maize, rice, potato,
known by the different shape of their starch granules. (See Fig.
1.) Alum has been occasionally added to flour, to enable the
baker to make a white and porous bread from damaged wheat
flour. It can be detected as follows:—Pour over the freshly cut
surface of a slice of bread some freshly prepared decoction of
logwood chips, and then a solution of carbonate of ammonia. If
alum is present, the bread turns a marked blue to violet colour; but
if the bread is pure, it is only stained pink.
The wheat grain may be used as food in its entirety. Thus
boiled in milk, after having been soaked in water, it forms the chief
constituent of frumenty. Usually it is converted into flour by
grinding or milling. A grain of wheat consists of three parts, an
outer envelope, the bran, consisting chiefly of indigestible cellulose,
and composing 13½ per cent. of the grain; the kernel, or endosperm,
which makes up 85 per cent. of the grain; and the germ, forming 1½
per cent. of the grain. In the old method of stone grinding, the bran
was removed, and the germ left along with the endosperm. In the
elaborate processes of modern roller milling, the bran is removed as
in the old grinding, because it cannot without the greatest difficulty
be reduced to powder; and the germ is also removed, because the
oil abundantly present in it is apt to become rancid and spoil the
flour, and because the soluble proteids in it are apt to change some
of the flour into dextrin and sugar, which become brown in baking
and spoil the appearance of the bread. The germ is easily removed,
because its toughness causes it to be flattened out in the milling,
while the endosperm becomes powdery. The central part of the
endosperm is the source of ‘patents.’ It is very rich in starch and
is used for making fancy breads and pastry. The outer part of the
endosperm is ‘households.’ ‘Households flour’ is subdivided into
(a) second patents, or ‘whites’; (b) first households; (c) second
households or ‘seconds.’ ‘Seconds’ is richest in gluten,17 ‘whites’
in starch. Ordinary bread is normally derived from a blend of
these three. Some ‘strong’ wheats, e.g. Australian, yield a
‘patents’ which is rich in gluten, and such flour is used for making
Vienna bread. ‘Strong’ wheats take up most water in baking, and
so yield most loaves per sack. ‘Seconds’ flour yields a bread which
is richer in proteid than most other kinds; but the dark colour of
the loaf makes it unpopular. Various schemes have been devised
to utilise the germ and the bran, which are ordinarily discarded. In
the preparation of Hovis flour the separated germ is partially cooked
by superheated steam. This kills the ferment contained in the
soluble proteids, and thus prevents it from changing starch into
maltose and dextrin. The action thus prevented is represented
by the following formula:—
STARCH.MALTOSE.DEXTRIN.
10 C12H20O10 + 6 H₂0 = 6 C12H22O11 + 4 C12H20O10.
The germ thus treated is ground to a fine meal, of which one part
to three of ordinary flour, forms Hovis flour. Other ‘germ breads’
are also in the market. In the making of Frame food the bran is
boiled with water under high pressure. The watery extract,
containing the mineral and part of the nitrogenous constituents of
the bran, is evaporated to dryness, and forms the basis of various
preparations. It is doubtful if this food possesses any great
value.
Brown bread is a somewhat vague expression, meaning either
an admixture of bran or of germ or of both with flour, or bread
made from whole wheat flour. In each of these cases the loaf would
be brown. The bran is rich in fat as well as in phosphates. It acts
as a mechanical irritant, ill borne by delicate stomachs, but very
useful where a tendency to constipation exists. The excess of
nitrogenous matter in brown bread and its richness in fat, do not
prove its greater nutritiveness, as it is present in a condition in
which only a portion is absorbable from the alimentary canal into
the circulation.
The harder wheats, such as Sicilian wheat, contain a larger
percentage of gluten; and from them macaroni and vermicelli are
obtained, which are nearly pure gluten. They are very nutritious
and useful foods. Semolina is prepared from wheat, the millstones
being left sufficiently apart to leave the product in a granular
condition. In malted breads, a syrupy infusion of malted barley
(malt extract) is added to the flour. Malt extract contains in
addition to malt sugar (maltose) and dextrins, a ferment (diastase)
which, like the saliva, is able to convert starch into the soluble
substances, maltose and dextrin (see formulæ above). The action
of this ferment is stopped by the temperature of baking. Hence
even when the malt extract is allowed a considerable time for its
operation on the dough, only about 10 per cent. of the starch in the
loaf becomes soluble, as compared with 4 per cent. in an ordinary
loaf.
Oatmeal, obtained from the common oat, contains very little
gluten, and so cannot be made into vesiculated bread. It contains18
a large proportion of other nitrogenous material and of fat. As
porridge and oatmeal cake it forms a very nutritious diet. The husk
ought to be carefully removed from the meal intended for human
food, as, although very nitrogenous, it acts as a mechanical irritant.
Groats consists of oats from which the husk has been entirely
removed. The substitution of rolling for grinding in preparing oats
for food and the application of heat during the rolling process, have
made oatmeal more digestible, as in Quaker, Provost, and Waverley
oats.
Barley contains very little gluten; on this account, like
oatmeal, it does not admit of being made easily into bread.
Malt is barley which has been made to germinate by heat and
moisture and then dried, “diastase” being formed in the process.
Extract of malt, containing diastase in an active condition, is
useful in cases of impaired digestion and deficient assimilation of
food.
Rye is rarely used in this country for making bread. In
Germany it is known as “black bread,” but its colour and acid
taste make it disagreeable, and it is laxative in its action.
Maize, or Indian Corn, is deficient in gluten, and so not
suitable for making vesiculated bread. Like oatmeal, it is made
into cakes, called in America “Johnny cake.” It contains much
fatty matter, and is largely used for fattening poultry and other
animals. Oswego flour and corn flour are maize flour deprived by
a weak solution of soda, of its proteids and fat; hominy contains all
its constituents. Maize is a cheap and nutritious food. When
wheat flour is dear, it is occasionally adulterated with maize. The
adulteration can be detected by the forms of the starch granules,
examined under a low power of the microscope.
Rice contains less proteids and fat than any other cereal. Its
chief value as a food depends on the large amount of starch it
contains (table, page 16).
Leguminous Foods.—The chief seeds belonging to this group
are peas, beans, and lentils. They contain a smaller proportion of
starch, and a larger proportion of nitrogenous materials than cereals.
Thus while flour contains 9.5 and bread 8 per cent. of proteid, lean
meat 15.18 per cent., and cheese about 30 per cent., peas and beans
contain 21 to 26 per cent. (green peas only 4 per cent., dried peas 21
per cent.) of proteid. The nitrogenous material exists chiefly as
legumin, which has been called vegetable casein. Although
leguminous seeds contain more nutritive material in a given weight
than cereals, dietetically they are inferior, owing to the fact that they
are less digestible, often causing flatulence and other dyspeptic
symptoms. Cereals, again, are more palatable than leguminous
seeds, and are more prolific, and consequently cheaper. In the
absence of animal food, legumens form a useful substitute. They
are advantageously diluted with oily substances, or with rice. The
farm-labourer’s dish of broad beans and fat bacon is founded on strict
physiological principles. A mixture of lentil and barley flour is
sold under the name of Revalenta Arabica. Lentil flour costs 2½d.,
Revalenta 3s. 6d. per lb. Green peas, French beans, and scarlet19
runners are much more easily digested than are dried peas or beans.
Lentils contain the largest proportion of proteid of any of the pulses.
They also contain very little sulphur, and so do not give rise to the
same liberation of sulphuretted hydrogen in the intestine, as other
pulses. The ash of the Egyptian lentil is particularly rich in iron.
Amylaceous Foods. Amylaceous or starchy substances are
contained in many of the preceding foods; but some other foods
consist almost entirely of starch. The chief of these are sago,
tapioca, and arrowroot.
Sago is obtained from the pith of the stems of various species
of palm; a single tree may yield several hundred pounds. Alone it
is easy of digestion. Boiled with milk it forms a light, nutritious,
and non-irritating food. Fictitious sagos are frequently sold, made
from potato starch.
Tapioca and Cassava are derived from the tubers of more
than one species of the poisonous family, Euphorbiaceæ. The juices
are removed, and the prussic acid removed by heat. Tapioca only
differs from cassava in being a purer form of starch; the latter is
more nutritious, and among the Indians takes the place of bread.
Arrowroot is obtained from the tubers of Maranta Arundinacea.
Tous-les-mois is a form of starch obtained from the tubers of a
West Indian plant, the Canna edulis.
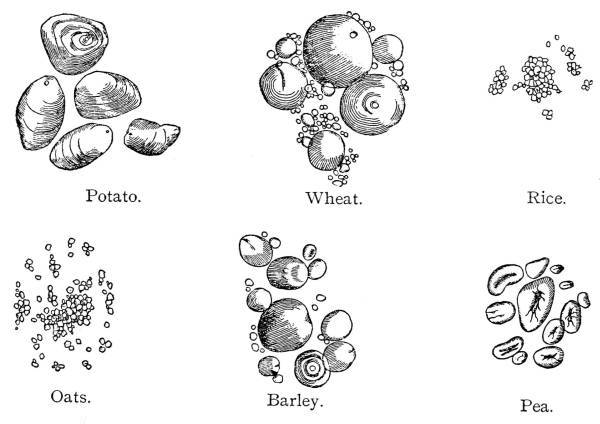
Fig. 1.—Different Forms of Starch Granules.
Potato. Wheat. Rice.
Oats. Barley. Pea.
The detection of the varieties of starch is usually possible
owing to their fairly characteristic appearance under the microscope.
Fig. 1 shows the most important starches. It must be noted that in
oats, maize, and rice the contour is completely marked by facets or
surfaces, while there are less complete markings in tapioca and sago.
In wheat, rye, pea, bean, barley, potato, and arrowroot the contour
is even, though there are minor differences of size and shape.
20
Other Vegetable Foods.—Green Vegetables contain comparatively
little nutriment, but form valuable additions to other
foods. Cellulose, which forms their main constituent, although
indigestible, forms a bulk in the alimentary canal, which is necessary
to ensure peristalsis. Concentrated nourishment can only be
digested in limited quantity, and is very apt to produce digestive
disorder. Cabbage contains 92 per cent. of water, and 2½ per cent.
nitrogenous matter. Carrots contain 6 per cent. and turnips 2 per
cent. of nitrogenous matter; parsnips are intermediate between
these. Green vegetables possess valuable anti-scorbutic properties.
They may be made an important vehicle for giving fatty food, by
adding butter, etc.
Rhubarb and sorrel contain oxalates and tartrates of potash and
lime, to which they owe their tartness. Spinach is cooling and
laxative, like rhubarb, but not tart. Sea-kale, artichoke, and
asparagus are all wholesome vegetables. Asparagus is somewhat
diuretic, and gives a peculiar, disagreeable odour to the urine.
Salads, such as mustard and cress, water-cress, endive, and the
garden lettuce are very useful as anti-scorbutics. Some of them
possess a peculiar pungency due to a volatile oil analogous to that
contained in horse-radish.
The Potato contains 26 solid parts in 100, of which nearly 20
are starch and 2½ nitrogenous matter. It forms one of our best-appreciated
vegetable foods, and as it possesses valuable anti-scorbutic
properties, its universal use is, perhaps, the chief cause of
the present rarity of scurvy. Alone, it possesses too small a
proportion of nitrogenous material to support life, but the addition
of butter milk makes up this deficiency; and these two together
form a sufficient diet to maintain life and health for a long time.
The Onion, Garlic, Leek, and Shalot, all members of the lily
family, are chiefly used as condiments. They contain an acid
volatile oil, which gives them a peculiar odour and flavour. By long
boiling, this is dissipated (as in the case of the Spanish onion), and
the onion is then fairly digestible, as well as nutritious.
Celery possesses a more delicate flavour and odour than the
preceding, but even the most tender celery is digested with
difficulty; less so, when boiled or stewed, or a constituent of soups.
Only four Fungi are, with us, commonly regarded as safe—mushrooms,
champignons, morels, and truffles; but there are many
others which are equally edible. The food value of fungi has been
exaggerated. They are difficult of digestion and contain little
nutritive material. Poisonous fungi usually have an astringent
styptic taste and a disagreeable pungent odour. In any doubtful
case it is better to abstain.
Oily Seeds contain a considerable amount of fixed oil which
renders them unfit for persons of weak digestion. The almond,
walnut, hazel-nut, and cocoa-nut are common examples. The
sweet almond, when eaten unbleached, occasionally produces nettlerash,
and its solid texture and large proportion of fixed oils render
it difficult of digestion. The chestnut contains less oil, but a large
amount of carbohydrate. It is extensively used as a food in Italy21
and some other countries. In the uncooked condition it is very
difficult of digestion.
Fruits are chiefly used as adjuncts to other foods; but the
vegetable salts and the cellulose and sugar which they contain,
make them very valuable. Cucurbitaceous fruits are used as
vegetables rather than as fruits. Vegetable marrow is wholesome
and agreeable, but not very nutritive. Cucumber is most digestible
when rapidly grown and freshly gathered.
Stone-fruits or drupes, such as the peach, nectarine, plum,
cherry, are rather luxuries than foods, like many other fruits.
Before ripening they are unfit for food; when ripening is complete,
the acids and astringent matter largely disappear. The date
contains chiefly sugar, and forms an important food in the East.
Pomaceous Fruits, as the apple, pear, and quince, are more
digestible when cooked; and, speaking generally, all fruit not
perfectly ripe should be cooked before eating. The presence of
vegetable acids in fruit soon converts the sucrose of cane sugar
into dextrose, a less sweet variety of sugar. It is therefore more
economical to sweeten after than before cooking.
The chief Berries are the grape, currant, gooseberry, cranberry,
and elderberry. The grape is the most important, and 1,500
varieties of it have been described. Its juice contains a large
amount of grape sugar (dextrose), and small quantities of glutinous
material, bitartrate of potash, tartrate of lime, malic acid, etc.
Besides the above fruits, we have strawberries, mulberries, figs,
plantains, melons, etc., which are all refreshing and anti-scorbutic.
The orange family furnishes us with the orange, lemon, citron,
lime, shaddock, and pomelo, of which the orange is by far the
most important, and possesses most valuable refreshing qualities.
Sugar exists in two chief forms, viz. sucroses and glucoses.
Sucroses, known chemically as disaccharids (Sucrose = C12H22O11;
compare starch = C12H20O10) are exemplified in cane, beet, maple,
malt (maltose), and milk sugar (lactose). Cane sugar has been
gradually displaced by beet sugar. The two are chemically identical,
and equally nutritious. Maltose is given in malt extract as a food,
and because of the digestive action of the ferment also contained in
the extract on starchy food. Thus:—
STARCH.MALTOSE.
C12H20O10 + H2O = C12H22O11.
Lactose is comparatively free from sweetness, and is hardly capable
of being fermented by yeasts.
Of Glucoses the best example is dextrose = C6H12O6, H2O,
which can be seen crystallised in dried raisins; it only possesses
one-third the sweetening power of sucrose. Starchy food becomes
changed into glucose by the action of saliva and pancreatic juice in
the alimentary canal. Grapes, cherries, gooseberries, figs, and
honey contain lævulose in addition to glucose (glucose = C6H12O6,
H2O, lævulose = C6H12O6). Lævulose resembles dextrose except in
being uncrystalline, and in its effect on polarised light. Many ripe22
fruits, such as pineapples, strawberries, peaches, citrons, contain
sucrose and lævulose, the latter being not quite so sweet as
sucrose.
In the alimentary canal sucroses are inverted into dextrose and
lævulose. Thus natural foods containing these sugars are more
readily assimilated than those containing sucrose.
The sweetening power of the varieties of sugar depends on
their degree of solubility in water. Sucrose is soluble in one-third
of its weight of cold, and in rather more of hot water. Dextrose is
soluble in its own weight of water; lævulose is more soluble, and
therefore sweeter than dextrose. Lactose requires five to six parts
of cold and two of hot water, and is therefore not so sweet as the
other varieties.
23
Diseases may arise from the noxious character or from deficiency
or excess of some particular food, or of the food as a whole.
Diseases from Unwholesome Food.—I. The Meat of
Diseased Animals.
(1) The flesh of animals which have not been slaughtered should be
prohibited from sale, whether death has resulted from accident or
disease. The meat from diseased animals is also generally
dangerous, sometimes owing to the drugs with which the animals
have been dosed before death, e.g. tartar emetic, or opium.
(2) Meat may be unwholesome from the presence of parasites.
Of these the most common is—
(a) The cysticercus cellulosæ, which is the undeveloped
embryo of the tape-worm; that from the pig becomes the tænia
mediocanellata. The cysticercus of the pig is the most common;
it forms a cyst about the size of a hemp-seed, commonest on the
under surface of the tongue. In hams oval holes are found or
opaque white specks, which are the remains of the cysts converted
into calcareous matter. When meat containing the cysticercus
alive (as in under-cooked or raw meat) is swallowed, it develops
into the tape-worm, which consists of a number of flat segments,
each capable of producing numerous ova of new cysticerci, with
a minute head at the narrow end surrounded by hooklets. A
temperature of 174° F. kills the cysticercus. Another kind of
tape-worm common on the continent, called bothriocephalus latus, is
derived from the cysticercus
of fish.
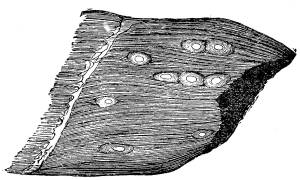
Fig. 2.
Cysticercus (“Measles”) in Pork.
(Natural Size.)
(b) The trichina
spiralis is not a solid
worm like the tænia, but
possesses an intestine.
In pork it forms a minute
white speck, just visible
to the naked eye, which
forms a nest, and in this
one or two coiled up
worms can be seen by a
magnifying glass in active
movement. They are
effectually killed by the
temperature of boiling water; but no form of drying, salting, or even
smoking at a low temperature is sufficient for this purpose. Boiling
or roasting does not suffice to destroy all the trichinæ unless the24
joint is completely cooked in its interior. When trichinous pork is
swallowed, the eggs develop in the alimentary canal in about a week
into complete worms, and in three or four days more each female
produces over a hundred young
ones. These burrow into every
part of the body, producing
great irritation and inflammation.
In one case after death
upwards of 50,000 worms were
estimated to exist in a square
inch of muscle. Most of the
cases of trichinosis have
occurred in Germany, from
eating imperfectly cooked
sausages. The pig becomes
trichinous by eating offal, and man is infected by eating pork.
This disease is rare in England.
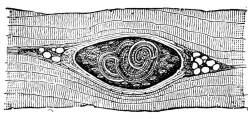
Fig. 3.
Trichinæ Capsulated in Flesh.
Magnified.
(3) Tuberculous Meat, from animals suffering from tuberculosis,
has been found to cause tuberculosis in small animals
experimentally fed on it. Koch has recently thrown doubt on the
communicability of bovine tuberculosis to man; but this point must
be regarded as still unsettled (see page 312). Sheep are rarely affected
by it, but it is very common in cattle, especially in cows, and it is a
serious economical question whether the meat of all such animals
should be condemned. The ideal would be to condemn all such
animals, as tuberculosis is an infective disease, and the bacillus
which causes it (as well as the toxic products of its activity) may be
present in meat which shows no actual signs of disease, except in
the lungs or other internal organs. In practice, however, the rules
laid down by the Royal Commission on Tuberculosis, in 1898,
should be followed for the present. These state that:—
“The entire carcase and all the organs may be seized (a) when there is
miliary tuberculosis of both lungs, (b) when tuberculous lesions are present on
the pleura and peritoneum, or (c) in the muscular system, or in the lymphatic
glands embedded in or between the muscles, or (d) when tuberculous lesions exist
in any part of an emaciated carcase. The carcase, if otherwise healthy, shall
not be condemned, but every part of it containing tuberculous lesions shall be
seized (a) when the lesions are confined to the lungs and the thoracic lymphatic
glands, (b) when the lesions are confined to the liver, (c) or to the pharyngeal
lymphatic glands, or (d) to any combination of the foregoing, but are collectively
small in extent.” They also add that any degree of tuberculosis in the pig
should secure the condemnation of the entire carcase, owing to the greater
tendency to generalisation of tuberculosis in this animal; and that in foreign
meat, seizure should ensue in every case where the pleura has been “stripped.”
(See also page 312.)
(4) Other Infective diseases besides tuberculosis may render
meat wholly or partially unfit for food. Of these pleuro-pneumonia
may not require condemnation of the entire carcase; but in the
following this course should be adopted, cattle-plague, pig typhoid
(pneumo-enteritis), anthrax, and quarter ill, as well as in sheep-pox.
In puerperal fever, actinomycosis, and sheep-rot (liver flukes) each
case must be decided on its merits.
25
II.—Decomposed Meat.—Putrid meat has often produced
diarrhœa and other severe symptoms. Putrid sausages are
especially dangerous, and incipient putridity seems to be more
dangerous than advanced.
Tinned Meats occasionally produce severe illness, which
has been in several cases fatal. It is important to secure a good
brand, and to eat the meat as early as possible after the tin is
opened. Tins in which any bulging is present, showing the presence
of putrefactive gases, must be rejected; and still more tins which
have been pricked and resoldered in a second place. All tinned
meats and fruits are stated by Hehner to contain compounds of tin
in solution. These do not seem to be perceptibly injurious, unlike
lead salts, which are now rarely found.
The general subject of Meat Poisoning has had much light
thrown on it during the last few years. Brieger, about 1886,
showed that during the cultivation of bacteria, alkaloidal bodies
known as ptomaines and leucomaines, were formed, which were
virulently poisonous. It was commonly supposed that the poisoning
occasionally produced by eating meat pies, sausages, hams, brawn,
and similar food, was due to these ptomaines. It is now known,
however, that there are far more important toxines than the
alkaloidal, which result from bacterial life in meat, etc. (see page
286). These are more closely related to substances of an albuminous
or proteid nature than the ptomaines. These toxines may be fatal
when as small a dose as a fraction of a milligramme (mgm. = about
1 ∕ 64 grain) is given subcutaneously. The evidence now shows that
neither ptomaines nor other toxines (albumoses) or any other
bacterial products besides these, cause the outbreaks of acute
poisoning occasionally traced to food, but that these are due to
bacteria. There is, in other words, actual infection, as well as
poisoning. The microbe chiefly found as the cause of these outbreaks
is the Bacillus enteritidis of Gaertner, and some allied microbes. In
an outbreak at Oldham, 160 pies made on a Thursday, from the
veal of a calf killed on the preceding Tuesday, were baked in
several batches, and of the persons eating these pies fifty-four became
ill. That the contamination was not introduced after cooking was
shown by the fact that several persons were made ill who ate pies
still warm from baking. The facts indicated that one batch was
imperfectly cooked, the time allowed being only twenty minutes,
as compared with fifty minutes allowed in corresponding cooking in
domestic life. Experimentally it has been found that an exposure
for one minute to 70° C. kills the Bacillus enteritidis of Gaertner.
That this bacillus was the cause of the outbreak was subsequently
shown by the fact that the serum of blood taken from some of the
patients showed characteristic clumping with a pure culture of this
bacillus, just as happens with the blood of a patient suffering from
enteric fever when a cultivation of the microbe of this fever is mixed
with it (see page 301). In this outbreak the symptoms were usually
diarrhœa, vomiting, intense thirst, desquamation of the skin, and a
slow convalescence, lasting from three to six weeks. (See page 26
for poisoning by Bacillus enteritidis sporogenes.)
26
III.—Meat injuries from the food eaten before killing.—Pheasants
fed on laurel, hares on rhododendron chrysanthemum,
and other animals fed on the lotus, wild cucumber, and wild melon
of Australia, have caused dangerous symptoms.
IV.—Fish, especially some kinds, occasionally produce nettlerash
and other disorders, especially in warm weather. Leprosy has
been ascribed to the eating of decomposing fish, but it occurs in
countries where a fish diet is impossible.
Shell-fish and crustaceans (as lobster, crab) are very prone to
produce evil results. Shell-fish (mollusca), such as mussels,
cockles, and oysters, are dangerous foods. They are generally
grown in estuaries, to which the sewage of towns has access; and
not infrequently cases of enteric (typhoid) fever, as well as
more acute attacks of diarrhœa and vomiting, have been traced to
them. Mussels and cockles are seldom sufficiently cooked to
render them safe; and oysters are eaten raw. They should never
be eaten, unless from personal direct knowledge it is certain that
they have been derived from an estuary in which there was no
possibility of contamination by sewage.
V.—Milk has been a common carrier of disease. Cows eating
the rhus toxicodendron get the “trembles,” and their milk produces
serious gastric irritation in young children. The milk of goats fed
on wild herbs or spurgeworts has produced severe disorders.
The milk of animals suffering from foot-and-mouth disease,
although frequently drunk with impunity, occasionally produces
inflammation of the mouth (aphthous ulceration). The milk derived
from cows fed on grass from sewage farms is, per se, as wholesome
as any other, and its butter has no more tendency to become putrid
than that derived from any other source.
The great dangers in respect to milk are of its becoming mixed
with contaminated water; or of its absorbing foul odours. The
absorptive power of milk for any vapour in its neighbourhood, is
shewn by exposing it in an atmosphere containing a trace of carbolic
acid vapour: the milk speedily tastes of the acid.
Milk also tends to undergo rapid fermentative changes, especially
in warm weather, or when tainted by traces of putrefying
animal matter. Diarrhœa in children is frequently due to such a
condition, or to the rapid decomposition of milk in an imperfectly
cleaned bottle. Milk should always be boiled in warm weather;
and it should never be stored in ill-ventilated larders, or where
there is a possibility of the access of drain effluvia; nor ought it to
be kept in lead or zinc vessels.
Epidemic diarrhœa has been ascribed by Klein to a microbe called the
Bacillus enteritidis sporogenes. This is not killed by heating the liquid containing
it to 80°C. for twelve to fifteen minutes, as is the typhoid bacillus and other non-spore-forming
bacilli. In an outbreak of diarrhœa among the patients in St.
Bartholomew’s Hospital, London, there was strong evidence that this microbe
taken in rice pudding had caused the mischief. Eighty-four patients and two
nurses were attacked, and the patients who had eaten rice pudding were almost
exclusively attacked. A portion of this pudding after being kept twenty-four
hours was found sour and acid. The Bac. enteritidis sporog. was found in it.27
Furthermore it was shewn that the temperature at which the rice puddings
were cooked never exceeded 98°C., whereas the spores of this microbe withstand
100°F. a considerable time.
Very many epidemics of enteric fever and scarlet fever, and a
smaller number of epidemics of diphtheria have been traced to
contaminated milk. Usually in enteric fever the contamination of
the milk was traced to the use of water “for washing the milk-cans,”
derived from specifically polluted sources, and doubtless the
water was the real source of the disease. In most of the milk
outbreaks of scarlet fever, either there was scarlet fever in the dairy,
or persons employed in the dairy were in attendance on patients
suffering from the disease; but in an outbreak connected with a
supply of milk from Hendon, it was suspected that a certain
eruptive disease of the udders of the cow might have been the cause
of scarlet fever in man, without infection from a previous case of the
disease. This point is still sub judice.
Tubercular disease of the intestines and mesenteric glands may
be produced by taking milk derived from tuberculous cows. This
was proved in the case of calves (page 311), and there are strong
reasons for thinking that the same is true for infants, though
doubt has been thrown by Koch on the communicability of bovine
tuberculosis to the human being. The only safe plan is to sterilise
the milk (page 13).
VII.—Vegetable Food (especially greens) is indigestible if
stale, and all mouldy vegetables are dangerous. Over-ripe and
rotten fruit is liable to produce diarrhœa; but the diarrhœa
prevalent in summer is due much less to this than to other
decomposing foods, particularly milk.
Poisonous symptoms have been produced by the admixture of
darnel (lolium temulentum) with flour.
The eating of damaged maize in Italy is the cause of an endemic
skin disease, called pellagra, which commonly proves fatal.
Ergotism is due to the growth on cereals (and most commonly
on the rye) of a poisonous fungus, the claviceps purpurea, which
produces a deep purple deposit on the grain. If bread made from
such flour is eaten for prolonged periods, severe symptoms result;
in some cases, a dry rotting of the limbs. There have been several
epidemics on the continent, due chiefly to eating bad rye bread.
Starvation Diseases.—Simple Starvation causes death in a
period varying with the previous state of nutrition. Usually death
occurs when the body has lost two-fifths of its weight, whether this
be after days, months, or years (Chossat). A supply of water
prolongs the duration of life, to as much as three times what it
would otherwise be. Good nourishment doubles the power of
resisting disease; while deficient food prepares the way for many
diseases. A large share of the decline in the English death-rate
during the last forty years is due to free trade, and the great
cheapening of wholesome food which has resulted from it.
An ill-balanced is more frequent than a deficient diet.
Deficiency of fat is more serious than deficiency of carbohydrates,
and deficiency of proteid is most serious.
28
Scurvy is caused by the absence of fresh vegetables. The use of
the potato and the orange, as well as of lime juice (the juice of
citrus limetta), has led to its extinction among adults in this
country. In former times, it caused more deaths among seamen
than all other causes put together, including the accidents of war.
In infants fed upon tinned foods, whether condensed milk or patent
foods, a form of scurvy still occurs. Infants fed on new-milk never
suffer in this way. If, therefore, it is necessary to feed an infant on
condensed milk for many consecutive months, potato gruel or raw
meat juice or fresh milk must occasionally be given.
Rickets is chiefly due to improper feeding in childhood. The
substitution of artificial foods (most of them containing starch) for
the natural milk is its chief cause. The lower incisor teeth of an
infant appear between the sixth and seventh months. Starchy food
given before this age is undigested. Such food likewise leads to less
fat and proteid being given, which are essential for growth.
Deficiency of lime salts in the food does not cause it, and giving
them in food or medicine will not cure it. Enrichment of the diet
by cream or failing this by cod liver oil is the best means of
preventing and curing it. Abundant fresh air and warm clothing
are also necessary.
Relapsing fever generally follows epidemics of typhus fever, and
is greatly favoured by starvation. Ophthalmia has been chiefly
prevalent in charity schools in which the children are underfed,
though its essential cause is contagion.
Diseases Connected with Over-Feeding.—A fire may go
out for want of fuel, or from becoming choked with ashes; and
it is the latter state of things which occurs in Gout and allied
diseases. Weakness is commonly complained of, but this is due to
excess of food embarrassing vital action; and abstinence and
exercise are required to restore the balance. Excess of nitrogenous
food—especially if combined with the use of sweet, or strong, or
very acid wines, and beer—is particularly prone to produce gout.
In these cases, animal food should only be taken once a day, and
vegetable food should be allowed to preponderate.
Obesity is favoured by excess of starchy food and sugar, and
by copious drinking of water or other beverages. The plan of
curing obesity by restricting oneself almost entirely to meat food is
only advisable, however, under certain conditions. Gall-stones are
favoured by rich foods and excess of sugar; also by alcoholic
indulgence. Dyspepsia is commonly due to loading the stomach
at too frequent intervals; but on the other hand, it not infrequently
leads to the taking of insufficient food, because of the discomfort
produced. The result of this is that a chronic starvation results,
with impaired vital powers. Dyspeptic patients should abstain
from pastry and from tea and coffee, except in small quantities.
Alcohol in any form, as a rule, does harm. Not uncommonly
mastication is imperfectly performed, and a good dentist may cure
the indigestion which has resisted all other treatment.
29
The importance of a duly proportioned and sufficient dietary
is shown by its great influence on health and constitution. An ill-proportioned
or deficient diet is certain to lead to failure of health.
The anatomy of an animal may be modified in the course of
generations by altered diet, as well as its character; thus, the
alimentary canal of the cat has increased in length to adapt it to
its omnivorous habits. In the case of the bee we have a still
more remarkable instance. If by any accident the queen bee dies,
or is lost, the working bees (which are sexually undeveloped) select
two or three eggs, which they hatch in large cells, and then feed
the maggot on a stimulating jelly, different from that supplied to
the other maggots, thus producing a queen bee.
The food of mankind varies naturally with—
I.—Climate. A cold climate leads to increased metabolism,
and consequently a large amount of fatty matter can be eaten
without producing nausea. Witness the difference between a
Laplander’s and a Hindoo’s diet.
The season of the year has likewise some influence. Vital
processes are more active in spring than autumn, and more food is
consequently required in the former season.
II.—Occupation. Although muscular exercise is not associated
with an immediate increase of elimination of urea, yet as a matter
of experience more nitrogenous food is required and can be
metabolised by hard workers than by idlers. The trappers on the
North American prairies can live for weeks together on meat alone,
accompanied by copious draughts of tea. They are constantly in
the open air, undergoing fatiguing exercises, in a dry and rare
atmosphere. For brain workers no special food is required.
Foods containing phosphorus have no special value, so far as is
known, for mental work. Such work, however, is apt to affect
digestion; consequently the digestibility of food is more important
for those engaged in sedentary occupations than its chemical
composition.
III.—Sex. As a rule, women require about one-tenth less
food than men, but probably this rule hardly holds good in the case
of women engaged in laborious work.
IV.—Age. Infants require only milk, and the less they have30
of any other food before a year old the better. Atwater has
calculated that——
- A child under 2 requires 3 ∕ 10 the food of a man doing moderate work.
- A child of 3 to 5 requires 4 ∕ 10 the food of a man doing moderate work.
- A child of 6 to 9 requires 5 ∕ 10 the food of a man doing moderate work.
- A child of 10 to 13 requires 6 ∕ 10 the food of a man doing moderate work.
- A girl of 14 to 16 requires 7 ∕ 10 the food of a man doing moderate work.
- A boy of 14 to 16 requires 8 ∕ 10 the food of a man doing moderate work.
Vital processes are more active in early life, and food is required
not only to carry on the functions of the body, but also to furnish
the materials for growth. Hence, while the proportion of proteids
to carbohydrates and fats should be—
As 1:5.3 in adults, it should be about as 1:4.3 in children.
After the age of thirty-five or forty, the tendency is to take too
much food. All the tissues of the body are established, and excess
of food (especially nitrogenous food) is liable to produce tissue
degeneration by loading the system with partially metabolised
matter, and may lead to gouty diseases. It is much safer to take
what may be regarded as too little than too much food after this
period.
Times for Eating.—The best arrangement seems to be to
have three meals, each fairly nutritious, and containing all the
constituents required. The Romans only had two meals daily,
prandium and cœna. This is common among the French at
present, but it tends to overloading the digestive organs at these
meals.
An ordinary full meal has usually passed from the stomach in
four hours. Fresh food ought never to be introduced before this
period; it is advisable to allow an interval of five hours between
meals for the healthy, so as to give time for the digestive organs to
rest, and for the absorption of food. The practice of taking tea
with the chief meal, or a “meat tea,” is bad. Tea is better taken
an hour or two after food.
Regularity in the time of taking meals is important, as the
digestive organs acquire habits like other parts of the body. Work
ought not, if possible, to be resumed immediately after meals, nor
active exercise of any kind. These tend to abstract blood from the
digestive organs, and so diminish the efficiency of digestion.
Vegetable and Animal Foods.—The fact that the food we
require can be obtained from the vegetable world has led to the
proposition that vegetable food should be taken alone. It is urged
in favour of this plan, that a large amount of suffering to animals
would be prevented. Also that animal food is not so economical
as vegetable, land being more economically employed in producing
corn than in feeding cattle. Thirdly, there is the indubitable fact
that health can be maintained for prolonged periods on vegetable
food (including nuts, cereals, fruits, etc.)
On the other hand, the chief objections to a purely vegetable
diet are that the undigested refuse is greater than with an equal31
quantity of animal food; that a longer time and more exertion than
for animal foods are required in digesting the most nutritious
vegetable foods, such as legumens, while other vegetable foods
do not contain a sufficient proportion of nitrogenous material.
Also, if one lived entirely on vegetable food, a greater bulk would
be required, and owing to the fact that such food is less easily
absorbed, satisfaction to the appetite would not so soon be produced.
Animal food has a great advantage as regards convenience.
Man is not an eating machine; he requires food which is easily
converted into the body substance, and this is supplied by the flesh
of animals, milk and eggs, with a due proportion of non-nitrogenous
food; sheep and oxen work up indigestible vegetable materials into
easily assimilable mutton and beef. The greater convenience of
animal food as a supply of proteid is shown by the following
examples of foods after the removal of water:—
| 100 parts of |
rice |
contain |
7 |
parts of proteid. |
| „ |
wheat |
„ |
16 |
” |
| „ |
pea flour |
„ |
27 |
” |
| „ |
dried lean beef |
„ |
89 |
” |
On the other hand, vegetable foods are a cheaper source, not
only of carbohydrates and fats, but also of proteids as well. Thus
the approximate cost of—
| 1 lb. |
of proteid in |
beef is 2s. 8d. |
| „ |
„ |
milk is 2s. 2d. |
| „ |
„ |
bread is 1s. 6d. |
| „ |
„ |
oatmeal is 7½d. |
| „ |
„ |
peas is 7d. |
Under the ordinary conditions of town life, there is considerable
danger of indulging in an excess of nitrogenous food, and vegetarians
may therefore do good by showing that meat is not
absolutely necessary, and can often with advantage be largely
replaced by vegetable food.
If we include milk, cheese, and eggs in the vegetarian diet, the
objections to it partially disappear; and it would be well if it were
much more widely known, especially among the poor, that on these,
together with vegetables, health can be maintained with the addition
of little or no meat.
The Determination of Diet.—The first principle in making
a dietary is that it must be mixed, containing all the necessary
constituents, proteids, hydrocarbons, carbohydrates, water, and
salts. No one of these alone will support life for any considerable
period. Carbohydrates (sugar and starch) can be most easily
dispensed with; fats, on the other hand, are essential for the
maintenance of health.
The next point is to ascertain the proportion in which these
different foods are required. Salts are commonly taken with other
foods, common salt being the only one taken alone. The amount
required is given on p. 7. The amount of water required varies32
with the season of the year, the amount of exercise and perspiration,
and other factors. As a rule, not more than two pints of water are
required per day, and still less if fruit is freely taken. We may
therefore confine our attention to the carbonaceous and nitrogenous
foods, and try to ascertain the amount of each of these required.
Every diet must be subjected to the following tests, to fully ascertain
its value:—
1. The Chemical Test.—The metabolism undergone by food
in the body being essentially a process of oxidation (though
partially modified and incomplete), the amount of heat yielded on
complete combustion of a food may be taken as a measure of its
value as a source of energy, of which heat and work are convertible
forms. The standard of heat production is the calorie, the amount
of heat required to raise the temperature of one gramme of water
1° C. This is the small calorie. The kilo-calorie (called the
Calorie) is the amount of heat required to raise 1 kilo (1 litre) of
water 1° C., or 1 lb. of water 4° F. In calculations on this basis,
allowance must be made for foods which are incompletely oxidised
in the body. Rubner has shown that the heat value of 1 gramme
(=15½ grains) of each of the chief food stuffs is as follows:—
| Proteid |
4.1 |
Calories. |
| Carbohydrates |
4.1 |
„ |
| Fat |
9.3 |
„ |
The method of applying this standard to a food is as follows: the
percentage of proteid or carbohydrate given in the following table is
multiplied by 4.1, and the percentage of fat by 9.3:—
|
IN 100 PARTS. |
|
WATER. |
ALBUMINATES
OR PROTEIDS. |
FATS. |
CARBO-
HYDRATES. |
SALTS. |
| Uncooked meat with little fat |
74.4 |
20.5 |
3.5 |
— |
1.6 |
| Cooked meat—without loss |
54 |
27.6 |
15.45 |
— |
2.95 |
| Salt beef |
49.1 |
29.6 |
0.2 |
— |
21.0 |
| White fish |
78.0 |
18.1 |
2.9 |
— |
1.0 |
| Bread, white wheaten |
40. |
8. |
1.5 |
49.2 |
1.3 |
| Wheat flour |
15. |
11. |
2. |
70.3 |
1.7 |
| Rice |
10 |
5 |
.8 |
83.2 |
0.5 |
| Oatmeal |
15 |
12.6 |
5.6 |
63.0 |
3. |
| Peas (dry) |
15 |
22 |
2. |
53. |
2.4 |
| Potatoes |
74 |
1.5 |
.1 |
23.4 |
1. |
| Butter |
8 |
2. |
88 |
— |
variable |
Eggs (including shell, for which deduct
10 per cent. |
73.5 |
13.5 |
11.6 |
— |
1 |
| Cheese |
36.8 |
33.5 |
24.3 |
— |
5.4 |
| Milk |
87.0 |
4. |
3.5 |
4.8 |
.7 |
Thus for bread—
| Proteid |
8 × 4.1 = |
32.8 |
| Fat |
1.5 × 9.3 = |
13.95 |
| Carbohydrate |
49.2 × 4.1 = |
201.72 |
|
|
——— |
| Total Caloric value of 100 grammes of bread = |
248.47 |
33
The total fuel value in Calories of one pound of certain typical
foods is given by Hutchison as follows:—Butter 3,577, peas 1,473,
cheese 1,303, bread 1,128, eggs 739, beef 623, potatoes 369, milk 322,
fish (cod) 315, apples 238.
2. The Physiological Test.—Not only is a proper proportion
of proteid, fat, and carbohydrates required, but these must be
capable of digestion and absorption and of oxidation in the body.
Cheese is a highly concentrated food, but its value is less than its
percentage composition would indicate, because of the difficulty of
digesting considerable quantities of it. Green vegetables consist
largely of cellulose, which is only imperfectly capable of absorption
into the blood, although it can experimentally be oxidised by
combustion. The proportion between absorbed food and food
rejected in the fæces can be ascertained by analysis. Many
experiments made on these lines show that on a purely animal diet
(meat, eggs, milk) but little nitrogen is lost, while with vegetable
foods (carrots, potatoes, peas, etc.) the waste of nitrogen is
considerable. Fats are very completely absorbed from the
alimentary canal. The amount remaining unabsorbed is greatest
with mutton fat (10 per cent.), least with butter (2½ per cent.).
Experimentally it has been found that an amount up to 150 grammes
(about 5½ oz.) of fat can be absorbed without appreciable loss.
Carbohydrates are very completely absorbed, even starchy foods
rarely escaping digestion. Completeness of absorption from the
alimentary canal is not desirable for all foods; a certain amount
of unabsorbed residue is required to stimulate peristalsis. With
a purely vegetable diet this amount is excessive, and there is
physiological waste of effort.
3. In practical dietetics the Economic test is important.
Carbohydrate is by far the cheapest food, and generally vegetable
are cheaper than animal foods. Thus a shilling’s-worth of bread
yields 10,764 Calories, while the same sum spent on milk would
only yield 1 ∕ 3, and on beef 1 ∕ 10 this number of heat units. Similarly a
shilling’s-worth of peas contains 572 grammes of proteid, about
double as much as the same money’s-worth of cheese; while to
obtain the same amount of proteid from eggs would cost more than
eight, and from beef more than five times as much as from peas
(Hutchison). The market price of foods is no certain indication
of their nutritive value. Thus haddock will supply as much
nutriment as sole at a fourth of the cost; Dutch as much nutriment
as Stilton cheese at less than half the cost. Similarly the most
economical fats are margarine and dripping.
4. An Examination of Actual Dietaries under various
conditions has strikingly confirmed the results obtained by other
methods. It has been found that (a) the potential energy required by
a healthy man weighing 11 stones, and doing a moderate amount
of muscular work is 3,000 to 3,500 Calories (=310 grains); and that
(b) about 20 grammes of nitrogen and 320 grammes (=4,960 grains)
of carbon are excreted by such a man. (c) Expressing the 3,000
Calories required in terms of grammes of food, it is found that 12534
grammes of proteid, 500 of carbohydrate and 50 of fat are necessary.
These facts are expressed in the following table (Hutchison):—
|
STANDARD AMOUNT OF
FOOD CONSTITUENTS REQUIRED
(IN GRAMMES). |
SAME AMOUNT OF FOOD FOOD IN TERMS OF |
YIELDING ENERGY IN Calories. |
| CARBON. |
NITROGEN. |
| Proteid |
125 |
62 |
20 |
512·5 |
| Fat |
500 |
200 |
─── |
2050· |
| Carbohydrate |
50 |
38 |
─── |
465· |
|
675 |
300 |
20 |
3027·5 |
Three of the best known standard dietaries give the amounts in
grammes of each food constituent as follows:
|
PLAYFAIR. |
MOLESCHOTT. |
ATWATER. |
AVERAGE. |
| Proteid |
119 |
130 |
125 |
125 |
| Fat |
51 |
40 |
125 |
72 |
| Carbohydrate |
531 |
550 |
450 |
510 |
| Calories |
3140 |
3160 |
3520 |
3273 |
Expressing the same facts in English ounces instead of
grammes, 42 ∕ 5 oz. of proteid, 2½ oz. of fat, and 18 oz. of carbohydrate,
would represent the ounces of each constituent required according to
|
(1) |
(2) |
|
AVERAGE OF ABOVE
THREE DIETARIES. |
HUTCHISON. |
| Proteid |
4 ∕ 25 |
4 ∕ 25 |
| Fat |
2 1 ∕ 2 |
1 ∕ 45 |
| Carbohydrate |
18 |
17 ∕ 35 |
| Ounces of dry food |
24 ∕ 910 |
23 ∕ 45 |
The chief point of divergence in the above standard dietaries is
in the relative proportion of carbohydrate and fat. Probably the
correct proportion between these is as 1 to 10; but it will vary
according to climate and other circumstances. Detailed examination
of a large number of dietaries shows that the amount of daily
proteid should be about 125 grammes, or 4⅖ozs. This is contained
in 20 eggs, or in 18 oz. i.e. about 4½ ordinary platesful of cooked
meat.
It must be noted that the 23-24 oz. of food given above as the
standard daily amount represents dry food. This represents 40 oz.
or nearly 3 lbs. of ordinary food.
The following example by Waller, gives a rather liberal35
standard English diet, for a man doing a moderate amount of
muscular work.
| |
|
CARBON. |
NITROGEN. |
| Foundation: |
1 lb. bread |
117 |
5.5 |
|
½ lb. meat |
34 |
7.5 |
|
¼ lb. meat |
84 |
— |
| Accessories: |
1 lb. potatoes |
45 |
1.3 |
|
½ pint milk |
20 |
1.7 |
|
¼ lb. eggs |
15 |
2.0 |
|
⅛ lb. cheese |
20 |
3.0 |
|
|
—- |
—— |
|
Total |
335 |
21 grammes. |
This divided up into meals works out roughly as follows
(Hutchison):—
| Breakfast |
 |
Two slices of thick bread and butter. |
| Two eggs. |
| Dinner |
 |
One plateful of potato soup. |
| A large helping of meat with some fat. |
| Four moderate sized potatoes. |
| One slice of thick bread and butter. |
| Tea |
A glass of milk and two slices of thick bread and butter. |
| Supper |
Two slices of thick bread and butter and 2 oz. of cheese. |
From the preceding data, practical problems as to dietaries are
easily solved. Thus if it be required to find
how much oatmeal, milk, and butter would be required to give a sufficient quantity of
albuminoids, fats, and carbohydrates to an adult male,
the calculation may be based on the figures in the table on
p. 32, or the following figures may, for the sake of convenient
calculation, be taken as representing the percentage amount of
each of these chief food principles contained in the foods named:—
|
ALBUMINOIDS. |
FATS. |
CARBOHYDRATES. |
| Oatmeal |
12 |
6 |
60 |
| Milk |
4 |
3 |
5 |
| Butter |
2 |
88 |
— |
- Let
- o = number of ounces of oatmeal required.
- m = number of ounces of milk required.
- b = number of ounces of butter required.
- Then
- (12o + 4m + 2b ∕ 100 = 4.5 ozs. of albuminoid
- (6o + 3m + 88b)/100 = 3 ozs. of fat
- (60o + 5m)/100 = 14.25 ozs. of carbohydrate,
according to Moleschott’s diet.
When these equations are worked out by substitution and transference—
- o = 19.2 ounces.
- m = 55.4 ounces.
- b = 0.24 ounces.
36
Similarly if it is required to find how much meat, bread, and butter of the following
percentage composition will be required to give a man a sufficient amount of albuminoids,
fats, and carbohydrates.
|
ALBUMINOIDS. |
FATS. |
CARBOHYDRATES. |
| Meat |
25 |
15 |
0 |
| Bread |
8 |
1.5 |
50 |
| Butter |
2 |
88 |
0 |
- Let
- m = number of ounces of meat required.
- b = number of ounces of bread required.
- B = number of ounces of butter required.
- Then
- (12m + 8b + 2B)/100 = 4.5 ozs. of albuminoid
- (15m + 1.5b + 88B)100 = 3 ozs. of fat
- 50b ∕ 100 = 14.25 ozs. of carbohydrates
When these equations are worked out—
- m = 6.28 ounces.
- b = 28.5 ounces.
- B = 1.15 ounces.
Relation of Food to Mechanical Work.—In the body the
movements of every part are constant sources of heat. It is evident
therefore that the potential energy of food can be expressed by (a)
the amount of heat obtained by its complete combustion, or (b) by
the amount of work capable of being obtained from it. Joule
discovered by exact experiment that the mechanical power obtainable
from a given amount of fuel is directly proportional to the
amount of fuel used, being in fact due to the oxidation of this fuel,
the heat produced being transformed into mechanical power. The
heat unit or calorie has been already given (p. 32). The gram-metre
is the work unit. The heat unit corresponds to 425.5 units of work.
Thus the same energy required to heat one gramme of water 1° C.
will raise a weight of 425.5 grammes to the height of 1 metre.
Conversely a weight of 425 grammes if allowed to fall from a height
of 1 metre, will by its concussion produce heat sufficing to raise the
temperature of 1 gramme of water 1° C. In England the amount
of work done is commonly expressed as foot tons, i.e. tons lifted one
foot; while in France it is similarly expressed as kilogrammetres.
Gramme-metres can be converted into foot-pounds by multiplying
them by .007233, and kilogrammetres into foot-tons by dividing by
311.
Frankland estimated that—
| 1 oz. dry albumin |
yields |
174 |
foot-tons |
of potential energy. |
| 1 oz. fat |
„ |
378 |
„ |
„ |
| 1 oz. starch |
„ |
135 |
„ |
„ |
| 1 oz. cane sugar |
„ |
129 |
„ |
„ |
| 1 oz. glucose or lactose |
„ |
122 |
„ |
„ |
In practical dietetics digestibility of food as well as chemical
composition is an important factor. Furthermore metabolism in the
body is not in every instance so complete as oxidation outside it.
Hence estimates of potential energy can only be regarded as37
theoretically correct. Examination questions like the following are
occasionally asked:—
A man does work equal to 176.8 foot-tons in a day. Supposing that he eats
only bread, how much will he require to give the amount of energy required, if
bread contains 8 per cent. proteid, 1.5 per cent. fat, and 49.2 per cent.
carbohydrate?
On the above basis, from 100 ounces of bread the amount of potential
energy obtainable is:—
| 8 × 174 |
= |
1,392 |
foot-tons |
| 1.5 × 378 |
= |
567 |
„ |
| 49.2 × 135 |
= |
6,642 |
„ |
|
|
——— |
| Total energy |
= |
8,601 |
„ |
obtained from 100 ozs. bread. |
Let b = number of ounces of bread required to develop 176.8 foot-tons of
energy.
Then 8,601: 100:: 176.8: b.
Therefore b = 2.05 ounces.
38
CHAPTER VI.
THE PREPARATION AND PRESERVATION OF FOOD.
Objects of Cooking.—Food may be taken in its crude
condition, as directly derived from the animal or vegetable world,
or after it has undergone a preparatory process of cooking. Man is
the only animal who cooks his food. Many foods, in the uncooked
condition are almost entirely incapable of digestion by him—such as
the proteid and farinaceous materials contained in the seeds of cereal
and leguminous plants. But cooking, as a preparatory help to the
digestion of food, is not equally required by all foods. Thus, fruit
is commonly taken uncooked, and does not undergo any important
alteration on cooking. Salads are taken uncooked, but not for their
nutritive properties so much as for a relish to other foods, and for
their quasi-medicinal properties. Milk, again, may be taken cooked
or uncooked. The oyster is the only animal which is eaten
habitually, and by preference, in the uncooked condition; and there
is a physiological reason for this universal custom. The large
fawn-coloured liver, which constitutes the delicacy of the oyster, is
little else than glycogen, associated with its appropriate ferment
diastase, so that the oyster is almost self-digestive. When cooked,
the ferment is destroyed, and digestion of the oyster becomes more
difficult.
Cooking is intended—1. To make the food softer, and in part to
mechanically disintegrate it, thus rendering it more easily masticated
and digested. In fact, cooking, in the best sense, is an artificial
help to digestion; and digestion may well be said to commence in
the kitchen.
2. To produce certain chemical changes. Thus, starch is partially
converted into dextrine; gelatin is formed from connective
tissue, etc.
3. To destroy any noxious parasites present in the food, or
obviate any ill effects from putrefactive changes. Diseased meat
chiefly produces bad effects when imperfectly cooked.
4. To make the food more pleasant to the eye and agreeable to
the palate. The improved savour in cooked meat, for instance,
has a very appetising effect, and consequently makes digestion
easier.
The Cooking of Flesh.—1. Roasting is, perhaps, the most
perfect way of cooking meat. It exalts its flavour more than any
other method. In roasting, place the meat at first sufficiently near
a brisk fire, so that the albumin on its surface may be readily
coagulated, and the juices retained in the interior of the joint. After39
about fifteen minutes, the joint ought to be removed somewhat
further from the fire, and allowed to cook slowly. Frequent basting
is desirable to obtain a good result. Brown meats, such as beef,
mutton, and goose, require a quarter of an hour per pound weight;
veal and pork require about ten minutes additional, to ensure the
absence of redness. White-fleshed birds require a somewhat
shorter time. The time required in roasting will be a little more if
the joint is large, or the fire not very clear. To ascertain if the
meat is sufficiently cooked, press the fleshy part; if it remains
depressed, it is done; if not done, it retains its elasticity. At the
first incision, gravy should flow out of a reddish colour.
The changes undergone during roasting are, that the connective
tissues uniting the muscular fibres is converted by the gradual heat
into gelatin, which is soluble and easily digested; the muscular
fibres, consequently, become more separable, and the myosin of
which they consist is rendered more digestible. The fat is partly
melted out of its fat cells, and partly combines with the alkali from
the blood-serum. Empyreumatic oils (i.e. fat partially burnt),
developed by charring of the surface of the joint, are carried off
when it is roasted in front of the fire; and so, to a large extent, is
acrolein. Acrolein (C₃H₄O) is always produced by the destructive
distillation of neutral fats containing glycerine, and is the cause
of the intolerably pungent odour accompanying the process.
Osmazome, a peculiar extractive matter, on which the flavour
and odour of meat depend, is developed better by roasting than
by any other method of cooking.
It is useful to remember, in buying beef or mutton, that 20 per
cent. must be allowed for bone and 20 to 30 per cent. for the loss
during cooking.
The following figures are by Johnston:
|
IN ROASTING. |
IN BAKING. |
IN BOILING. |
| 4 lb. of |
mutton |
lose in weight |
1 lb. 6 oz. |
1 lb. 4 oz. |
14 oz. |
| „ |
beef |
„ |
1 lb. 5 oz. |
1 lb. 3 oz. |
1 lb. |
Thus roasting is the least economical method of cooking. The
chief loss, however, is of water; the dripping and gravy are
recoverable.
2. Baking of meat in a closed oven does not produce so
agreeable a result as roasting in front of an open fire. The oven
ought always to be very hot before the meat is put in, in order to
rapidly coagulate its surface. Baked meat may have an unpleasant
flavour, owing to its saturation with empyreumatic oils, which escape
in open roasting. The unpleasant flavour can be prevented by
covering the meat with a layer of some non-conducting material, as
a pie-dish or a crust, no empyreuma being then formed. Baked
white of egg, as in the dish of fried ham and eggs, is one of the
most indigestible forms of albumin obtainable.
3. Boiling of meat requires the same time as roasting. If the
flavour and juices are to be retained, the joint ought first to be
plunged into soft boiling water, and then, after three minutes,
allowed to stand aside in water at 170° Fahr. The preliminary40
boiling forms a coating of coagulated albumin over the joint.
Where there is no thermometer to guide the cooking—after the
preliminary boiling for three to five minutes, add three pints of
cold water to each gallon of boiling water, and retain at the same
temperature for the rest of the process, i.e., at about 170° Fahr.
If the meat is boiled in an inner vessel surrounded by water
(water-bath), the temperature of the inner vessel does not rise above
160°-170° F. Ordinary “simmering” means that the meat is kept
all the time at a temperature of 212° F. and is thus spoilt. The
boiling of an egg is an example of the same point. If an egg is kept
in water at a temperature of 170° F. for 10 to 15 minutes, its contents
form a tender jelly, while an egg kept in water at 212° F. for the
same length of time is hard and tough. An egg is more digestible
when cooked in water at 170° F. for 10 minutes than when boiled
in water for 2½ minutes.
The use of soft water for cooking purposes is always advisable;
otherwise a longer period must be allowed. A preliminary boiling
for a few minutes renders hard water softer, and the addition of a
little carbonate of soda has a like effect.
When meat is inserted in water at a temperature below its
boiling point, the juices are gradually extracted, while the meat is
left a mass of indigestible fibres. A good soup is produced, but the
meat is almost valueless. In order that the soups and broths may
be nutritious, the less heat is employed in their preparation the
better. If a soup is strained to make it clear, much of the most
valuable part is removed.
Stewing is a process intermediate between boiling and baking.
It possesses the great advantage over dry baking that no
empyreumatic gases are produced, and there is no charring. The
temperature of the stew-pan ought never to be above 180° Fahr.;
at this heat the roughest and coarsest kinds of meat are made
tender. The only objection to stewing is that the meat becomes
saturated with fat and gravy, and is too rich for weak stomachs. It
is advisable to stew lean meats only.
Hashing is a process of stewing applied to meat which has
been previously cooked. The consequence of this double cooking,
is that the meat becomes tough and leathery. A modified hash in
which the meat is simply well warmed throughout is preferable.
Frying, unless carefully done, renders meat difficult of
digestion, each fibre becoming coated with fat. The art is to “fry
lightly,” that is, to burn quickly and evenly, so that no charring is
produced. Two methods of frying are described. In the first, the
substance to be fried, as an omelette or pancake, is placed with a
little fat or oil in a frying-pan. This is really a modified process of
roasting, the fat merely serving to prevent the object from adhering
to the shallow pan. In the second, the substance to be fried is
immersed in fat; for this purpose a frying kettle is required. Olive
oil or good cotton seed oil is best for use in the frying-kettle. Lard
is a bad material for frying; both it and butter are apt to burn
unless heated slowly. Dripping is a good substance for frying.
The fat used must be heated to from 350° to 390° F., and then the41
substance to be fried, e.g. a sole, plunged into it and left for two or
three minutes. In this process the substance of the sole is really
being steamed by the steam generated in the substance of the sole.
6. Broiling and Grilling are really processes of roasting
applied to small portions of meat. In grilling, it is important that
the gridiron should be hot before putting anything on it. An
external coagulation of albumin is produced, as in good roasting
and boiling.
The Cooking of Mixed Dishes.—A few instances may be
given of common errors in preparing compound dishes. An egg in
a custard, or just coagulated in a poached egg, is a light and easily-digested
food; baked half an hour in a pudding, it is much less
digestible; fried with ham, it is almost as indigestible as leather.
Spices, if mixed with a dish before it is boiled, lose nearly all their
flavouring power, while they remain irritating. They ought to be
added near the end of the cooking process. A soup containing
vegetables, as well as meat juices, should be prepared in two parts.
The vegetables require prolonged boiling; gravy is spoilt by this.
Similarly, the jam in a tartlet, if inserted before baking, loses its
proper fruity flavour; and oysters baked in a beef-steak pie are
indigestible.
The Cooking of Vegetable Foods.—Bread is either
vesiculated or unvesiculated; the latter being what is called
unleavened bread. Vesiculation of bread has usually been produced
by fermentation of some of the sugar of the flour. The starch first
becomes sugar (dextrose) and then the growth of the yeast plant in
the dough splits this up into alcohol and carbonic acid gas. The
carbonic acid percolates the substance of the dough, rendering it
porous. When it has “risen” sufficiently, the dough is placed in
the oven. The heat of the latter kills the yeast plant, thus
preventing any further fermentation, but at the same time expands
the carbonic acid gas in the bread, rendering the latter still more
porous, and drives off in a gaseous condition the greater part of the
alcohol produced by the previous fermentation.
It is objected to this plan of making bread, that a little of the
sugar is wasted in producing alcohol and carbonic acid. To remedy
this, another plan is sometimes adopted, as first proposed by Dr.
Dauglish. In it the dough is charged with carbonic acid dissolved
in water under considerable pressure. The gas escapes in the
substance of the dough, and on baking expands as in the ordinary
method of making bread. Bread made in this manner, is called
“aerated bread.” Nevill’s bread has a solution of carbonate of
ammonia incorporated in the dough, which is dissipated by heat,
thus causing vesiculation of the bread.
On the continent, a mixture of hydrochloric acid and carbonate
of soda is commonly used, carbonic acid and common salt being
formed in the dough. Thus NaHCO₃ + HCl = NaCl + H₂O + CO₂.
The hydrochloric acid employed should be perfectly pure and free
from arsenic. Baking powders are also largely used for making
cakes. “Self-raising” flour is flour with which baking-powder has
already been mixed. Most baking-powders consist of a mixture of42
carbonate of soda and tartaric acid or bitartrate of potash, diluted
with starch. When wetted, carbonic acid gas is evolved. A few
contain alum, which is now an illegal material for this purpose.
Ten pounds of flour ought to make thirteen to fourteen of
bread. The use of stale bread is much more economical than of
newly-made bread; besides this, it is more digestible. Newly-made
bread is more palatable than stale, but it is more cohesive, and does
not crumble into separate particles like stale bread. The consequence
is, that it is less digestible, being less easily penetrated by the saliva
and other digestive juices. The effect of toasting is to render
bread more friable, and consequently more digestible. It ought,
however, to be thin and eaten soon after it is made; when thick
and kept too long, it becomes tough and leathery.
Pastry is less easily digested than ordinary bread. The lard or
dripping added renders it more flaky and less easily pulverised;
and, in addition, the fat coats over the starch cells; and thus the
action of the digestive juices on the pastry is impeded.
Potatoes ought to be boiled in their jackets, or steamed, to
avoid loss of nitrogenous material and salts. Moist heat causes
the starch granules to swell, and ultimately softens and bursts the
cellulose envelopes in which these are contained. Dry heat, as
when potatoes are baked, converts starch into a soluble form, and
ultimately into dextrine (= C₆H10O₅), an intermediate stage towards
the formation of dextrose (i.e. glucose = C₆H12O₆).
Peas and Beans ought to be boiled slowly and for a long
time to render them more digestible. If old, they ought to be
soaked in cold water for twenty-four hours, then crushed, and
stewed. Hard water must be avoided in the cooking of peas and
beans as well as of other vegetables, as the lime-salts form
insoluble compounds with legumin.
Green vegetables require thorough and prolonged cooking.
This renders their tissues softer and more easily attacked in
digestion. The members of the cabbage tribe and carrots can
hardly be boiled too long. Soft water ought always to be used;
this is one reason why steaming is preferable. Before boiling, all
vegetables should be well washed in cold water. A little vinegar
will remove any insects present.
Cooking Apparatus.—The apparatus required in cooking may
be divided into kitchen utensils and cooking ranges.
To ensure good cooking, perfect cleanliness of all apparatus
is indispensable. The use of the frying-pan, gridiron, spit, and
oven has been sufficiently indicated under the description of the
different methods of cooking. The form of stove to be used for
cooking meat is gradually being settled against the old open stove.
Although this secures a somewhat more savoury joint than when
meat is baked, it is extravagant in working. The closed kitchener
in which coal is employed is less economical than a gas stove at the
present price of gas, if the latter is carefully used.
Various appliances for economising fuel have been devised, and at
the same time of allowing of the prolonged action of a moderate degree
of heat. These are usually constructed on the principle of an ordinary43
bath, consisting of a double pan, with a layer of water between the
two compartments. Warren’s cooking-pot belongs to this type.
The Aladdin oven consists of an iron box with an opening above to
let off superfluous steam. This box is surrounded by another
composed of non-conducting material, while a lamp below furnishes
the heat. Dr. Atkinson has calculated that in an ordinary oven
2 lbs. of fuel must be expended for every pound of food cooked,
while in his Aladdin oven 2½ lbs. of fuel will cook 60 lbs. of food.
Time is an important element in cooking. Food is most thoroughly
cooked and most digestible when subjected to a temperature below
that of boiling water for a prolonged period.
The Preservation of Food.—All organic foods tend rapidly
to decompose and putrefy. Putrefaction only occurs when a warm
and moist substance is exposed to the air. The problem of
preserving any food, therefore, may be solved (1) by keeping it at a
very low temperature, (2) by desiccating it, or (3) by boiling or
steaming it so as to destroy any microbes in the food which would
otherwise start putrefaction, and then fastening it in an air-tight
case.
Milk is commonly preserved as condensed milk, and in this
condition is very valuable. A pure condensed milk is now supplied,
prepared without the addition of sugar or any antiseptic, but in
which, as in other condensed milks, all disease-producing or
decomposition-producing microbes have been destroyed during the
process of concentration. Milk may also be desiccated; in this
condition it is difficult of digestion.
In addition to the household methods of preserving fruits, large
quantities of fruits—both moist and dry—are now imported,
protected by syrup or sugar, in sealed canisters; and they retain
the original flavour almost unchanged.
The preservation of meat is effected by—
1. Drying.—This must be done rapidly. It is a process
which is best applicable to fish, but has been applied also to beef.
Dried Hamburg beef is used for making sausages. Pemmican,
largely used by Arctic voyagers, consists of a mixture of meat and
fat, dried and powdered along with some spices; it is generally
eaten with some kind of meal.
2. Cold.—Frozen meat now forms a very large part of the
food of the English people. If the meat has been frozen before
rigor mortis (rigidity after death) has commenced, it keeps well; if
frozen later, it rapidly decomposes after being thawed. Freezing
arrests putrefaction and tends to conceal its odour. Hence the bad
condition of frozen fish may not be detected until it is cooked.
In cooking frozen meat, time should be allowed for thawing to
occur, before the meat is placed in the oven. Much of the
ill-founded prejudice against frozen meat arises from inattention
to this point. Frozen meat is equal in nutritive value to and does
not lose more in cooking than fresh meat.
3. Salting may be done with brine or saltpetre (nitrate of
potassium); the latter does not decolourize the meat like the
former. Salted meats have lost much of their nutritive material,44
in the form of albumin and salts, and the remaining meat is harder
and more difficult of digestion than fresh meat.
4. Immersion in antiseptic liquids or gases, as sulphite of
soda, is objectionable, on account of the addition of extraneous, and
not altogether innocuous, salts. Boric acid powder is largely used
for sprinkling on meat, particularly rabbits, etc., and for preserving
hams and other meats. Its use is to be deprecated. All such
meats should be thoroughly washed with water, before being
cooked.
Solutions of boric acid and borax are frequently added to milk.
Their use is objectionable (a) because they tend to conceal incipient
decomposition, but do not prevent its possible evil effects, and (b)
because they enable the farmer to palm off dirty milk on the public.
Were the addition of preservatives to milk forbidden, the farmer could
perfectly well keep his milk sweet until it reached the town-consumer
by adopting strict measures of cleanliness, and by cooling
his milk before it leaves the farm. At the least it should be made
obligatory on the milk retailer to declare the presence of preservatives
in milk sold by him.
The presence of borax or boric acid can be detected by evaporating the milk
to dryness, incinerating and then moistening the ash with a drop of strong
sulphuric acid. If a little alcohol be now added, on applying a light, a green
flame indicates boric acid. Milk or cream containing boric acid turns blue
litmus paper red.
Formalin is also sometimes used as a preservative for milk in
very weak solution.
Its presence can be determined by diluting the milk with water in a test-tube,
and running strong sulphuric acid down the side of the tube, taking care
to prevent mixing. At the junction of the acid and diluted milk a violet ring is
seen if formalin is present.
Salicylic acid was formerly used as a milk preservative, but is
now seldom used except in beers. All these preservatives are
objectionable in milk, although their injurious action may be
difficult to prove.
5. Coating with fat or gelatine has only succeeded in
conjunction with the exclusion of air. This process is especially
applicable to fishes, as tinned sardines. In a modified form, it is
useful in coating potted meats, etc.
6. Heating and Air-tight Cases.—Tinned meats prepared
according to this method are imported in large quantities. In the
process of preparation, the cases are packed with meat and filled up
with gravy, and then closed with a cover which is hermetically
sealed, except at one point. The case is then heated to 250° Fahr.,
in order to drive out all air, and destroy any putrefactive germs
present. The open point is sealed while the gravy is still boiling,
thus making the case completely air-tight. Albumin is coagulated
at about 170° Fahr.; the higher temperature, which it is found
necessary to employ, overcooks the meat and renders it less
digestible (see also p. 40).
45
CHAPTER VII.
CONDIMENTS AND BEVERAGES.
Condiments, etc.—The name condiment is used in various
senses by different writers. In its strictest sense it is a substance
containing a volatile oil or ether, which may be taken with salt, and
the object of which is to excite the senses of taste and smell, and
consequently produce an appetising effect. This definition excludes
spices, substances allied to condiments, but usually taken with sugar,
as cinnamon, ginger, etc.; also flavouring agents, such as vanilla;
and acids, such as vinegar and lemon-juice. If we use the word in
its widest sense, to include these various groups of substances, we
find that all condiments are taken with the object of improving the
taste or flavour of food, or of assisting its digestion; but that they
are not foods in the sense of supplying any elements towards
building up the body or maintaining its heat. The only partial
exception is lemon-juice, the salts of which have a quasi-medicinal
use.
Taste is usually a compound sensation, the organs of which are
the nerves of taste and smell. True taste is confined to the
appreciation of sensations of bitter and sweet; but the flavour of
meats is nearly entirely appreciated by the sense of smell. This is
shown by the fact that meats appear tasteless and insipid, during
“a cold in the head.” In the appreciation of acid, astringent, and
fiery substances, the sense of touch is also employed. The excitement
of these different nerves results in a stimulus which is carried
up to the central nervous system, and causes by reflex action an
increased flow of the digestive juices. Hot substances, like cayenne
and ginger, also cause an increased flow of gastric juice, by directly
congesting the mucous membrane. This action is not so desirable
as that through the influence of the nervous system. All natural
foods are sapid and possessed of flavour, and thus stimulate
secretion; but any local irritating effect ought to be avoided.
1. Condiments proper comprise chiefly mustard, pepper,
cayenne, garlic, onion, capers, mint, sage, morels, mushrooms,
truffles. The last three on the list are also foods, but are more
commonly used as condiments.
All these act as stimulants to the digestive organs, and in small
quantities aid digestion. The active principle of mustard and
horse-radish is sulphocyanide of allyl. Horse-radish is not so
wholesome as mustard, the scraped root being apt to adhere to the
stomach like the skins of grapes, and produce indigestion. Pepper
contains an acrid resin, a volatile oil, and an alkaloidal substance,46
called piperine. Cayenne contains an analogous substance, called
capsicin. Cayenne, unless in extreme moderation, is harmful, as its
small particles adhere to the mucous membrane of the stomach, and
may set up considerable irritation.
2. Spices are those condiments which contain an aromatic
oil, and which harmonize with sugar. They are, as a rule, less
irritating to the stomach than those of the pepper group.
Cinnamon, cloves, camphor, ginger, and curry powder are the chief
of these. Curry powder really belongs to both the first and second
divisions. When genuine, it is said to contain turmeric, cardamoms,
ginger, allspice, cloves, black pepper, coriander, cayenne, and a few
other substances.
3. Flavouring agents, such as vanilla, lemon peel, and fruit
essences, are used to give a pleasant flavour to various dishes.
4. Acidulous substances are taken chiefly because of their
sharp and agreeable taste. Vinegar is the chief acid employed. It
is produced by the action of a fungus (Mycoderma aceti) on alcoholic
liquids, as wine, or beer, C₂H₅OH (alcohol) becoming C₂H₄O₂
(acetic acid). It is also produced by the destructive distillation of
wood. In small quantities it does not stop digestion, but, by
exciting the nerves of taste, may be of actual service. It helps to
soften the vegetable fibres in a salad; and is also useful for the
same purpose with hard meats, as lobster, etc. In large quantities
it diminishes the power to assimilate food.
Good vinegar ought not to contain less than 3 per cent. of acetic acid; and
sulphuric acid beyond 1 in 1000 in vinegar is to be regarded as an adulteration. A
specific gravity below 1015 indicates the addition of water.
Citric acid and lemon-juice are useful for their refreshing properties,
and the latter also because of its alkaline salts.
Oils, such as olive oil, have been sometimes classed under
condiments, but as they have great nutritive properties, this is
hardly accurate. For the same reason, salt is not classed under
this head.
BEVERAGES.
Water is the universal beverage, and for healthy persons is
preferable to any other. All other beverages necessarily contain it
as their basis.
It will be convenient to consider first aërated and other natural
waters; then tea, coffee, and cocoa; and finally, alcohol.
1. Aerated Waters contain carbonic acid (carbon dioxide) in
solution, which gives to them their characteristic sharp taste and
sparkling character. Thus distilled water charged with gas is sold
as Salutaris or Puralis water. Soda water contains three to five
grains, and medicinal soda water fifteen grains of bicarbonate of
soda to the bottle. Potash water contains fifteen grains of bicarbonate
of potash to the pint, in each case carbonic acid being
dissolved under pressure. In lemonade, ginger-beer, etc., the basis
is sweetened water, rendered tart by the addition of an acid, and47
finally charged with carbonic acid. Lemonade frequently contains
acetic or phosphoric acid instead of citric or tartaric, and ginger-beer
the same constituents with some added tincture of ginger.
Home-made lemonade prepared from fresh lemons is a much more
wholesome drink. Ginger-beer (stone ginger) is produced by the
fermentative action of yeast on a solution containing sugar, bruised
ginger, tartaric acid, and oil of lemon. It usually contains at least
two per cent. of alcohol.
Natural Mineral Waters usually contain common salt
(chloride of sodium) and alkaline salts of soda or lime, and are
impregnated with carbonic acid gas. Apollinaris, Rosbach, and
Johannis possess these characteristics. The carbonic acid in
natural waters is partially combined, and is given off more gradually
than that in artificial mineral waters.
In all the preceding waters there is considerable carbonic acid.
This acts as a sedative to the mucous membrane of the stomach,
and is useful in indigestion. An aërated water added to milk
renders it more digestible by diluting it, and by preventing the
formation in the stomach of a heavy clot of casein. In the making
of artificial aërated waters, it is essential that the water employed
should be pure, that the acid used in generating the carbonic acid
should be free from arsenic or other impurities, and that the water
should not be allowed to come into contact with lead at any stage,
as in pewter fittings. One per cent. of proof spirit is allowed in
temperance beverages by the Excise.
TEA.
Tea is the leaf of an evergreen shrub, the Camellia thea, which
is cultivated in China, Japan, British India, Ceylon, Java, and
other countries. The tea leaves, as seen in this country, uncurl in
hot water. They are lanceolated, with a serrated edge, and the
veins do not extend to the edge of each leaf. By these characteristics
they may be distinguished from foreign leaves, e.g., the sloe
and willow used as adulterants (Fig. 4). The use of old and
exhausted leaves can be detected by a determination of the percentage
of soluble matter dissolved by boiling water from a given
weight of tea. This on evaporation to dryness should be 28 to 30
per cent. of the total weight of the original tea. The presence of
clay, iron dust or other forms of dust is detected by igniting a given
amount of tea and determining the amount of ash. This should be
only about six per cent.
In black tea, the leaves are dried in the sun, rolled and allowed
to become soft and to ferment. During this process, some of the
tannin appears to be converted into less soluble forms. The leaves
are afterwards sun-dried, and these “fired” in a furnace. Green tea
leaves are dried in the fresh condition over wood fires. Indian teas
have more “body” and astringency than China teas. The smallest
and topmost leaves of the tea plant give the finest sort of tea
(Orange Pekoe); next to this comes Pekoe; the next largest leaves48
producing Souchong; after these Congou; while the coarser leaves
nearer the base used to yield Bohea, which is now seldom seen.
Tea consists of three important constituents—volatile oil,
theine or caffeine, and tannin—and soluble and insoluble extractive
matters.
| The |
amount |
of |
caffeine |
varies |
from |
2 to 4 |
per |
cent. |
| „ |
„ |
„ |
tannin |
„ |
„ |
10 to 12 |
„ |
„ |
| „ |
„ |
„ |
volatile |
oil is about |
½ |
„ |
„ |
(1) Volatile Oil gives the aroma and flavour to each particular
tea. It is this which causes the headache, trembling, wakefulness,
and restlessness, occasionally produced by tea, especially by green
tea.

Leaves of
(A) Elder. (B) Tea. (C) Tea. (D) Sloe. (E) Elder.
Fig. 4.
(2) Theine or caffeine, is an alkaloidal crystalline principle.
Its composition is represented by the formula C₈H10N₄O₂, H₂O.
Ceylon tea, broken leaf contains 4·03 per cent., Assam (Indian) tea,
broken leaf 4·02 per cent., while Chinese teas contain from 2·89
(Moyune Gunpowder) to 3·74 (Moning, black leaf) per cent. of
caffeine (Allen).
Theine is the most important constituent of tea and coffee. It
is a stimulant, but unlike alcohol, acts even more upon the central
nervous system than upon the heart. It removes the sense of
fatigue, and may, especially if taken in excessive doses, produce
sleeplessness. Its stimulant action on the heart is followed by
increased flow of urine, and it thus helps in the removal of waste
products from the system. The effect on the tissue-changes of the49
body is somewhat doubtful. It has been stated to arrest or
diminish the waste, i.e., the metabolism, constantly going on in the
system, and so diminish the amount of food required to repair this
waste. This is highly improbable; we cannot conceive the
likelihood of the development of energy without a corresponding
expenditure of material, and that is what would be the case if
theine increased the activity of various organs while retarding their
waste. The experiments of Conty and Guimarès on the action of
coffee show that this (and tea has the same essential constituent)
does not diminish tissue waste. It does not prolong life in starvation,
though it may lessen the feeling of hunger. Hence tea and
coffee, which owe their value mainly to the caffeine or theine
contained in them, are in no sense foods.
(3) The amount of Tannin varies from 12·31 in Ceylon tea
(broken leaf, Pekoe) to 11·76 in Moning, black leaf, and 9·9 per
cent. in Natal Pekoe Souchong (Allen). The difference in tannin
between Chinese and Indian teas is not therefore so great as is
usually supposed. Tannin is a powerful astringent, and possesses
a bitter styptic taste, and a constipating effect on the bowels. Its
amount is increased by long “brewing,” as is shown by the
following results (Hale White):—
|
Three Minutes’
Infusion. |
Fifteen Minutes’
Infusion. |
| Finest Assam |
11·30 per cent. |
17·73 per cent. |
| Finest China |
7·77 per cent. |
7·97 per cent. |
| Common Congou |
9·37 per cent. |
11·15per cent. |
The Mode of Preparation of Tea is important. It is clear
that the percentage of tannin to weight of leaf used in making the
infusion increases with the protraction of the infusion. On the
other hand caffeine is so soluble that it is nearly completely dissolved
as soon as infusion has begun. Dittmann found that five
minutes infusion of Indian tea extracted 3·63 and ten minutes
infusion 3·73 per cent. of caffeine. The Chinese put the tea leaves
in a cup, and having poured boiling water on them, drink the
resulting infusion after a very short time, without adding anything.
The Russians drink the infusion with a squeeze of lemon, and with
or without sugar. We add cream or milk and generally sugar,
and so render it more nutritious, though the delicate flavour is
veiled. The Chinese plan of infusion for a short time is the best,
as it ensures the extraction of the aromatic and stimulant principles
of the tea with only a proportion of the tannin.
In making tea it is important to use a tea-pot which is quite
dry, in order to avoid mustiness; to pour a small quantity of
boiling water into the tea-pot and then out again, so that the
infusion may be made at the temperature of boiling water; and to
use water which has only freshly come to the boil, and so has not
been rendered flat, and not to infuse longer than five minutes. For
persons of weak digestion, the best kind of tea is that obtained by
pouring boiling water on the leaves, and then immediately pouring
the resulting infusion into another hot tea-pot. In all cases where50
tea has to be kept a considerable time, it should be poured into a
second tea-pot, the leaves being left behind.
Indigestion is not an uncommon consequence of tea-drinking;
caused by the excess of tannin in the tea, by the other constituents
of the tea, or more commonly by the practice of drinking tea in
small sips, with bread and butter. The tea infusion usurps the
place of the saliva, the secretion of saliva remaining partially in
abeyance. The presence of tannin in tea renders it an undesirable
part of a substantial meal. Tannin coagulates albumin, and
retards its solution by the digestive juices. Hence “high teas”
and “tea-dinners,” unless the tea is very weak, are objectionable.
The practice of drinking tea with every meal is inexcusable.
For quenching thirst during active exercise, and rendering
possible prolonged exertions, tea is unsurpassed.
COFFEE.
Coffee is the seed of the berry of the Caffea Arabica. Each
berry contains two seeds, or beans as they are sometimes
incorrectly called. The coffee is prepared by roasting the seeds
until they assume a reddish-brown colour, in which process they
lose 15 per cent. in weight and gain 30 per cent. in bulk. During
the process of roasting, a volatile oil having a powerful aromatic
smell is developed. This is not produced in such large quantities
from fresh seeds; the best time for roasting varying, however, for
different varieties of coffee.
The amount of Volatile Oil in coffee is much less than in tea.
As it is elicited during the process of roasting, this should be done
with nicety and care. It is effected in an iron cylinder made to
revolve over a fire. After the roasting, the sooner the seeds are
ground the better the coffee. When it cannot be immediately used,
it should be kept in closed canisters, and not in paper or open jars.
In addition to the volatile oil, which is contained in roasted
coffee in the proportion of about 1 part in 50,000, coffee contains
caffeine, of which there is ¾ to 1 per cent., and an astringent
acid, called caffeo-tannic or caffeic acid, which differs from ordinary
tannin in that it does not blacken a solution of an iron salt.
The chief adulteration of coffee is Chicory, which is thought
by some to improve the coffee. It is generally harmless, though in
some people it produces heartburn and diarrhœa. Chicory is
prepared from the root of the wild endive. It contains a volatile
oil and a bitter principle, but no caffeine. It is, therefore, of no
utility as a stimulant. Its presence can be detected by shaking a
little of the suspected coffee on to the surface of the water in
a wine-glassful of cold water. Coffee swims on the surface,
and gives little or no colouration to the water; while chicory
sinks, and gives a deep red tint. The aqueous extract of pure
coffee (extracted by boiling water) is, when evaporated, 25 to 30
per cent. of the weight of the original decoction of coffee; while
that of chicory is 65 to 70 per cent.; and on this basis, as well as
on the fact that a filtered decoction of 10 grammes of coffee in 100
c.c. of distilled water, cooled to 60° F. has a specific gravity of 1009,51
while that of a similar solution of chicory would be 1021, the
proportion of chicory in a mixture of coffee and chicory can be
calculated. The microscopical appearances of the two powders
differ, coffee showing hexagonal cells and no laticiferous vessels,
unlike chicory. There is no law against selling mixed coffee and
chicory, if the fact that it is a mixture is stated; and the
proportion of the two unfortunately is not required to be stated.
As a pound of coffee costs five times as much as a pound of chicory,
it is obviously to the purchaser’s advantage to make his own
mixture in the proportions desired.
The Preparation of Coffee ought to be effected as in the case
of tea—by making an infusion and not a decoction, i.e. by pouring
boiling water on the coffee and allowing it to stand, but not
continuing the boiling. Continuance of boiling dissipates the
delicate aroma.
Inasmuch as coffee contains a much smaller percentage of
theine than tea, more of the former must be used to obtain a
beverage equally refreshing with tea. Two ounces to a pint of
boiling water are required. The infusion thus made should be
mixed with an equal part of boiled milk. The coffee ought, if
possible, to be freshly roasted.
The colour of coffee is no guide to its strength. Many of the
black coffees, especially “French coffee,” owe their colour to the
caramel (burnt sugar) contained in the chicory mixed with them.
Coffee has similar properties to tea, with some minor differences.
(1) Like tea, it is restorative and sustaining in its action, but seems
to act more quickly than tea. (2) Unlike tea, it does not tend to
produce perspiration, but rather a dry hot skin. (3) With some it
is decidedly laxative; while tea, especially if badly made, has an
opposite effect; but this is not always true. (4) It seems to have
a greater power of antagonising the effects of alcohol than tea; and
is a valuable antidote, after the action of an emetic, in poisoning by
opium or arsenic or alcohol.
As a rule, coffee is not so prone to disorder the digestion as
tea, but this is not universally true, and in some persons it always
produces “biliousness.” When taken in excess, it produces—besides
indigestion—palpitation, restlessness, irritability, sleeplessness,
and a condition of general nervous prostration; in fact,
similar symptoms to those produced by a prolonged over-indulgence
in tea.
While the consumption of tea is rapidly on the increase, that
of coffee is steadily diminishing. This is partly owing to the
greater expense of coffee—a larger quantity being required to form
a good beverage; and partly to the greater difficulty in preparing
good coffee.
COCOA.
Cocoa, or more properly cacao, is obtained from the seeds
of the Theobroma Cacao—a native of the West Indies, Mexico,
and the central parts of America. Its name Theobroma was given
it by Linnæus, and means the “food of gods.” The fruit is a large52
leathery capsule, having nearly the form of a cucumber. It contains
from 25 to 30 seeds, each about the size of an almond. Before
using, these are roasted like coffee berries, and a peculiar aroma is
developed in this process as in the case of coffee. The beans or
seeds are then manufactured into three different products. (1)
They are simply deprived of their husks and broken to pieces; this
forms Cocoa-Nibs. (2) They are ground, husk and all, between
hot rollers into a paste, and mixed with starch and sugar; this
forms Cocoa. (3) They are shelled and then ground into a paste,
as in making cocoa; sugar and some seasoning, usually vanilla,
being subsequently thoroughly mixed; this paste is Chocolate.
The purest form is the cocoa-nibs. When these are boiled in
water, a brownish decoction is formed, with the fat as a scum at
the top; this may be removed, and the decoction flavoured with
milk and sugar. In this form, cocoa can be taken by invalids with
weak digestion, who would be nauseated by the fat of ordinary cocoa
or chocolate.
The best cocoa is prepared as above; but the lowest quality
contains the husks of the beans, with hardly any of the beans in it;
a somewhat better, though still inferior sort, is made from the
smaller fragments of the nibs, and a good deal of husk. In some
cases the cacao butter is removed during the process of preparation,
and starch or sugar substituted. This form is less likely to disagree
with dyspeptics than whole cocoa.
The action of the Volatile Oil (not the cacao-butter) developed
during roasting, is probably similar to that of tea and coffee,
though it is less in amount. The bitterness is greater than that of
coffee, but the astringency less than in either tea or coffee.
The Concrete Oil, or fat of cocoa, forms about half its weight.
It is white, and not apt to turn rancid, and possesses an agreeable
flavour. Cocoa also contains a certain amount of starch and cellulose.
Theobromine is a white crystalline alkaloid, the exact
analogue of caffeine. The latter, in fact, is methyl-theobromine—that
is, theobromine plus the theoretical group CH₂. Theobromine
possesses similar properties to caffeine. It amounts to 1.5 to 2 per
cent. of the whole bean. The ordinary preparations of cocoa differ
considerably in composition as may be seen from the following
table of per centage composition (Ewell). In each instance other
nitrogenous and non-nitrogenous constituents go to make up the
total 100:
|
FAT. |
FIBRE. |
CANE-SUGAR. |
ASH. |
ADDED STARCH. |
| Fry’s Cocoa Extract |
30·9 |
3·9 |
— |
4·2 |
None. |
| Schweitzer’s Cocoatina |
31·1 |
3·7 |
— |
6·3 |
Do. |
| Rowntree’s Cocoa Extract |
27·6 |
4·4 |
— |
8·5 |
Do. |
| Van Houten’s Cocoa |
29·8 |
4·4 |
— |
8·6 |
Do. |
| Epps’s Prepared Cocoa |
25·9 |
1·5 |
26 |
3·1 |
Much arrowroot. |
Some of the preparations of cocoa (e.g. Van Houten’s) have
added to them alkaline salts to increase their solubility. Cocoa is
not such a valuable food as might appear from the large amount of53
fat in it, because only moderate quantities of this can be taken
without deranging digestion. In Vi-Cocoa a certain amount of kola
is added, which contains a considerable proportion of caffeine.
The addition of such a drug to a beverage is distinctly to be
deprecated.
Minor Stimulants.—Beverages containing theine, or some
analogous principle, appear to be employed in most countries. In
moderate doses, they may assist the assimilation of other foods, but
their main influence is on the nervous system. Theine-containing
substances may be described as both sedative and exciting. They
are sedative, in that they allay nervous irritability, and tend to
“take the edge off” the disturbance caused by outward circumstances;
and they are exciting, inasmuch as they are known to form
an admirable antidote to the stupefying effects of opium or alcohol.
The wakefulness from tea is an instance of the same thing, while
the allaying of sensations of cold and hunger by a cup of tea is an
instance of the sedative effect.
In Brazil, Guarana (from Paullinia sorbilis) is used as a drink; it contains
theine, the quantity of which is twice as much as in good black tea, and five
times as much as in coffee. Like green tea, a cup of guarana infusion is sometimes
extremely valuable in nervous headaches.
In Peru, the natives use the leaves of the Coca plant (Erythroxylon coca),
which must be carefully distinguished from cocoa. It is chewed somewhat in the
same way as the betel-nut. It contains two alkaloids—cocaine and hygrine, as
well as tannin. In its stimulant action it resembles tea and coffee. The active
principle of this plant, Cocaine, is a valuable local anæsthetic. Internally it
has been taken as a stimulant and restorative. Various wines containing Coca,
with vaunted restorative powers, are advertised. They are mischievous when
taken frequently. Nature’s remedy for fatigue, whether mental or body, is rest
and recreation. Stimulants of this class, even though they enable work to be
continued for awhile, eventually increase the exhaustion for which they are
taken.
The Kola-nut is used in some parts of Western Africa as a stimulant. It
is about the size of a pigeon’s egg, and has a bitter taste. The natives of Guinea
generally take a piece of the seeds before each meal, and sometimes nibble it
throughout the day.
Kava is prepared from the root of a kind of pepper. The natives of the
Fiji islands commonly indulge in it. Its effects resemble those of coffee. In
large doses, it destroys the power of walking, and may possibly produce impairment
of vision.
The leaves of the Ilex Paraguayense, Ilex Gongorrha, and Ilex Theezans are
made into the beverage commonly known as Paraguay tea or maté.
The leaves of the Hydrangea Thunbergii are made into a beverage,
which is designated in Japan “the tea of heaven.”
Among certain nations of Asia, the Betel-nut (from a palm called Areca
Catechu) is chewed, after mixing small fragments with pepper and quicklime,
and rolling in a palm leaf. The saliva is tinged blood-red, and a narcotic effect
is said to be produced.
The dried flowering tops of the Indian Hemp (Cannabis Indica) are smoked
by the Malays and others, or made into a beverage, called haschisch, which
produces a kind of intoxication, in which murder has often been committed
(hence, assassins equals haschascheens).
The Kamtschatkans drink an infusion made from a fungus, known as the
Fly Agaric (Amanita Muscaria), thus producing an intoxication similar to
that from haschisch.
Opium in small doses is a stimulant, in large doses narcotic. The crude
drug is sometimes taken, and less frequently the active principle, Morphia. It
is frequently smoked, as well as taken internally. It is to be feared that secret54
opium taking is considerably increasing. The taking of morphia, especially
hypodermically, is too common. Generally it has been first prescribed for
neuralgia or some other complaint causing acute pain; and the patient, having
experienced relief by its means, is tempted to revert to the practice apart from
medical advice. Such a line of action is most pernicious. Eventually both
the physical and the moral nature of the victim are shattered by it; and to break
off this insidious habit, when once thoroughly established, is most difficult.
Tobacco may be conveniently mentioned here, though its usual effects are
certainly not stimulant. It is smoked, chewed, or taken as snuff; when indulged
in to excess it produces serious depression of the heart’s action, with frequent
intermittence. In moderate doses it is sedative as well as slightly laxative.
Prolonged indulgence in tobacco has produced many cases of incomplete
blindness (tobacco amblyopia), in some cases it comes on with much smaller
doses, and in all cases is only curable by ceasing to smoke. There is no
sufficient ground for the statement that cigarette smoking is more injurious
than smoking tobacco in a pipe or cigar, unless in the former case the smoke
is inhaled into the lungs. The practice of smoking is injurious to growing boys,
and should be strictly forbidden.
Other Drugs are now not infrequently taken, apart from medical advice.
Of these the most commonly used are Antipyrin and Phenacetin, for
headaches. Their use is injurious, and should not be entertained as a frequent
practice. Sleeplessness frequently leads to the practice of taking chloral or
sulphonal, or occasionally the inhalation of chloroform to induce sleep. (See
also page 259). Remedies to induce sleep should never be taken except under
immediate medical advice. They are only justifiable in extreme conditions,
and if frequently taken tend to aggravate the conditions for which they are
given.
55
Properties of Alcohol.—When a saccharine solution is
subjected to the influence of warmth and moisture, and exposed to
the air, it rapidly undergoes a process of fermentation. The most
favourable temperature is about 70° Fahr. The ferment or agent
exciting the change in the sugar is derived from the atmosphere;
it is a minute fungus (torula cerevisiæ), the spores of which are
constantly floating in the air. When once fermentation has started,
exposure in the air is no longer necessary; the process continues in
closed vessels. The essential change occurring in the vinous
fermentation is that grape sugar (C₆H12O₆,H₂O) becomes split up
into alcohol (C₂H₅OH) and carbonic acid (CO₂). Thus—
|
C₂H₆O |
 |
Two of alcohol |
|
C₂H₆O |
| C₆C₆H₁₂O₆ = |
|
CO₂ |
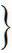 |
Two of carbonic acid. |
|
CO₂ |
There are other fermentations allied to the vinous. Thus the
Acetous fermentation results in the conversion of alcohol into
vinegar, as in the souring of beer or wine. The Lactic fermentation
leads to the conversion of milk-sugar into lactic acid, with consequent
souring of the milk.
Alcohol, or more correctly ethylic alcohol, is a colourless
liquid, having a pleasant vinous odour, and evaporating rapidly on
exposure to air. It burns with a bluish sootless flame, and is a
capital solvent for resins and other substances.
Rectified spirit is absolute alcohol mixed with 16 per cent. of
water. Proof-spirit is a mixture of 42·7 per cent. by volume of
absolute alcohol, and 57·3 per cent. of water. Thus the ratio of
alcohol to proof-spirit being as 1: 1·76, the amount of alcohol in
any liquid being given, the amount of proof-spirit can readily be
calculated. The fermented drinks containing alcohol may be
classed as (1) malt liquors, (2) wines, and (3) distilled spirits. The
relative properties of these will be considered afterwards; in the
next two sections will be considered the effects of diluted alcohol in
whatever form it is taken.
Effects of Moderate Doses of Alcohol on the System.—In
studying the physiological effects of alcohol, one has to guard
against the fallacy that these are the same, only differing in degree,
whatever the dose may be. The effects of large doses of alcohol are
almost exactly the reverse of those produced by small doses. It56
will be necessary to define, therefore, what we mean by a moderate
dose. By a moderate dose, we understand the amount of alcohol
which can be taken without any alcohol being eliminated in the
urine. Dr. Anstie found that 1½ ounces, that is three tablespoonsful,
of absolute alcohol, taken in twenty-four hours, caused
its appearance in the urine; and Dr. Parkes and Count Wollowicz
obtained almost precisely the same result. Anything below some
quantity between 1 and 1½ fluid ounces per day can be disposed of
in the system, and is probably oxidised like ordinary foods.
The amount of alcohol, in the form of alcoholic beverages,
corresponding to this maximum dose of absolute alcohol is
approximately as follows:—
| One imperial pint |
(20 fluid ounces) |
of bottled beer |
(5 per cent. of alcohol). |
| One tumblerful |
(10 fluid ounces) |
of claret, hock, and
and other weaker wines |
(10 „ ). |
| 2½ glasses |
(5 fluid ounces) |
of port, sherry, and
other strong wines |
(20 „ ). |
| One glass |
(2 fluid ounces) |
of brandy or whiskey |
(50 „ ). |
It will be understood, therefore, that in describing the effects
of a moderate amount of alcohol on the system, an amount below
1½ ounces of absolute alcohol per day is meant, freely diluted, and
taken as a rule with meals.
1. Effect on the Stomach.—In very small quantities, alcohol
seems to stimulate digestion in the same way as mustard. But
like all other artificial helps to digestion, it is best avoided in the
healthy condition.
2. The Effect on the Liver is similar to that on the stomach—a
temporary redness and congestion being produced; this effect
soon disappearing if the dose is small and well diluted. But in all
cases where there is a tendency to biliousness, even small doses of
alcohol are injurious.
3. The Effect on the Heart and Blood-Vessels is first to
increase the force of the heart’s action and the rapidity of the
pulse. The stimulation of the heart is rapidly followed by a
universal dilatation of the small arteries of the body, which diminishes
the blood-pressure. Parkes and Wollowicz found that the daily
administration of from 1 to 7½ ounces of rectified spirit raised the
pulse rate by ten beats per minute, as compared with other periods;
and that this effect was followed by a period of depression in which
the beat was both slower and feebler than usual.
4. The Effect on the Nervous System varies. In persons
unaccustomed to its effects, even small doses dull the power of
thought and the rapidity of perception, owing to the paralyzing
effect which it exerts on nerve cells. In most cases, however,
it at first produces increased rapidity of thought and excites the
imagination, though even here it makes it more difficult to keep to
one train of thought. This is clearly owing to the more rapid
circulation of blood through the brain. Dr. E. Smith’s experiments
show that it diminishes the acuteness of the senses. Its
influence even in dietetic doses, on the capacity for mental work,
is slightly to diminish it.
57
5. The Effect on the Muscular System is never beneficial.
Even when only small quantities are taken, the power of controlling
delicate movements is slightly diminished. For persons engaged
in laborious occupations, a small quantity does not produce much
apparent effect, but where the quantity exceeds two fluid ounces per
day the capacity for strong and sustained muscular work is
manifestly lessened (Parkes). This effect is probably due partly to
the dulling of the nervous system, rendering the muscles less
amenable to the will, and partly to the over-excitation of the heart
causing palpitation and breathlessness.
6. The Effect on Metabolism is to diminish it, thus favouring
the deposit of fat in the tissues. It acts as a poison to the protoplasm
of the cells of the body, diminishing their power to break
down the floating nutriment, especially fat and carbohydrate.
The Effect on the Temperature is to lower it; but unless
the dose is excessive, this effect is hardly appreciable. The
resistance to excessive cold is diminished by even moderate doses of
alcohol, still more by large doses. In the Arctic regions, this has
been abundantly proved. This effect is produced, notwithstanding
the fact that alcohol becomes oxidised in the system. The dilatation
of the surface blood vessels leads to a greater loss of heat than that
produced by the oxidation of the alcohol.
Effects of Immoderate Doses of Alcohol on the System.—Bearing
in mind the definition given of a moderate dose, one is
bound to admit that a large number of individuals exceed this
amount daily, apparently without any very serious results. The
system becomes habituated to large doses, and if the occupation is
a laborious one, they may in part be oxidised in the system. Such,
however, are exceptional cases. In the majority of cases evil
results are by no means confined to those who indulge in very large
quantities of alcohol at varying intervals. In fact these very often
escape comparatively free, while others who never take a quantity
sufficient to incapacitate them for their work, are sowing the seeds
of chronic and oft incurable disease. The labourer who has a
drinking bout at intervals is thoroughly nauseated; and the
condition of liver and stomach induced, enforces abstinence on him
for a time sufficient to bring his organs back to a normal condition;
while the city merchant who indulges more moderately, but whose
organs are almost continuously impregnated with alcohol, becomes
gouty and prematurely old.
The Stomach may become acutely inflamed, when a large dose
of alcohol is taken. The chronic irritation of alcohol, especially
when taken apart from meals, causes atrophy of the walls of the
stomach, and a change analogous to that in the liver.
The Liver, when alcohol is daily taken immoderately, becomes
seriously diseased. In some cases it becomes large and fatty; in
others the chronic irritation excites an overgrowth of fibrous tissue
between the lobules of the liver, which, gradually shrinking, squeezes
the liver cells and causes them to atrophy, at the same time
obstructing the small branches of the portal vein in the substance
of the liver. The consequence of this obstruction to the flow of58
blood through the liver is that all the organs from which the portal
vein brings blood become overloaded with blood, and vomiting of
blood and dropsy of the abdomen occur at a later period.
The Lungs are irritated to a less extent by alcohol in large
doses. The tendency to chronic bronchitis is increased, followed
by emphysema, and sometimes an overgrowth of fibrous tissue
(cirrhosis) like that in the liver occurs.
The Heart and Blood-vessels tend to become diseased, owing
largely to the gouty condition of system developed.
The powers of Metabolism are diminished. Corpulence is,
consequently, a common result of alcoholism. There may also be
fatty deposit in the internal organs, such as the heart. This must
not, however, be confounded with a much more serious condition,
fatty degeneration of the heart, in which the substance of the
muscular fibres becomes partially converted into fat, and which
also is sometimes due to alcoholism.
The Nervous System is more prone to suffer in chronic
alcoholism than any other part of the body, except perhaps the
liver. The first effect of a large dose of alcohol is to stimulate the
nervous system, as already described. This is followed by a dulling
of the nervous faculties, which comes on rapidly in proportion to
the amount taken. The phenomena of intoxication are unhappily too
familiar to require description, mental incoherence and muscular
incoordination (lack of control over the muscles) being the most
prominent features.
When the dose of alcohol is still larger, a condition of profound
unconsciousness is produced (coma), which may be difficult to
distinguish from other forms of unconsciousness.
Delirium Tremens is another nervous condition, which may
rarely follow a single debauch, but much more commonly affects
the chronic toper. In some cases the immediate exciting cause is
a mental shock, or lack of food, or a surgical injury. Alcoholic
subjects suffering from any acute disease are liable to this form of
delirium, and their chance of recovery is greatly diminished.
Insanity of a more prolonged character than that characterising
delirium tremens is an occasional result of alcoholism.
Besides the nervous diseases already named, a chronic
thickening of the membranes covering the brain and spinal cord,
gradually progressing and finally fatal, is often the consequence
of prolonged alcoholic indulgence.
Various Degenerative Diseases are produced by alcohol.
It has been well called by Dickinson the very “genius of
degeneration.” Such degenerations are by no means confined to
the intemperate; they are seen in those who are of what would
usually be considered moderate habits. The stomach, liver, lungs,
and probably the kidneys, are the main organs to suffer in this
way. It is probable that the effect on the kidneys only occurs
when a gouty condition is developed. In all these cases there is
an overgrowth of fibrous tissue, with atrophy of the proper gland
structures.
59
Gout is the common nemesis of those indulging in alcoholic
beverages, more especially wine and beer, due to the excessive
formation or retention of urate of soda in the body. This produces
inflammation of the joints, and other evils—among them the gouty
kidney, named above, which is always ultimately fatal. Rigid
arteries are likewise commonly due to alcoholism and gout. If
one of these bursts in the brain, apoplexy results.
Longevity is diminished by immoderate indulgence in alcohol.
The statistics of Temperance Insurance Societies, show much
better results among teetotalers than among moderate drinkers.
It is only fair to add that although the latter are supposed to
consist of moderate drinkers—and particular enquiries are always
made on this point before insurance—it is probable that a large
proportion of them exceed 1½ ounces of alcohol per day. Making
due allowance for this fact, the statistics show a great superiority
in the expectation of life of teetotalers.
Factors Modifying the Effects of Alcohol.—1. Age
and Sex.—Until adult life is reached, total abstinence from alcohol
should be enforced. The delicate nervous system of children is
easily disturbed by it, and it appears in some measure to retard
growth. Another argument against giving alcohol before adult
age is reached, is still more important. It is at this period of life
that habits are chiefly formed, and a craving for alcohol may be
insidiously produced, destined to have most baneful results.
Old people, if ordered spirits for medical reasons, should drink
them well diluted.
Women are much more easily affected by alcohol than men,
and if they acquire the habit of excess, the hope of reformation
is even less than with men.
2. Exercise has a most important influence in modifying the
effects of alcohol. Those of sedentary occupations and living in
towns, cannot oxidise as much as those engaged in active out-door
work, and are consequently much more prone to suffer. A game-keeper
in the Scotch Highlands may possibly live to a good old
age, notwithstanding the fact that he consumes an amount of
whiskey that would have sent a sedentary man to his grave in
the course of a few years.
3. The Condition of the Stomach has also great influence.
When the stomach is empty, alcohol produces at once a powerful
reflex stimulation of the heart, and becomes quickly absorbed into
the circulation. Thus intoxication may be produced by a quantity
that would have had little effect if taken with a meal.
4. The State of Concentration or Dilution modifies greatly
the action of alcohol, the local action on the stomach and the
reflex stimulation being much greater than when it is concentrated,
and injurious effects being much more likely to occur.
5. Cold and Heat modify the action of alcohol. A smaller
quantity of hot spirits and water will intoxicate than of cold; the
heat stimulating the heart, and so making the absorption of the
alcohol more rapid. A glass of hot spirits and water will often
cause sleep, by drawing the blood towards the abdominal organs.60
The fact that persons, who have been drinking spirits in a warm
room, on going out into the cold air become suddenly intoxicated,
seems opposed to what has been already said. But probably this
is due to the cold causing contraction of the arteries of the skin,
and so driving more of the blood loaded with alcohol to the
internal organs and the brain (Brunton).
6. Mental Occupation has some influence in modifying the
effects of alcohol. Topers have found that if they try to converse
during their debauch—the conversation implying increased
functional activity of the brain, and therefore a freer circulation
of blood in it—intoxication occurs much more readily, than when
the mind is not active.
7. Disease modifies greatly the effects of alcohol. In some
diseases, as in inflammation of the lungs and in fevers, it can be
given in large quantities without producing intoxication; and in
these conditions it lowers the temperature. In other diseases,
especially gout and kidney disease, its use is nearly always followed
by bad results.
The Advisability of Alcohol as an Article of Diet in
Health.—In dealing with this difficult point, two sets of facts
require consideration, those obtained as the result of Physiological
observations (see page 56), and those which are the result of
Experience. There can be no doubt that the former are much
more reliable than the latter. Experience is very prone to give
fallacious results, especially when questions of appetite are
concerned. In making a trial of abstinence, the mistake has been
commonly made of only prolonging the investigation for a few
weeks, and then comparing results. Such a method is, however,
very unfair, and is certain to lead to an unreliable conclusion.
The records of experience under certain conditions have,
however, been so extensive, as to lead to trustworthy results.
It has been abundantly proved that prolonged muscular work is
best undergone during total abstinence from alcohol; and that
the extremes of heat and cold and the exposure and exertions of
marching armies, are best borne under similar conditions.
The artificial character of town life is commonly adduced as
an argument for the moderate use of alcohol. In the case of
healthy workers, this does not hold good; many of our hardest
workers and thinkers take no alcohol.
The universality of the habit of taking stimulants is a curious
argument on the same side, though if the habit be bad, this can
be no more reason for continuing it than can the prevalence of
vice be an excuse for indulgence in it.
The two chief physiological points bearing on the advisability
of alcohol as a part of one’s daily diet are—its food properties, and
its effect on the appetite and digestion.
It has been already stated that a quantity of alcohol under 1 or
1½ ounces may become oxidised in the system, and may thus form
a source of heat. But in all probability, although it may be
regarded as a food, it is a most inconvenient one, inasmuch as it
diminishes the oxidation of other foods. It has been aptly com61pared
in this respect to sulphur, which is an oxidisable material,
but which, when it is burnt in a chimney, in which the soot is on
fire, will put an end to the combustion of the latter. Its value as
a food, under normal conditions, is practically nil.
Its Effect on the Digestive Organs is three-fold. (a) The
contact of alcohol with the mucous membrane of the mouth and
stomach, acts as a reflex nervous stimulus, which in moderation
excites an increased flow of gastric juice. (b) It also increases the
activity of the movements of the stomach. In cases of weak
digestion, therefore, small doses of alcohol may, at times, be useful.
(c) The effect of alcohol on the food taken varies with its degree of
dilution. Concentrated alcohol coagulates albumin, and so stops
digestion; largely diluted alcohol has no such effect.
The late Dr. Parkes, the greatest authority on the dietetic use
of alcohol, has summarised the argument as to the dietetic use of
alcohol as follows:—
“But what, now, should be the conclusion as to the use of
alcohol in health after growth is completed? Admitting the
impossibility of proving a small quantity to be hurtful, and at the
same time acknowledging the dangers of excess, there arises an
argument which seems to me somewhat in favour of total abstinence.
No man can say when he has passed the boundary which
divides safety from harm; he may call himself temperate, and yet
may be daily taking a little more than his system can bear, and be
gradually causing some tissue to undergo slow degeneration. He
may be safe, but he may be on the verge of danger.
“This uncertainty, coupled with the difficulty at present of
saying what dietetic advantage is gained by using alcohol, seems to
me rather to turn the scale in favour of total abstinence instead of
moderate drinking. But if any one honestly tries, and finds he is
better in health for a little alcohol, let him take it, but he should
keep within the boundary line, viz., that 1½ ounces of pure or
absolute alcohol in twenty-four hours form the limit of moderation.
I do not then think he can do himself any harm.”
The Varieties of Fermented Drinks.—The three chief
kinds of alcoholic beverages are malt liquors, wines, and ardent
spirits. In addition, we may mention cider and perry, which are
the fermented juices of apples and pears respectively; and koumiss,
which the Tartars prepare by fermenting mare’s milk, though it may
also be made from the milk of other animals.
All Beers, Ales, and Porters are prepared from malt, which is
the germinating grain of barley. The fermentation of the sugar
in the barley produces alcohol, the amount of which varies in
different cases. In Pilsener beer it is 3½ per cent. of absolute
alcohol; in stout and porter 5 to 6 per cent. The hop which is
added to the fermenting barley, gives to beer its characteristic
bitterness.
London Porter is coloured with black or roasted malt; stout
is only a stronger form of porter. Bottled ales are generally stronger
than those on draught, and being slightly effervescent, may agree
better.
62
The effect of alcohol in beer is modified by the hops, which help
in producing drowsiness. Beer has a marked tendency to produce
obesity, more so than any other alcoholic beverage. Its influence
in the production of gout is also very great.
Substitutes for Malt have been largely used. Thus by the
action of sulphuric acid on starch, an artificial form of sugar is
produced, which is largely used in place of malt for making beer.
Many recent cases of poisoning by arsenic have been traced to the
use of impure sulphuric acid in manufacturing this form of sugar.
The detection of arsenic in organic liquids requires great care, as
so many compounds of arsenic are volatile, especially in the presence of
chlorides, as in beer. To detect arsenic in beer a pint of the beer is evaporated
to dryness, and treated with 20 c.c. of strong sulphuric acid, heated, and
20 c.c. of strong nitric acid added drop by drop. Violent action occurs: if
possible 20 c.c. more of nitric acid are worked in. Transfer the liquid to a
small flask, and expel the nitric acid by boiling. By this means all chlorine
is expelled, the arsenic is oxidised and the organic matter destroyed. SH₂ gas
is now passed into the acid liquid for some hours, the precipitated sulphur
and any sulphide filtered off and extracted with ammonia, which dissolves any
sulphide of arsenic. The liquid so obtained is subjected to Marsh’s test. (See
page 216.)
In the making of beer from malt, the first stage is to malt the
barley, i.e. leave it spread on floors for ten days after soaking.
This allows germination to take place, in which process the
insoluble starch is converted into starch, dextrine, maltose and
glucose. After the dried malt has been screened to break off the
sproutings, the brewer places it in the mash-tub, with water, at a
temperature of 160° F. This completes the transformation of the
starch into glucose. The wort is now boiled to stop the process,
and the albumin from the grain is thus coagulated. Hops are
added at this stage. The boiled liquid is passed into shallow
vessels and cooled. The proper temperature for “top” yeast is
60° F., for “bottom,” or Bavarian yeast, a much lower temperature
is desirable. When the desired temperature is reached, the liquid
is run into the fermenting tun along with yeast. The varieties of
beer are due in part to the degree of completeness of fermentation of
sugar allowed. If too complete, the beer does not keep well.
Wines are produced by the fermentation of the juice of the
grape. The wine produced may be bottled before or after fermentation
is complete; in the former case, an effervescing wine is
produced, such as the sparkling wines of the Rhine and the
Moselle, or champagne. When the sugar is nearly all fermented a
dry wine is obtained, of which Bordeaux and Burgundy, Hock and
Moselle, are examples.
The difference in colour between red and white wines is produced
by allowing the juice in the former to ferment in contact with
the skins, from which the colouring matter is extracted by the alcohol.
Both red and white wines may be obtained from either red
or white grapes. From the skins also are extracted a salt of iron,
and a peculiar form of tannin. Tartaric and acetic acids, and
tartrate of potass, are present in varying quantities in wines; in old
wines the tartrate separates as bitartrate of potass, forming with63
tannin and colouring matter the “crust” of port and other wines.
The “bouquet” of wines is due chiefly to certain volatile bodies,
such as pelargonic ether. The proportion of alcohol in wines varies
from 6 to 14 per cent. As fermentation is stopped by the presence
of 14 per cent. of alcohol, any larger amount of alcohol than this
must have been added to the wine.
Wine, like beer, has a strong tendency to produce gout, especially
the sweet and strong wines. It has not, however, the same
tendency to induce obesity.
Spirits differ from the two last groups, to begin with, in the
amount of alcohol they contain. Thus, English beers contain from
3 to 6 per cent., German beers from 2 to 5 per cent., wines from
8 to 20, and all kinds of spirits from 37 to 58 per cent. of alcohol.
They differ in the absence of the bitter principle of beer and much
of the salts and sugar and ether of wines. They are all prepared
by the distillation of some previously fermented liquor. Brandy
ought to be made by the distillation of wine; and then contains,
besides alcohol and water, small quantities of acetic, œnanthic,
butylic, and valerianic ethers. But much of the brandy sold is
simply made from potato spirit, by the addition of acetic ether,
burnt sugar, etc. The starch of potatoes is converted into dextrin
and dextrose by dilute acids, and then fermentation allowed. By
the use of patent stills, all bye-products can be separated, a fairly
pure alcohol known as silent spirit being produced. This is largely
employed in manufacturing spirits and in fortifying wines.
Whiskey is prepared from malted barley, or from a mixture of
grains, to which a sufficiency of malt to convert their starch into
sugar has been added. In grain whiskey the distillation is effected
by steam in a patent (Coffey’s) still, which separates most of the
bye-products (fusel oil, etc.) from the spirit. In malt whiskey,
distilled in the old-fashioned pot-still, these bye-products are not
separated.
The improvement of whiskey effected by keeping is not due
(Bell) to the diminution of fusel oil. Such a diminution does not
occur. The percentage of alcohol diminishes by keeping, 6 to
8 per cent. proof spirit being lost by five years’ storage in wood.
“Fusel oil” is a mixture of alcohols of higher boiling point than
ethylic alcohol (amylic, propylic, etc.). Even in a bad whiskey
not more than 1 ∕ 10 per cent. of fusel oil is present (about one grain in
a glassful). Experimentally no marked effects have been produced
by fusel oil, when it is less than 1 per cent. Possibly the presence
of furfurol, of which there is a trace in malt whiskey, which disappears
on keeping, may partially explain the disagreeable flavour
of new whiskey. But it is fairly clear that those who argue that it
is bad whiskey and not good whiskey which does harm are speaking
without knowledge. It is not the quality but the quantity of
whiskey which is responsible for so much moral and physical evil.
Gin and Hollands are obtained from barley, and flavoured with
juniper berries and other materials. The oil of juniper stimulates
the urinary excretion.
Rum is obtained by the distillation of molasses, and is usually64
kept for a long time in oak barrels. It is said thus to acquire more
astringent matters than other spirits contain.
The legal limits of dilution of whiskey, brandy and rum is
down to 25 degrees under proof, and of gin down to 35 degrees
under proof. (For definition of proof spirit, see page 55). The
amount of alcohol in an alcoholic liquor is determined by distillation
of 100 c.c., making up the distillate to 100 c.c. by the addition of
distilled water, and then taking the specific gravity of a portion of
this liquid by the aid of the specific gravity bottle. The percentage
of alcohol corresponding to a given specific gravity is given in tables
prepared for this purpose.
Prolonged indulgence in spirits produces the various organic
diseases already described, and unless well diluted they are more
harmful than beers or wines. They differ from wines and beers in
not tending to produce gout, and from beer in not leading to obesity.
65
Uses of Water.—Water is a prime necessity of life. In its
absence life can only exist in lowly organised beings, and in them
only in a dormant state. From a hygienic point of view, the uses
of water are four-fold:—(1) It is an essential part of our food,
not only serving to build up the tissues of the body, but also
preserving the fluidity of the blood and aiding excretion of effete
matters. (2) It is necessary for personal cleanliness, of which
the importance can scarcely be exaggerated. (3) In the household
it is essential for cooking, as well as for washing the house, the
linen, and various utensils. (4) By the community at large it is
required for water-closets and sewers, for public baths, for cleansing
the streets, and for horses and other domestic animals, as well as
in many manufacturing processes. It is obvious that the water to
be used for domestic and general purposes, need not be so pure as
that for drinking purposes. Hence, a double supply was proposed
for London in 1878, by the Metropolitan Board of Works—a less
pure river supply for general purposes, and a deep chalk-well supply
for drinking purposes. The scheme, however, rightly fell through,
because of the expense of a double source of supply, and the danger
that the impure water would, through carelessness or ignorance, be
often used for drinking purposes, when it happened to be nearest at
hand.
Quantity of Water Required.—The quantity of water
required for all purposes has been variously stated by different
authorities. The quantity required for drinking purposes is found
to bear a relation to the weight of the individual, being nearly half
an ounce for every pound weight, or 1½ gills for every stone weight.
Thus, a man weighing 150 lbs. would require 3¾ pints. Of this
water, about one-third is taken in the food; the remainder,
averaging 2½ pints, being required as drink. If we add the water
required for other purposes, according to De Chaumont, 1 gallon is
required for drinking and cooking, 2 gallons (not including a bath)
for personal cleanliness, 3 gallons for a share of utensil and house
washing, 3 gallons for clothes washing; and if a general bath be
taken, 3 gallons more; making a total of about 12 gallons, to which
5 gallons must be added if there is a water closet.
In hot summer weather the consumption is about 20 per cent.
above the average of the year; and frost often increases the amount
30—40 per cent. above the average, owing to the bursting of pipes,
or the loss from taps foolishly left open to prevent bursting.
66
Water companies usually reckon 30-60 gallons for each
individual, to allow for the water required for scavenging and
manufactories and for waste. In large houses and hotels where baths
are freely used, often as much as 70 gallons per head is used, and in
hospitals the amount averages from 60 to 90 gallons per head. The
following is Parkes’ estimate of the daily allowance for all
purposes:—
|
GALLONS PER HEAD
OF POPULATION. |
| Domestic supply |
12 |
| General baths |
4 |
| Water-closets |
6 |
| Unavoidable waste |
3 |
|
— |
| Total house supply |
25 |
| Municipal purposes |
5 |
| Trade purposes |
5 |
|
— |
| Total |
35 |
It has been proposed to put a water-meter to each house, so
that the rate may be in proportion to the amount of water used.
The plan is objectionable for two reasons: 1st—Because it tends to
restrict the necessary use of water for purposes of cleanliness. A
scant supply of water is always followed by uncleanliness of house
and person, with its consequent diseases; at the same time closets
may be imperfectly flushed, and may become choked. 2nd—Because
of the primary expense of the meter, and of its maintenance.
Sources of Water Supply.—All our drinking water is
obtained in the first instance, by a natural process of distillation on
a large scale. The sun is constantly causing evaporation from sea
and land. The vapour produced, being condensed by a lower
temperature, returns to the earth as snow, dew or rain. All these
natural products have been at times utilised as sources of drinking
water.
1. Dew has on rare occasions been utilised at sea by hanging
out fleeces of wool at night and wringing them out in the morning.
A much better plan is—
2. The Distillation of Sea-water. This can easily be
managed now that steam power is so largely used. It has even
been employed on land, when it was necessary temporarily to
continue the use of water derived from an impure source. The first
part distilled should always be rejected, as it is always impure.
Distilled water is “flat” in taste, owing to its containing no
dissolved gases. It can be aërated by letting it drop a considerable
distance from one cask into another, through small openings in the
upper one, and by filtration through charcoal. Non-aërated water
is not easily absorbed into the circulation, and occasionally causes
illness.
3. The utility of Melted Snow and Ice is obviously very67
limited. Moreover, its use is not free from danger if the ice is
derived from contaminated water. Outbreaks of enteric fever have
been traced in the United States to the taking of ice obtained from
impure water.
4. Rain-water is a much more important source of water
supply, and after passing through the soil it constitutes the chief
part of the water we drink. The term, however, is properly
restricted to the water collected immediately after its descent from
roofs, etc. Its purity depends on three conditions—the character of
the air it passes through, the cleanliness of and absence of lead
from the channels through which it runs, and the condition of the
water-butts in which it is stored. Rain-water is soft; in fact, too
soft to be pleasant to the palate. In passing through the air, it
carries with it a certain proportion of its constituents; in towns
especially ammonia, soot, etc.; near the sea, it generally contains
some salt; and being soft and having dissolved oxygen from the
air, it dissolves an appreciable amount of lead from roofs or
gutters.
The Rivers Pollution Commissioners found that out of eight
samples of stored rain-water only one was fit to drink. They came
to the conclusion that rain-water, collected from the roofs of houses
and stored in underground tanks, is “often polluted to a dangerous
extent by excrementitious matters, and is rarely of sufficiently good
quality to be used for domestic purposes with safety.” Also, that
in Great Britain, and more particularly in England, we shall “look
in vain to the atmosphere for a supply of water pure enough for
dietetic purposes.”
The use of rain-water for drinking purposes is only justified in
isolated country houses where no better source is available; and
under these circumstances the greatest care should be taken to
prevent contamination with lead or organic impurities.
The amount of water falling on any impervious material
obtainable from rain can easily be estimated, if the amount of
rainfall and the area of the receiving surface are known. The
average annual rainfall in this country is 33 inches (see page 236).
We may assume the amount practically available to be 20 inches per
annum, and the area of the receiving surface 500 square feet. Multiply the
area by 144, to bring it into square inches, and this by the rainfall, and the
product gives the number of cubic inches of rain which fall on the receiving
area in a year. One cubic foot, or 1,728 cubic inches, of water being equivalent
to 6·23 gallons, the number of gallons of water can be easily calculated. To
calculate the receiving surface of the roof of a house, do not take into account
the slope of the roof, but merely ascertain the area of the flat space actually covered
by the roof. This may be done roughly by calculating the area of all the rooms
on the ground floor, and allowing an additional amount for the space occupied
by the walls. It has been estimated that, even if a rain-water supply for towns
were desirable, the amount collected from the roofs of houses would scarcely
average two gallons per person daily—assuming the average rainfall to be 20
inches, and that there was a roof area of 60 square feet for each individual.
The amount practically available from rain falling on different
soils varies with their porosity and slope. Thus, according to68
Professor Rankine, the proportion of the total rainfall available is
as follows:—
- Nearly the whole on steep surfaces of granite, gneiss, and
slate;
- From three to four-fifths on moorland hilly pastures;
- From two-fifths to half on flat cultivated country; and
- None on chalk.
By available rainfall is meant the amount remaining after
allowing for percolation, etc., which can be stored in reservoirs.
5. Upland Surface Water is the water collected in hilly
districts, as on moorlands, at the head of a river. By its utilisation
for drinking purposes, the sources of water for the river are interfered
with, and any water company or local authority using such a
source is, therefore, required to run into the stream a quantity of
water equal to a third of the available rainfall. The limited and
regular supply thus furnished to the stream is found to be
advantageous for industrial purposes as its flow is equalised, and
the violence of floods mitigated.
In the utilization of upland surface water the water from the
surrounding hills is collected at the bottom of a valley, in an
artificial, strongly-constructed lake; or in a natural lake, as in
Loch Katrine (from which Glasgow is now supplied).
Upland surface water is nearly always soft. Its use is much
more economical than that of hard water. It may be brownish,
from the presence of peat, but this is not objectionable, so far as
health is concerned. Its occasionally solvent action on lead is a
more serious objection. The population of many parts of Yorkshire
and Lancashire have suffered severely from chronic lead poisoning,
due to the action of certain upland surface water on lead service
pipes. Only the waters giving an acid reaction possess this plumbo-solvent
power. (See also page 82.)
6. Springs supply water which, originally derived from rain-water,
has percolated through the soil until it reaches some
impervious stratum, and has then run along this, until it arrives
at the point at which the impervious stratum reaches the surface of
the soil. A spring is thus the outcrop of the underground water.
Springs are divided into (1) land springs, and (2) main springs.
The former flow from beds of drift or gravel lying on an impervious
stratum. They are very subject to seasonal variation, and may dry
up in certain years; while main springs occurring in chalk, greensand,
or other regular geological formation, constantly supply a
certain amount of water. Springs often occur in connection
with “faults” in geological strata, and then may appear on table-lands
and high elevations, unlike springs caused by alternation of
strata in valleys of denudation. The two kinds of springs are shewn
in Fig. 5 and 6.
In the land spring water crops out at the point where the
porous stratum ceases. Deep springs may crop out in the same
way as land springs, except that they appear at the bottom of
deeper strata. Or they may be formed by faults. Both these are69
shown in water having percolated through the chalk beneath the
superficial clay, is stopped at the “fault” by the lack of continuity
of the chalk stratum, and is consequently confined under pressure.
It therefore makes its way to the surface, forming a spring. In its
passage underground, water (owing partly to the carbonic acid it
has obtained from the air and soil), is able to dissolve small
quantities of chalk, sulphate of lime and of magnesium, and traces
of oxide of iron, aluminium oxide, and silica. Spring-water possesses
an equable temperature, generally about 50° Fahr., while impounded
or river water is always warm in summer and cold in winter.
Spring water is well-aerated, while river water, and still more rain-water,
are flat.
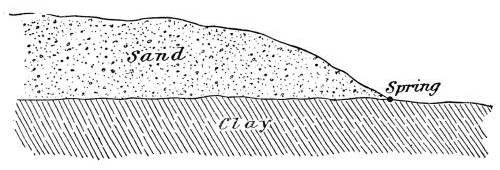
Fig. 5.—Land Spring.
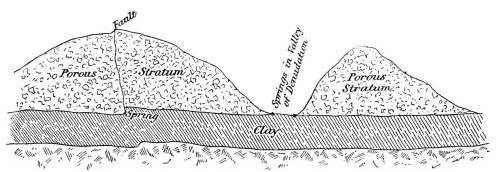
Fig. 6.
Main Springs formed in Valley of Denudation and by a Fault.
7. Wells may form the best or worst sources of water-supply
according to their depth and the means of protection against
contamination. There are two kinds—Surface wells and deep wells.
Surface Wells do not usually descend further than 15 or 20
feet, and have no impervious stratum between the source of water
and the surface of the well. They catch the subsoil or underground-water,
which percolates into them from the surrounding soil, and
the character of the water they receive will therefore vary with the
nature of their surroundings. If there is a cesspool near, this may
simply drain into the well. All the soakage from a considerable
distance may find its way into the well. In villages and isolated
places the water of surface wells is commonly contaminated. One
hole may be dug in the garden for a well, and another for a cesspool,
while there is possibly a farmyard near at hand—the soakage from70
the cesspool and farmyard soaking into the well. Danger may
also arise from more distant contamination. The ground water
which is tapped by the well is an underground stream flowing
towards the nearest brook. Heavy rains swelling the ground water
may wash impurities from cesspools, leaky drains, etc., at a
considerable distance, and carry these into wells lying between
these sources of pollution and the brook into the bed of which the
underground water ultimately discharges. The danger of contamination
of the water in the well by the contents of the cesspool is
much greater in the relative position shown in A than in the
position shown at B, Fig. 7. After heavy rain, when the underground
water is swollen, the danger of contamination is still further
increased. The model bye-laws of the Local Government Board
state that a cesspool must be at least 40 feet distant from any well,
spring, or stream. Probably this is insufficient for safety; cesspools
ought to be entirely forbidden. If necessary to retain a surface well,
it should be protected nearly to the bottom with brick, lined with an
impervious layer of cement so as to prevent water from entering
the well except near its bottom. In modern wells iron cylinders
are employed to line the upper part of the well; and large glazed
earthenware pipes arranged vertically and with water-tight joints
are sometimes used for the same purpose.
Fig. 7.
Showing Varying Danger of Contamination of a Shallow Well,
according to Level of Underground Water and Relative Position of
Cesspool and Well (after Galton).
Deep Wells are made by digging a surface well, as above,
except that the ground water is prevented from entering the well by
means of impervious steining; and then boring from the bottom
down through the subjacent impervious stratum until a water-bearing
stratum is reached. The difference between a surface well
and a deep well is shown in Fig. 8 by A and B. Where the water71
in this stratum is retained under pressure, deep wells are known as
Artesian Wells. Such Artesian wells have been sunk in London.
Rain, falling on the chalk hills which lie to the south and north of
London, percolates through the chalk downwards, and then laterally,
until it lies in the concave London basin. Here the clay stratum
above it prevents its escape upwards; and being confined under
considerable pressure, it rises to the surface, or into a well in the
superficial gravel, when the clay is tapped. In Fig. 8, B is an
Artesian well if the pressure is such as to make the water rise
through the London clay, when this is cut through and the
underlying chalk is reached. C is a well in the chalk, which does
not pass through an impervious stratum, and therefore comes within
the above definition of a surface well; but as regards depth required
to be dug before water is reached it is more like a deep well.
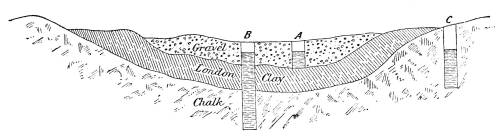
Fig. 8.
Showing Difference between Deep and Superficial Well.
A.—Surface Well in Gravel. B.—Deep Well, going through Gravel
and Clay to Chalk. C.—Well in Chalk District.
Among the deepest Artesian wells are Grenelle (1,800 feet), and
Kissingen (1,878 feet.) The sinking of a deep well and severe
pumping of its water may exhaust all the neighbouring wells for
two or three miles. There is also danger of contamination from
neighbouring cesspools when the upper part of the deep well is not
properly constructed. The area exhausted by a deep well undergoing
pumping is represented by an inverted cone, having a very wide
base, and with a convex inner surface pointing towards the well.
For country places deep-well water is much preferable to water
from streams, as streams are very liable to be contaminated by the
sewage of houses higher up in their course, or even by that of
houses close by. A good well should be at least thirty feet deep—preferably
fifty feet and should always be lined with impervious
material, except near its bottom. The absolutely water-tight and
impervious condition as well as the distance of all drains or
cesspools in the vicinity should be ascertained before deciding
whether the drinking water from a given well is above suspicion.
The direction of flow of the underground water should also be
determined. This may be done by measuring the level of all the
wells in the neighbourhood. Possible sources of pollution at points
from which ground water is flowing towards the well are much more
dangerous than those nearer than the well to the river towards
which the underground water is flowing (see Fig. 7). Steam
pumping greatly increases the area from which contamination may
be derived.
72
An excellent plan to obtain water for villages, in a gravelly
soil, is to sink a Norton’s Abyssinian tube well for fifty or sixty
feet.
In towns it is preferable to trust to the public water supplied,
rather than to any private well; and in villages, a general supply
from a pure source should also be provided.
The water is obtained from a well by a pump or a draw-well.
The former is a safer as well as a less laborious plan. The pump
should be fixed some distance from the well, and the aperture
through which the pump pipe passes should be rendered water
tight. Lead pipes should be avoided, as well water not infrequently
has plumbo-solvent properties.
8. Rivers and running streams originate in upland surface water
or springs, and their water should be of the same quality as these.
Unfortunately, they acquire a large amount of impurities in their
course. Towns commonly pour their more or less clarified sewage
into them; and the discharge of crude sewage from hamlets and
single houses on the banks is still far from uncommon. With the
more rigid enforcement of the Rivers Pollution Acts, this pollution
of rivers will become less frequent; but river water previously
contaminated by even small amounts of sewage cannot be regarded
as an ideal source of water-supply.
If no contamination be present in the water of a river, it forms
a good source of water-supply; being running water, it is always
fairly well aërated, and is not usually so hard as spring-water.
Even if sewage has entered a river, it is asserted that it becomes
a safe source of water-supply, after passage through filter beds, the
sewage having been got rid of in four ways.
1st.—By subsidence, the organic matter settling to the bottom.
2nd.—By the influence of water-plants, which assimilate ammonia,
nitrates, etc., and give out nascent oxygen.
3rd.—Oxidation. Doubtless a large amount of the nitrogenous
matter does become oxidised in its course down a river, and in this
condition is harmless. The river Seine becomes greatly polluted
as it passes through Paris, but so far as chemical analysis can
determine its condition, it is purer 30 miles below the city than
it was before it received the sewage of the city.
4th.—It is highly probable that the germs (or micro-organisms)
of enteric fever and other diseases known to be propagated by
polluted water, are practically or wholly destroyed in the struggle
for existence with the natural micro-organisms of river-water.
When to this is added the fact that river-water supplied to large
communities is carefully filtered through sand, after having been
stored in reservoirs, in which the chief impurities have time to
settle, it is not surprising that the experience of those communities
like London, which are supplied with river-water, usually shows
no evidence of evil ascribable to drinking this water. For over
30 years the inhabitants of London have been drinking filtered water
from the river Lea and from the Thames above Teddington, and
this gigantic experiment on a population which has increased from73
2½ to 5 millions has not been accompanied by any conclusive
evidence of evil effect.
In regard to the comparative merits of the various waters
described, it will be useful to give here the classification made by
the Rivers Pollution Commissioners in their sixth report:—
| Wholesome |
 |
1. Spring Water |
 |
Very palatable. |
| 2. Deep-well Water |
| 3. Upland Surface Water |
 |
Moderately palatable. |
| Suspicious |
 |
4. Stored Rain Water |
5. Surface Water from
Cultivated Land |
 |
Palatable. |
| Dangerous |
 |
6. River Water to which
Sewage gains access |
| 7. Shallow-well Water |
Passage through certain geological strata has a great influence
in rendering water palatable, colourless, and wholesome by percolation.
The following strata are said by the Commissioners to be the
most efficient:—(1) Chalk, (2) oolite, (3) greensand, (4) Hastings
sand, (5) new red and conglomerate sandstone. Fissures or cracks
in these strata may cause the water to pass through them unpurified
by filtration.
74
CHAPTER X.
THE STORAGE AND DELIVERY OF WATER.
The methods of storing and delivering water will vary with its
source. In rural districts, deep wells and springs are the best
sources of supply; but in large towns they are found to be
insufficient for the wants of a rapidly-increasing population; and
they can only be multiplied in a given district within certain limits,
as every well drains a large surrounding area. The supply from
surface wells in gravel or sand beds or in chalk districts is liable
to fail in seasons of drought; but deep wells in oolite or chalk
formations, and in the new red sandstone, generally yield a
constant and abundant supply.
When the water is supplied from upland surfaces, springs,
or small streams, a collecting reservoir is required. This is
generally a natural valley below the level of the source of supply,
but of sufficient elevation above the place supplied to allow the
water to be distributed by gravity, without any pumping apparatus.
The reservoir should be large enough to hold five or six months’
supply, and its embankment should be perfectly water-tight, and
of great strength.
When water is collected from upland surfaces, it is important
to know the amount of rainfall to be reckoned on. If we know
the area of the surface which drains towards the reservoir, and
the average rainfall, the total rainfall is easily calculated. This
will, however, differ greatly from the available rainfall, owing to
the losses from penetration into the ground, evaporation, and other
causes. The amount lost will vary, according to the season, from
one-half to seven-eighths of the total rainfall; and according to
the soil (page 68). The proportion of percolation in the chief
water-bearing strata surrounding London varies from 48 to 60 per
cent. (Prestwich). It is less when the ground is steep and the
rainfall rapid, and usually less in winter than in summer.
Water collected near its actual place of fall, and from
uncultivated districts, is always purer than that collected further
from its source, and from cultivated land.
From the collecting or impounding reservoir, water is carried
by the aqueduct or conduit either directly into the service-pipes,
or when the pressure is too great, into a second service-reservoir,
resembling the impounding reservoir in general structure, and
capable of holding a few days’ supply.
This must be high ground, above the level of the highest75
houses to which water has to be supplied, as water cannot rise
above its own level. When this cannot be arranged, the water
is pumped into tanks at a higher level, and distributed from
them.
The greatest hourly demand for water being double the
average hourly demand, the water-mains supplying a town must
have double the discharging power that would be required,
supposing the demand was uniform. The first requisite of a
supply of water is that it should be abundant, and sufficient in
amount for any extra strain on its capacities. Water ought to
be laid on to every house, and to at least two floors of the house.
Anything preventing free access to water, militates against
cleanliness.
Cast-iron is the most serviceable material used in the
construction of the main water-pipes; it is coated with pitch, or
Dr. Angus Smith’s varnish, or with magnetic oxide of iron (Barff).
The service-pipes to each house are generally made of lead, and
the ease with which this material can be bent and curved, and
carried to the different floors of a house, makes its use very
convenient. Lead pipes, furthermore, can be easily obliterated
in case of bursting, and so any waste of water and flooding of
the house minimised. Some kinds of water, unfortunately, act on
and dissolve lead; this is especially true of soft waters and those
containing organic matter. Shallow wells, being very liable to
organic pollution, ought never to have the supply-pipe of their
pumps made of lead. With hard waters, lead pipes may generally
be used safely. When the quality of the water renders lead pipes
objectionable, the use of iron, tin, zinc, tinned copper, earthenware,
gutta-percha, and other materials, has been suggested. Of these,
cast and wrought-iron pipes are the most serviceable, or pipes
composed of an inner lining of block-tin and an outer of lead, a
layer of asbestos intervening to prevent galvanic action between
the metals. According to Rawlinson, “supply-pipes of wrought-iron
are cheaper, stronger, and more easily fitted than service-pipes
of lead;” but it is urged against them by Perry, that with soft
water they become choked by rust in a few years. If galvanized
they are more durable. Cast-iron pipes are rusted less easily
than wrought-iron.
When the water-supply is from a river, filtering beds are
needed, in addition to the parts of a water-service hitherto
described. Moreover, since the river is usually at a low level,
the water, after passing through the filtering beds, requires to be
pumped into raised tanks, from which it is delivered.
In laying down water-pipes, in the streets and to houses, it
is very important to make the distance between them and all drains
and gas-pipes as great as possible. Suction of gases or liquids
may occur into leaky pipes, even though these contain water, and
still more when they are empty; and disease has occasionally been
traced to this source. Thus if sewers and water-mains are laid
in the same trench, foul matters which have escaped into the soil
from the former may be sucked into the latter. This may happen76
if the water-mains are leaky, even when they are running full.
Experiments have shewn that the flow of water causes a partial
vacuum and insuction at the defective points. During intermissions
of supply when the mains are partially or entirely empty,
the danger of leakage into them is still greater. Coal-gas has been
similarly sucked into water-mains.
The pipes bringing the water to a house may be kept
constantly filled with water, or only for a limited time once or
twice a day. The intermittent system of supply necessitates the
provision of cisterns or water-tanks, in which water can be stored
in the intervals of flow of water. With a sufficient and properly-distributed
public supply of water, no cistern ought to be required.
Cisterns.—Cisterns for the supply of potable water may be
made of iron, slate, stone, glass, glazed earthenware, or brick lined
with Portland cement. Other materials have been used, as timber,
lead, and zinc. Timber is inadvisable, as it easily rots; lead is very
objectionable, owing to the possible solvent action of the water on
it. Zinc or galvanized iron cisterns are also acted on by soft
water; but they may be used with most waters. Galvanized iron
is iron coated with a thin layer of zinc. Iron cisterns soon rust;
but this may be prevented by giving them a coating of boiled
linseed oil before they leave the foundry. Stone cisterns are too
heavy for use, except in basements. Slate cisterns are good, but are
apt to leak; the points of leakage have occasionally been stopped
with red lead, which is attacked by the water, and thus lead
poisoning results. If the slate is set in good cement (not mortar,
as this makes the water hard), it is a good material for a cistern.
Every cistern should have a well-fitting lid, always kept closed,
to avoid the entrance of dust of various kinds, or even dead cats,
birds, etc. Noxious gases may be absorbed by the stagnant water.
The cistern should be easy of access. If it is indoors, the cistern
room should be well ventilated; and in any case the cistern should
be periodically visited and cleaned out. When the cistern is full, a
ball-tap prevents any further flow of water; and if this does not act
properly, an overflow pipe carries off the excess of water.
Cisterns badly arranged or neglected have been in the past a
common source of disease. (1) The overflow pipe should not pass
into any part of the water-closet apparatus or the soil-pipe, or into
the supply pipe to the water-closet.
Where the overflow-pipe discharges into the soil-pipe or closet
pan, foul gases or even solid particles may find their way into the
cistern.
(2) No water-closet ought to be supplied from the same cistern
as supplies drinking water, as the pipe leading down to the closet
may when the cistern is accidentally empty carry noxious effluvia
into the cistern. A separate flushing cistern capable of discharging
two to three gallons of water should be provided for each closet.
With a constant supply of water, cisterns are only required for
water-closets and for hot-water apparatus (see pages 168 and 164).
Constant and Intermittent Services.—With an intermittent
service of water, during the intervals of supply, water is77
only obtainable from cisterns, water-butts, etc. The objections
against this system are that—(1) The cisterns required are
expensive, and liable to get out of order and become foul. (2) Their
overflow pipes may improperly communicate with the soil pipe or with
some other part of the drainage, instead of opening into the external
air. (3) Putrid gases, from neighbouring ventilating-pipes or other
parts of the drainage system, are liable to be absorbed by the
stagnant water in the cistern. (4) The chief objection to an
intermittent supply is that, during the intervals in which the water-mains
are empty, foul air and liquids from the contiguous soil and
drains are liable to be sucked through imperfect joints into the pipes.
(5) In case of fire, the supply of water in the system is insufficient.
In certain towns rates of insurance against fire have been reduced
on replacing an intermittent by a constant service of water.
On the other hand it is urged that more expensive fittings are
required for a constant service; and that, when taps are left open or
pipes burst, the waste of water is much greater than with a cistern
supply. The balance is decidedly in favour of a constant supply
without storage cisterns. Where storage cisterns are in use, the
taps for drinking-water should be connected with the “rising-main,”
before it supplies the cistern.
The Advantages of the Constant Service may be thus
summarised:—
(1) Owing to the absence of cisterns, the risks connected with
stagnant water, and with improper arrangement of overflow pipes,
are obviated.
(2) The risk of suction into supply mains of external contaminations
is reduced to a minimum, since the pipes are never empty.
(3) The pipes are less liable to rust. Air in the presence of a
little moisture, causes rapid corrosion.
(4) There is an abundant supply of water in case of fire.
Of course, when there is a temporary stoppage of supply, as for
repairs, some of the dangers incurred by an intermittent supply will
arise.
78
Properties of Water.—When pure, water is colourless, or
bluish when seen in large quantity. It should be quite inodorous.
If, after keeping it for some time in a perfectly clean vessel, or if on
heating it a smell is developed, the water is bad. Its taste should
be pleasant and sparkling from the atmospheric gases dissolved in
it. Bitterness generally indicates the presence of sulphate of
magnesium (Epsom salts). Saltness is always a suspicious
property, except in water obtained in the neighbourhood of salt
mines or brine springs, or near the sea. It should be soft to the
touch, and should dissolve soap easily. It should be bright and
clear, and contain no suspended matters. Clear water is not
necessarily pure, but turbid water is always to be rejected; the only
exception being the brownish-tinged water from moors, which is not
hurtful. In all other cases, printed matter should be legible through
at least 18 inches of water in a clear glass cylinder. Thoroughly
dissolved organic matter is less dangerous than suspended; the
turbidity of water is therefore of great importance. But water may
be bright and sparkling and apparently perfectly clear, and yet
highly dangerous. The most important of the physical properties of
water in regard to health are the absence of smell and turbidity,
and these can be ascertained by even the most inexperienced. The
chemical tests for the more important impurities are given (pages 85
to 87).
The impurities of water may be classed under four heads—gaseous,
mineral, vegetable, and animal.
The gases ordinarily present in water cannot properly be
regarded as impurities, inasmuch as they are always present, and
greatly increase its palatableness. The dissolved nitrogen and
oxygen bear to each other the proportion 1·42 to 1; where sewage
contamination occurs, the oxygen will be diminished or disappear,
owing to oxidation of the organic matter.
The amount of carbonic acid gas in water varies greatly. It
may be considerable in chalk waters, and in contaminated well-water.
Mineral Impurities.—Mineral impurities are dissolved by
water in its course through the soil, and so will vary with the
character of the latter. 1. The water obtained from granitic
formations contains very little mineral matter, often not more than
two to six grains per gallon. Clay slate water is also generally very
pure, as is the water from hard trap rocks. 2. The water from79
millstone grit and hard oolite is very pure, often containing only four
to eight grains per gallon, chiefly calcium and magnesium sulphate
and carbonate. 3. Soft sand-rock waters usually contain thirty to
eighty grains per gallon of sodium salts, with a little lime and
magnesia. 4. Loose sand and gravel waters vary greatly. They may
be almost free from mineral matter, or the solids may be more than
seventy grains per gallon, including much organic matter. 5.
Waters from the lias clays vary somewhat, but commonly contain a
large quantity of calcium and magnesium sulphates. 6. Chalk
waters generally contain from seven to twenty grains of calcium
carbonate, with smaller quantities of other salts. 7. Limestone and
magnesian limestone waters differ from the last, in containing more
calcium sulphate and less calcium carbonate, as well as much
magnesium sulphate and carbonate in the dolomite districts. 8.
Selenitic waters contain calcium sulphate in considerable quantities.
9. Clay waters usually possess the characters of water from surface
wells, and are objectionable. 10. Alluvial waters generally contain a
large amount of various salts, including the various calcium,
magnesium, and sodium salts. 11. Artesian well water varies greatly
in composition. It may contain a large amount of sodium and
potassium salts, or a small quantity of iron, or calcium salts.
The commonest and most important mineral constituent of
water is calcium carbonate, next to this calcium sulphate. These
two salts are the chief causes of hardness of water. For practical
purposes as regards use in domestic matters and in manufactures,
the most important classification of waters is into hard and soft.
The degree of hardness varies within wide limits—from rain-water,
which has no hardness at all, to the water from new red sandstone
rocks which sometimes possesses a hardness of 90 degrees;
or wells in the gravel, in which it may be as much as 152 degrees.
The following classification of waters, according to the degree of
hardness, beginning with the least hard and gradually increasing
in hardness, is from the sixth report of the Rivers Pollution
Commissioners:—1. Rain-water. 2. Upland surface. 3. Surface
from cultivated land. 4. River. 5. Spring. 6. Deep-well.
7. Shallow-well water.
Calcium carbonate is the most common cause of hardness, and
the hardness produced by it is remediable by boiling or chemical
means. Calcium carbonate (chalk) is rendered soluble in water,
by the carbonic acid contained in the latter, a double bicarbonate
being thus formed. The air contained in the interstices of the
soil through which water passes, often contains 250 times as
much carbonic acid as ordinary air. The water, in percolating
through the soil, dissolves this carbonic acid, and thus is able to
take up a considerable amount of chalk.
The amount of hardness in any given water is expressed in
degrees, one degree being equivalent to a grain of calcium carbonate
in a gallon of water. Clarke’s soap test is employed to detect
the amount of hardness. It consists of a solution of soap of a
known strength. Soft water will form a lather at once with this;
hard water will only form a lather after all the calcium salt is80
neutralised. The amount of Clarke’s solution required before a
lather is produced, will give an estimate of the amount of hardness.
To Determine the Total Hardness take 70 c.c. of the water and place
in a stoppered bottle. From a burette run in a sufficient quantity of the standard
soap solution (of which 1 c.c. equals 1° of hardness), to produce a lather on shaking
the water, which remains unbroken after standing five minutes. Thus, if
7·5 c.c. of the soap solution were required, the hardness is 6°·5, as 1 c.c. of
the solution is required to produce a lather in soft water. The 6°·5 means
6·5 milligrammes of calcium carbonate in 70 c.c. or 6·5 grains in a gallon of
the water.
To Determine the Permanent Hardness boil 70 c.c. of the water
in a flask for half-an-hour; allow the precipitated carbonates of calcium and
magnesium to settle. Some of the latter will be re-dissolved. Carefully
decant, and make up the liquid to the original 70 c.c. with distilled water.
Filter through fine filter paper and estimate hardness as above.
The amount of soap wasted in consequence of the hardness of
water is very great. Thus, in the case of water of one degree of
hardness, as every gallon contains one grain of chalk, 7,000 gallons
would contain 7,000 grains—that is, a pound. But every grain of
chalk wastes 8 or 9 grains of soap; therefore, a pound of chalk,
contained in 7,000 gallons, would waste about 8½ pounds of soap.
But nearly all waters are harder than this, and they not uncommonly
possess a hardness of 20° or more. If the hardness be 20°, the
waste would be 170 pounds of soap. This quantity would be easily
used annually in a family of seven or eight persons, if we include
the washing of clothes. The amount of money thus wasted can be
easily estimated.
Not only does soft water require less soap, but it is much more
suitable for making tea and soup, and for boiling meat and
vegetables—both time and fuel being saved. The reason why
better tea is made when a little carbonate of soda is added to the
water is that the chalk is by this means precipitated.
Carbonate of calcium is precipitated from water by boiling it;
carbonic acid being driven off, the neutral salt falls to the bottom of
the vessel. This is the origin of the “fur” inside kettles, which
lessens their conductivity to heat, and renders necessary a greater
consumption of fuel.
The chalk may also be removed by adding to the water, while
still in the reservoir, some milk of lime—that is, quicklime made
into a milky solution with water. This is done on a large scale at
various waterworks. The reaction may be expressed thus:—
Calcium bicarbonate + calcium oxide = calcium carbonate +
calcium carbonate.
The calcium carbonate, as it is precipitated, carries down with
it organic and other matters, thus clearing and purifying the
water.
The hardness due to calcium sulphate is not removable by
boiling. It is, therefore, called permanent hardness, to distinguish
it from the temporary hardness of chalk waters, which is
removable by boiling. It may, however, be partially removed by
the addition of washing soda to the water, as well as the nitrate
and chloride of calcium which are also present. The magnesium81
salts are not removable by boiling or soda. This is shown by the
fact that the “fur” inside kettles does not usually contain
magnesium salts.
The amount of hardness varies greatly in different waters. In
the deep wells in magnesium limestone, it varies from 14°-57°;
in the deep wells from chalk beds, it varies from 13° to 27° and
may be higher. In the water from Bala Lake, Wales, the
temporary hardness is 0°·1, the permanent hardness 0°·3; in the
Loch Katrine water there is no temporary hardness, 0°·9 permanent
hardness; in the water from the new red sandstone (Nottingham),
the temporary hardness is 9°·6, permanent 10°·2; in a chalk spring
at Ryde, temporary hardness 16°·7, permanent 3°·9 (Wanklyn).
The total hardness in the metropolitan water supplies from the
rivers Thames and Lea, varies from 13°·2 (Southwark Company)
to 14°·6 (New River Company); in the Kent Deep Wells 20°·1;
in deep wells from the chalk at Brighton it varies from 12° to 13°.
In all these, the hardness is chiefly temporary.
The amount of permanent hardness is always great in water
from clays, as the London, Oxford, Kimmeridge, and Lower Lias
clays; or in places where there are large deposits of calcium sulphate,
as at Montmartre, near Paris (hence the name Plaster of Paris,
given to desiccated calcium sulphate). Water from fissures in the
clay often contains, also, a large amount of organic matter.
Chlorides are always present in small quantities in water.
As a rule the presence of more than 1 grain per gallon, i.e. ·7 parts
per 100,000 of water, indicates contamination with some animal
refuse, unless the water is derived from new red sandstone, or brine
springs, or from the neighbourhood of the sea. This rule does not,
however, hold universally good. The absence or the presence of
only a minute quantity of chlorides indicates the probable absence
of animal contamination; but in exceptional cases waters of the
highest organic purity may contain more chlorides than the same
bulk of sewage.
To determine the amount of Chlorine take 70 c.c. of the water, add
a few drops of solution of potassium monochromate (KCrO₄). From a burette
run in gradually a standard solution of silver nitrate (of such a strength that
1 c.c. of the solution is equivalent to 1 milligramme of chlorine). The silver
solution forms milky chloride of silver (AgCl) by combination with the
chlorine of the chlorides in the water. When all the chlorine is thus combined,
the next drop of the silver solution forms a deep red tint with the chromate.
The number of c.c.’s of the silver solution required to produce this effect,
equals the number of milligrammes of chlorine in 70 c.c. of water, or the number
of grains of chlorine in a gallon of water. To convert this into parts per 100,000,
divide by 7 and multiply by 10.
To express the amount of chlorine in terms of common salt (NaCl),
multiply the parts per 100,000 of chlorine by 1·65.
Nitrates in any water are suspicious; but their import varies
with the circumstances under which they occur. A minute quantity
of ammonium nitrate is present in nearly all waters; and the water
of deep wells, especially of wells in the chalk, which, as a rule
is perfectly free from sewage, may be highly charged with nitrates.82
Nitrates, when derived from sewage, represent a completely oxidised
condition of its nitrogenous matter. Crude sewage generally contains
no nitrates. Nitrites as a rule indicate more recent contamination,
and therefore greater danger than nitrates. The presence of more
than a trace of phosphates is a strong indication of contamination
with sewage matter.
To determine the amount of Nitrites and Nitrates the best known
methods are by the indigo, the phenol-sulphuric, the aluminium, or the zinc-copper
couple tests. For nitrites the metaphenylene-diamine test is employed
(page 85). The following qualitative tests will suffice for elementary work.
Nitrates. An equal amount of a solution of brucine is added to the
suspected water in a test-tube, then a little pure sulphuric acid is poured
down the side of the tube. A pink zone is produced if nitrates are present
in considerable amount.
Nitrites. A few drops of each of diluted sulphuric acid and of
metaphenylene-diamine solution give a red colour with water after standing
for a few minutes, if nitrites are present.
Lead is an occasional contamination of slightly acid waters.
The purest and most oxygenated waters act most readily on lead;
as also those containing organic matter, nitrates or nitrites. Waters
containing chlorides also act on lead, the chloride of lead being
sufficiently soluble to produce poisonous symptoms. Upland
surface waters derived from moorlands in certain districts, e.g.
around Sheffield, have been found to be capable of dissolving
considerable lead from lead service-pipes. The water taken first
from the tap in the early morning is the most heavily charged with
lead. Such waters are very soft; but other moorland soft waters do
not dissolve lead. It is the water having a slightly acid reaction
which possesses this property. The source of this acid, whether
sulphuric acid from the products of combustion in a neighbouring
town, or an organic acid, is uncertain. The plumbo-solvent action
of such water is greatest in autumn, when the amount of acid is at
its maximum. The property of dissolving lead is removed by
passing the water on a large scale over filters of sand, spongy iron,
chalk, or limestone. The addition of a small quantity of carbonate
of soda has the same effect. In such districts the use of tin-lined
iron pipes for domestic services has been recommended, but these
are liable to fracture when bent. Pipes consisting of an outer case
of lead and an inner pipe of tin with a layers of asbestos between
have also been placed on the market. (See also page 68.)
Hard waters have the least action on lead; a coating of
insoluble carbonate of lead being formed on the interior of the pipe,
which prevents any further action. Thus the use of lead pipes for
water containing carbonates or sulphate, or calcium phosphate, is
comparatively safe. Hard water containing carbonic acid gas under
pressure will dissolve a small amount of carbonate of lead; this
explains the cases of lead poisoning from soda water which was
formerly supplied in syphon bottles with lead tubes.
Lead is dissolved much more easily by water if other metals are
in contact with it, as iron, zinc, or tin, galvanic action being thus83
set up. Zinc pipes containing some lead are very dangerous,
especially with the distilled water used on board ships.
To determine the presence of lead in water, place a given quantity,
say 100 c.c. in a white dish, and stir with a rod dipped in a solution of
ammonium sulphide; if the water becomes coloured, this is generally due to
the presence of iron or lead. If the colour remains after adding a drop or
two of hydrochloric acid, lead is present.
To determine the amount of lead, a standard solution of lead acetate
containing 1 ∕ 10 milligrammes of lead in 1 c.c., is made by dissolving ·183 gramme
of crystallised lead acetate in a litre of distilled water. Place 100 c.c. of the
water to be examined in a Nessler glass, acidify by a few drops of acetic acid;
now add ½ c.c. of a saturated solution of ammonium sulphide. A brownish-black
discoloration is produced if lead is present. To a second Nessler glass,
containing 100 c.c. of distilled water, the same amounts of acetic acid and
ammonium sulphide are added, and then a sufficient quantity of the standard
lead solution is added, until the tints of the contents of the two Nessler glasses
are identical. The amount of the standard solution added being known, we
know the amount of lead in 100 c.c., and the amount per litre (1,000 c.c.)
will be tenfold. Thus if 2 c.c. of the solution were required for matching colours,
there were ·2 parts of lead per 100,000 of water, or ·14 grains per gallon.
Traces of Iron are sometimes present in water, giving it an
astringent taste. Such water is apt to turn brown; and tea made
from it is very dark.
Organic Impurities.—Organic impurities may be either
vegetable or animal, the latter being by far the most dangerous.
The water from moorlands is often brown, but this is not noxious.
Growing plants, again, may be beneficial to water, by absorbing
dissolved organic matter, and aiding its oxidation. Decaying
vegetable matter is objectionable in water, and may set up
diarrhœa.
The most important organic impurity of water has an animal
origin—from sewage; the liquid or solid excreta (i.e. the urine or
fæces) gaining accidental access to the water. Besides sewage, the
eggs of various intestinal worms have been swallowed with water;
and in a few cases, even leeches. But whatever the source of the
organic matter contained in water, it contains nitrogen as an
essential constituent; and tends under the influence of warmth, and
therefore especially in summer, to undergo putrefactive changes,
owing to the action of bacteria. These split up the more complex
molecules of organic matter into simpler matter; ammoniacal
compounds and salts, of which the most important are nitrites and
nitrates, being final products of their activity. The detection of
nitrates, and still more of nitrites, is important, as they may
indicate the occurrence of previous sewage contamination. These
products are quite harmless in water, except as an indication that
the water has been polluted, and that possibly a certain proportion
of the nitrogenous matter in the form of the complex organic
matter forming the germs of such diseases as enteric fever, may still
be present. Organic matter may be suspended or dissolved, the former
being most dangerous to health. The germs or microbes causing
disease consist of suspended, i.e. particulate matter. The amount
of organic matter is determined by the amount of free ammonia84
and albuminoid ammonia which are present (Wanklyn’s process),
by Frankland’s combustion process, or by Forschammer’s oxygen
process; all of which give indications, rather than an exact estimate
of its amount.
METHODS OF WATER ANALYSIS.
The following scheme of qualitative examination may be followed, when
an immediate opinion is required as to a water. It can only be trusted when the
examination shows pollution. The following results will be obtained, for instance,
when a minute quantity of urine is added to a gallon of water.
(1) The water has a faint odour.
(2) Its colour is greenish yellow in bulk.
(3) On adding a few drops of Nessler’s solution, a deep yellow colour
appears.
(4) A few drops of an acid solution of permanganate of potassium become
yellow when added to it.
(5) Acidify some of the water in a test-tube with nitric acid, then add silver
nitrate solution. Distinct cloudiness is produced, much greater than
with pure tap water.
(6) Addition of hydrochloric acid and barium chloride solution shows a much
greater quantity of sulphates than the same quantity of tap water.
(7) A quantity of the water evaporated in a porcelain dish over a Bunsen’s
flame gives a white residue, which speedily turns brown, with a
urinous odour.
(8) Ignite the ash and add some nitric acid to oxidise it more completely.
Then dissolve in distilled water, and add acid molybdate solution. A
yellow colour, followed by a precipitate, indicates high phosphates and
sewage pollution.
The Complete Systematic Examination is (a) physical, (b) bacteriological,
and (c) chemical. Of the physical tests, colour, which should never be yellow
or brown except for peaty water, is important. Taste is a somewhat uncertain
guide, but any badly-tasting water should be rejected. The odour on heating
to 80° F. in a closed flask may indicate pollution. The degree of hardness can
be roughly tested by rubbing between the hands. The absence of turbidity is
most important, as suspended impurities are more dangerous than all others.
Printed matter should be legible through a column of 18 inches of water.
Microscopally the suspended matter in water which has been allowed to
settle should be examined. Particles of vegetable matter, e.g. fibres of cotton,
linen, cells of potato, or spiral cells of cabbage, are important as indicating
domestic impurities. Bits of wool, hair, wings and legs of insects and epithelium
may be discovered. The presence of algæ, diatoms and desmids, or of water-fleas,
cannot be held to indicate pollution, as these are found in all running streams
and in many wells. The eggs and embryos of worms are much more serious.
Bacteria are almost invariably present in water. The majority of these
micro-organisms are harmless. But as they may number among them the germs
producing diseases like enteric fever and cholera, the estimation of their number
and particularly of any deviation from the number usually present in a given
water, and if possible the detection of special disease-producing bacteria, are
very important. This method has been made more practicable since Koch’s
method of “plate cultivation” of bacteria was discovered. A small quantity
of the water to be examined (kept surrounded by ice until this test is applied,
to prevent multiplication of bacteria in the bottle), is mixed with sterilised
gelatine which has been melted over a water bath. Then the mixture is
spread in a thin layer on a glass plate and allowed to solidify, having been
covered to prevent atmospheric germs from settling on the gelatine. The
bacteria in the water thus become fixed, each growing and forming “colonies”
dotted over the plate. These colonies can be recognised by their size and
appearance, and by sub-culturing according to recognised methods. The85
number of such colonies, and the number of bacteria, from which, presumably,
such colonies sprang in 1 c.c. of filtered Thames water is usually
much below 100; in the water before filtration many thousands are present.
It has been suggested that no water should be regarded as wholesome which
contains more than 100 bacteria in each c.c.
This standard is, however, obviously arbitrary. Chalk water ought to
have a smaller number than this; river waters may have more, and yet be
wholesome. Everything depends on the character of the bacteria found. The
detection of the Bacillus coli communis, which is present in sewage, and normally
in the human intestine, is very suggestive of contamination by sewage. The
bacteriological method of examination of water is still in its infancy.
CHEMICAL ANALYSIS.
(1) The total solids are ascertained by evaporating a given quantity of
the water to dryness, and weighing.
(2) Determination of Chlorine (see page 81).
(3) Determination of Hardness (see page 80).
(4) The Determination of Nitrites is based on the reddish-brown
colouration produced when an acid solution of metaphenylene diamine is
brought into contact with a weak solution of nitrous acid. 100 c.c. of the water
under examination are placed in a clean glass cylinder. Add 1 c.c. of H₂SO₄
solution (1 in 3), then 2 c.c. of metaphenylene diamine solution (5 grains in
1 litre of water with a little H₂SO₄ added). Stir well with a glass rod. If a
colouration is produced at once, a smaller quantity of water must be taken, and
made up to 100 c.c. with pure distilled water. The quantity of nitrous acid
present is measured by introducing different fractions of a c.c. of the standard
sodium nitrate solution3 into similar glass cylinders. Each is then made
up to 100 c.c. with distilled water, and the metaphenylene diamine solution
and acid added as before. The colour develops slowly; time must, therefore,
be allowed in matching.
(5) The Determination of Nitrates can be conveniently made by the
following method. When phenyl-hydrogen sulphate solution is poured upon
a nitrate, and sulphuric acid is formed, picric acid is formed:—
(C₆H₅)HSO₄ + 3 HNO₃ = C₆H₂(NO₂)₃OH + H₂SO₄ + 2 H₂O.
The addition of free ammonia in excess forms yellow ammonium picrate,
the intensity of the colour of which is an index of the picrate, and of the
nitrate from which it was produced. (a) Evaporate 25 c.c. of the water under
examination, and (b) 5 c.c. of standard KNO₃ solution (containing 1 part N
in 100,000) to dryness in two porcelain dishes over the water bath. Add
1 c.c. of phenyl-sulphate solution to each of these as soon as cool, stir well
with a glass rod, then add 1 c.c. distilled water to each dish and 3 drops of
strong H₂SO₄. Next add 25 c.c. of water to each dish, and after heating for
five minutes over the water bath, add solution of ammonia to each dish in
excess. A yellow colour is produced in proportion to the amount of nitrate
present. Transfer the liquids to glass cylinders, and dilute each to 100 c.c.
Take 50 c.c. of the solution showing the least colour, and dilute the other with
distilled water, until it has the same tint.
Supposing the 100 c.c. of the sample required to be diluted to 150 c.c.—
Then the amount of N will be 150 ∕ 100 × 5 ∕ 25 = ·3 parts per 100,000.
If the two solutions (a) and (b) when diluted have the same tint, then the
Amount of N in the sample = 5 ∕ 25 = ·2 parts per 100,000.
(6) Determination of Organic Matter. Frankland’s combustion
process involves the use of delicate and costly apparatus, and is seldom86
employed. In this process the organic carbon is evolved as carbonic acid,
and the nitrogen as such.
Wanklyn’s ammonia process is based on the reduction of organic
matter to ammonia. Part of this ammonia, free or saline ammonia, is
simply combined with carbonic, nitric, or other acids, or is easily derived
from the urea of urine, CH₄N₂O + 2H₂O = 2(NH₄)₂CO₃. Another part is only
set free when the water is boiled with a strongly alkaline solution of permanganate
of potassium. This is called the albuminoid ammonia.
In carrying out this method, a retort is taken, and after having been washed
out, first with a little sulphuric acid, and then with some of the water to be
analysed, 500 c.c. of the latter is put in, and the retort is connected with a
condenser, and distillation begun; 50 c.c. of the distilled water is collected in
a cylindrical glass tube called a Nessler glass. To this 1½ c.c. of Nessler’s
reagent (mercuric iodide dissolved in a solution of potassic iodide and made
alkaline by potass) are added. A rich brown colour is produced, if any
ammonia is present in the distillate. The amount of ammonia in the distillate
is determined by exactly imitating its colour by adding a known quantity of
a standard solution of ammonium-chloride to 50 c.c. of ammonia-free distilled
water, and then Nesslerising as before. Each c.c. of the dilute standard
ammonium chloride solution is equivalent to ·00001 gramme of ammonia (NH₃).
If the first 50 c.c. of water distilled over gives only a slight colouration with
the Nessler solution, no more water needs to be distilled over for free ammonia.
If more is present, two more 50 c.c.’s must be distilled over, and the amounts
of the standard solution required for imitating the test in each Nesslerised
50 c.c. added together. Thus, if 2 c.c. were needed. This
= ·00002 grm. NH₃, which is contained in 500 c.c. of the water
= ·00002 × 200 = ·004 parts saline NH₃ in 100,000 of water.
The free ammonia having been distilled over, 50 c.c. of an alkaline
permanganate solution (containing 8 grammes KMnO₄ and 200 grammes of NaOH
in 1100 c.c. of distilled water, boiled until the bulk is reduced to 1,000 c.c.) is
poured into the retort, and distillation is begun again. Three successive 50 c.c.’s
of water are collected, and then the distillation stopped. Each of these is
Nesslerised, and the tint imitated as before with standard ammonia solution.
The three amounts of ammonia thus found to be present are added together;
and when multiplied by 200, we obtain the amount of albuminoid ammonia in
100,000 parts of water. This test is universally employed by water analysts
along with the next test.
The amount of Oxygen Absorbed from permanganate of potassium is
regarded as an approximate test of the amount of organic matter in water.
Qualitatively this forms a favourite method of testing the purity of water. Two
glass cylinders are taken, one filled with distilled water, one with the water to
be tested. To each is added a given small amount of an acid solution of
permanganate of potassium. The distilled water to which permanganate has
been added will retain its pink colour; while, if the water being tested is very
impure, it will speedily become decolourised. The rapidity and degree of
decolourisation are a rough test of the amount of impurity. A rapid decolourisation
proves the presence of organic matter having an animal origin, or of
sulphuretted hydrogen, iron, or nitrites. Sulphuretted hydrogen is rarely
present, and can be easily recognised by its smell; iron or nitrites are readily
distinguished by their appropriate tests. In the absence of these, the rapid
discolouration is an indication of animal contamination.
To Determine the Amount of Oxygen Absorbed, two glass-stoppered
bottles, each holding about 350 c.c. are required. Into one, 250 c.c. distilled
water, and into the other the same amount of the water under examination
are placed. To each are then added 10 c.c. of standard permanganate of
potassium solution4 and 10 c.c. of a standard pure 25 per cent sulphuric acid
solution. The two bottles, after being shaken, are placed in a water-bath at87
27°C for four hours. At the end of this time add a few drops of potassium
iodide solution to each bottle. The pink is now replaced by a yellow colour.5
A standard thiosulphate solution (Na₂S₂O₃, 5H₂O)6 is placed in a burette.
From this the thiosulphate solution is run into the control bottle until the yellow
colour almost disappears. Now a few drops of starch solution are added, and a
blue colour is produced. The thiosulphate is then added cautiously until all
the blue colour disappears. The amount of thiosulphate necessary for this is
read off on the burette. The same process is repeated with the bottle containing
the sample of water. The starch acts as an indicator. The amount of iodine
liberated is an index of the amount of permanganate in the water, which has
not been used up by its impurities. The amount of iodine liberated is measured
by the amount of thiosulphate required to decolourise the solution. Thus—
2 Na₂S₂O₃ + I₂ = 2 NaI + Na₂S₄O₆.
Suppose that 20 c.c. of thiosulphate solution were required to decolourise
the iodine liberated in 250 c.c. of a sample of water, while the distilled water
required 25 c.c. Then 25 c.c. thiosulphate represents 10 c.c. of the permanganate
solution = ·001 grains of available oxygen.
25-20 = 5
As 25 c.c. = ·001 grm. O, 5 c.c. = 5 ∕ 25 of ·001 = ·0002 grm.
This is the amount of O absorbed by 250 c.c. of the sample.
Therefore the amount of O absorbed by 100,000 c.c. of the sample. = ·08 grm.
It is usual to make a similar determination of the amount of oxygen
absorbed in fifteen minutes.
The Interpretation of Results of analysis is more difficult
than the analysis. A single analysis may be misleading, unless the
source of the water is known. Constancy in composition or analysis
is almost as important a criterion of purity as the actual character
of the constituents. A knowledge of the source is essential in
interpreting results of analysis, as the chemical composition of water
varies with its source. The following rules are only approximately
correct, and are subject to the above general considerations. The
total dissolved solids in river-water are usually 10 to 30 parts in
100,000. Shallow well-water may contain from 30 to 200 parts or
even more, and deep well-water from 20 to 70 parts.
Saline Ammonia in water is commonly of animal origin,
ammonia (NH₃) being one of the first products of decomposition
of nitrogenous animal refuse. Upland surface water usually
contains about ·002 parts per 100,000, but it may reach ·008
or more if the land over which the water passes has been
manured. Shallow well-water may be free from ammonia, or this
may be very excessive in amount. Deep well-water may contain no
ammonia or any amount up to ·1 per 100,000. Its presence is
suspicious if the albuminoid ammonia is above a trace, or if the
oxygen absorbed is appreciable in amount. Generally water is
suspicious if saline ammonia is up to ·01 per 100,000. Albuminoid
Ammonia indicates the amount of organic nitrogenous matter present
in the water. It should not exceed ·005 parts per 100,000, while at
the same time the saline ammonia should not usually exceed ·01 per88
100,000. For Oxygen consumed the following table of the weight of
oxygen required for 100,000 parts of water is given by Clowes and
Coleman:—
| UPLAND SURFACE
WATER. |
WATER FROM
OTHER SOURCES. |
Water of
Great purity |
Not exceeding ·1 |
Not exceeding ·05 |
| Medium purity |
From ·1 to ·3 |
From ·05 to ·15 |
| Doubtful purity |
From ·3 to ·4 |
From ·15 to ·20 |
| Impure |
Exceeding ·4 |
Exceeding ·20 |
The presence of more than 1 and still more so of 2 grains of
Chlorine per 100,000 of water is most suspicious, except in saline
districts. Nitrites if present in an appreciable quantity indicate
comparatively recent contamination by sewage. In deep well-water
they may be produced by deoxidation of nitrates. Nitrates in
upland surface waters should not be equivalent to more than ·03 of
N. per 100,000; in shallow well-waters the amount varies greatly;
in deep well-waters it may be excessive. As a rule it ought not to
be equivalent to more than 5 parts of N. per 100,000 of water; but
the significance of nitrates depends greatly on the source of the
water and on the amount of the other constituents present.
Chemical analysis alone cannot ascertain the safety of a given
drinking water. A minute amount of impurity inappreciable to
analysis may be competent to produce disease; while another water
may be drunk with impunity, which contains considerable organic
matter. Chemical analysis “can tell us of impurity and hazard,
but not of purity or safety” (Buchanan). An accurate opinion as
to the character of a drinking water can only be expressed when one
knows the amount of each chief constituent (as above), and whether
these amounts deviate from the same water at other times or from
other waters in the vicinity.
89
CHAPTER XII.
ORIGIN AND EFFECTS OF THE IMPURITIES OF
WATER.
Origin of Impurities of Water.—Parkes classifies impurities
of water as:
1. Those Received at the Source.—The character of water
varies with the geological structures through which it has passed;
with its origin from the subsoil or cultivated land, or deep wells, or
graveyards, or near the sea, etc. It is a mistaken policy to
commence with an impure water and proceed to purify it; though
communities supplied from rivers may be compelled to submit to
this. They must then insist on the most stringent measures of
purification (see p. 96). Inorganic impurities are of much smaller
consequence as regards health than organic; hence the great
advantage of deep well-water over river water. It has been
suggested, however, that when deep well-water becomes polluted,
it is more dangerous than equally polluted river-water, because in
the latter the normal bacteria of water are more abundant, and
possibly interfere with the continued life in water of disease-producing
bacteria. This statement is unproved; and if correct, is
rather an indication for further precautions being taken to prevent
access of pollution to deep wells, than in favour of the continued use
of river-water.
2. Impurities of Transit from Source to Reservoir,
acquired during the flow in rivers, canals, or other conduits. These
impurities have been broadly divided by the Rivers Pollution
Commissioners into “sewage” and “manufacturing;” the
former including the solid and liquid excreta, the house and waste
water, etc.; the latter including the refuse from manufacturing
processes, as from dye and bleaching works, tanneries, etc.
3. Impurities of Storage, whether in wells, reservoirs, or
cisterns. Organic impurities are commonly received at this stage.
A well, for instance, drains the soil around it in the shape of an
inverted cone, with a very broad base, unless the entrance of water
from its sides is prevented.
4. Impurities of Distribution. Lead, and occasionally other
metals, are dissolved by certain waters. If the pipes are left empty,
as with an intermittent supply, sewage may be drawn into them; in
a few cases coal-gas has found its way into the water pipes
(page 76).
Effects of Impure Water.—1. Effects of Mineral90
Impurities. Suspended Mineral matters in unfiltered water occasionally
produce diarrhœa. The hill diarrhœa of some parts of
India has been traced to water containing fine mica particles in
suspension.
Hard water is said by some to be hurtful, but the salts
causing hardness are probably innocuous when not amounting to
more than 12 or 16 grains per gallon. Persons in the habit of
drinking hard water find soft water unpalatable. Hard water has
been thought to favour gout and calculus (stone), but this is not so.
The salts producing permanent hardness are said to be injurious,
producing indigestion, but this is doubtful in the amounts ordinarily
drunk.
Goitre, a swelling of the thyroid gland in the neck, is often
associated with the use of drinking water from magnesian limestone
formations; but that any kind of excessively hard water causes
goitre is very doubtful.
Lead dissolved in water may produce serious and lasting
ailments, and they are often present for a long time before their
cause is detected. The amount of lead capable of producing
poisonous symptoms has been as little as 1 ∕ 100 grain per gallon of
water (Dr. Angus Smith). According to De Chaumont, 1 ∕ 10 grain per
gallon, that is 1 part in 700,000 is usually required to produce such
symptoms. In the well-known case of the poisoning of Louis
Phillippe’s family at Claremount, there was 7 ∕ 10 grain of lead in a
gallon of water; and this affected 34 per cent. of those who drank
it. The symptoms produced by lead poisoning are those of indigestion,
accompanied by colic; a blue line at the junction of the
gums with the teeth; “wrist drop,” a paralysis of the muscles of
the forearm, or some other paralysis; and if the poisoning is
continued, attacks of gout, followed by its usual consequences,
chronic kidney disease. The latter affections chiefly occur when
the poisoning is continued for a long time, as in the case of painters
or type-setters: poisoning from water is generally discovered before
any other than dyspeptic symptoms and colic are produced.
The presence of traces of iron in water may give it a slightly
astringent taste; and such water is liable to cause headache and
constipation.
2. Effects of Vegetable Impurities.—Living plants are
unobjectionable, but decomposing vegetable matter may produce
diarrhœa and other severe symptoms.
3. Effects of Animal Impurities.—Animal impurities of
water are by far the most important from a sanitary point of view.
They are most commonly derived from leaky drains or cesspools, or
from surface accumulations of filth. The quality of the contamination
is more important than its quantity; and this will explain why
water containing a large amount of sewage may be drunk for
a prolonged period with impunity, while at another time the least
trace, if it contain the active germs of disease, will lead to serious
mischief.
Suspended animal impurities are much more dangerous than
those completely dissolved. Hence the examination of the colour91
and turbidity of drinking water is very important. Fæcal contamination
is by far the most dangerous of all, and chiefly so when it is
derived from a patient suffering from some communicable disease,
like enteric fever or cholera.
Certain Parasites occasionally are swallowed with water in
the form of embryo or egg. The liver fluke, round worm, and less
frequently other kinds of entozoa have been introduced in this way.
The occasional swallowing of small leeches has occasionally given
rise to hæmorrhage.
Diarrhœa may be caused by animal contamination of water.
It most often occurs in summer, when all the circumstances are
favourable to active fermentative changes. The summer diarrhœa
of infants is caused by similar changes in milk or other foods. The
presence of fœtid gases in water may lead to diarrhœa. This may
occur when the overflow pipe of a cistern opens into the soil pipe or
into the trap of the W.C.
Dysentery, like cholera and enteric fever, may be propagated
by water contaminated with the stools of a patient suffering from
the same disease.
Malaria or Ague has been stated to be caused by the water
of malarious marshes. The evidence on this point requires revision,
in view of the part which the mosquito is now known to play in the
propagation of this disease (pages 282 and 307).
Enteric (otherwise called Typhoid) fever is most often due to
the drinking of water contaminated with sewage.
The balance of evidence is in favour of the view that in order
to produce enteric fever water must be contaminated with the stools
or urine of a patient who has suffered from this disease. Numerous
instances are on record in which villages, the inhabitants of which
drink sewage-contaminated water, have remained free from enteric
fever, until a patient suffering from it has come to the village, when
the spread by water has been very rapid. Occasionally no known
contamination from a case of enteric fever has preceded the outbreak
of this disease which has been caused by sewage-contaminated
water. It must be remembered on this point that the urine of an
enteric fever patient may occasionally contain large numbers of the
bacillus causing this disease for several months after the patient is
well (page 301).
The contamination of water with sewage may occur in
various ways. In country places surface wells and small streams
commonly supply the drinking water, and these are frequently
contaminated. The illustration (Fig. 9) shows the percolation
of excretory matters from an out-door closet through the porous
gravel, into a neighbouring well; the result being an epidemic of
enteric fever among those who drank the water of the well.
Alterations in the level of the subsoil water are sometimes followed
by an outbreak of enteric fever (p. 70). A sudden fall of rain
occurs, and the excess of water in the soil absorbs the soakings
from country privies or cesspools, and carries them into the nearest
well. The percolation of tainted water through a considerable tract
of land, possibly along fissures, is sometimes insufficient to purify it,92
as proved by a remarkable epidemic in the small village of Lausen,
in Switzerland.
In other cases sewage gains access into leaky water-pipes.
Formerly contamination was occasionally due to improper
connection between the overflow pipe of the cistern and the soil-pipe,
or to the water-closet being flushed by a pipe directly
connected with a water-main (as in the Caius College outbreak at
Cambridge), or connected with the drinking-water cistern (page 76).
Milk may, by the admixture of water, become contaminated
with enteric matter, and produce widespread epidemics. Where the
water is very impure, the small amount used in washing cans may
suffice to cause infection.
Cholera was first proved by Dr. Snow, in 1849, to be due to the
specific contagium of cholera gaining an entrance into drinking
water. This contagium is derived as in enteric fever from the
intestinal evacuations, the urine, and the vomit of patients suffering
from the same disease.
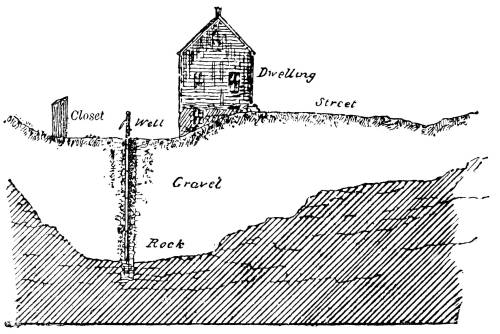
Fig. 9.
The close connection of the spread of cholera with an impure
water supply has been repeatedly shown in this country. The
cholera epidemic of 1854 was very severe in the southern districts
of London. At that period these districts were supplied with
water by the Southwark and Vauxhall Company, deriving its water
from the Thames at Battersea, and by the Lambeth Company,
having its intake at Thames Ditton, where the water was purer.
The two companies were acting in rivalry, so that in many streets
their mains ran side by side; and houses in the same street similar
in all other respects, received a different water supply. An
investigation of the distribution of cholera in these districts gave
the following results:—
|
POPULATION
IN 1851. |
CHOLERA
DEATHS IN
14 WEEKS. |
CHOLERA DEATHS
PER 10,000
OF POPULATION. |
| Houses supplied by Southwark Co. |
266,516 |
4,093 |
153 |
| Houses supplied by Lambeth Co. |
173,748 |
461 |
26 |
93
The facts, when examined in detail, brought out still more
strikingly the exemption of the houses supplied by the Lambeth
Company; the infection picking out in a given street the houses
supplied by the Southwark Company. The great epidemic of
cholera at Hamburg in 1892 proves the same point. Hamburg,
Wandsbeck and Altona are three towns adjoining each other, and
really forming one large community; but while Hamburg suffered
terribly, the two other towns had no cases of cholera, except the
few that were brought into them. In all respects except water-supply
the conditions were alike; but Wandsbeck obtained filtered
water from a lake, Altona obtained filtered water from the Elbe
below the town, while Hamburg was supplied, previous to the
epidemic, by unfiltered water from the Elbe just above the town.
Diphtheria and scarlet fever have never been traced to
polluted water.
Effects of an Insufficient Supply of Water.—The
influence on personal health is most baneful. Water is used
sparingly for purposes of cleanliness, with the necessary results that
cutaneous diseases become more common, and the whole body
suffers; the linen is imperfectly and infrequently washed; the
house becomes dirty; drains are imperfectly flushed; the streets
are not cleaned; and the whole atmosphere becomes loaded
with impurities. According to Parkes, it is probable that the
almost complete disappearance of typhus fever from civilized and
cleanly nations, is not merely owing to better ventilation, but also
to more frequent and thorough washing of clothes.
Insufficient cleansing of the surfaces of streets and of sewers,
owing to a deficient supply of water, has a very important influence
on the spread of enteric fever and epidemic diarrhœa. A heavy fall
of rain often causes a rapid diminution in the prevalence of the
latter disease.
94
CHAPTER XIII.
THE PURIFICATION OF WATER.
When a public water-supply is provided, it may reasonably be
expected to be furnished pure and fit for use; but this, occasionally
is not so. The reports, for instance, of the condition of the London
Water Supply, occasionally show that it is turbid and contains a
slight excess of organic matter. This is especially the case when,
after heavy rainfall, storm-water is brought into the reservoirs, and
owing to deficient storage, sufficient time is not allowed for deposit.
Rain-water always and other waters frequently require to be purified
before use.
Methods of Purification.—The only certain way of
obtaining pure water is by Distillation; but this plan is scarcely
applicable to water on a large scale. Furthermore distilled water
is not so palatable as ordinary water. The distillation of water is
more especially required on board ship, during long voyages. It
should be followed by the use of some measure to secure efficient
aeration.
2. Boiling water serves to remove the temporary hardness,
and the chalk carries down with it a large proportion of any organic
matter that may be present. Boiling deprives the water of its
dissolved gases, and renders it flat; it is desirable, therefore, to aerate
it by filtration or from a gazogene after boiling. All the microbes
which are known to produce disease are destroyed by efficient boiling.
Certain putrefactive microbes are more persistent of life, owing to
the fact that they form spores, which are not killed at the
temperature of boiling water. Tyndall showed that by boiling the
liquid containing these spore-forming microbes on three successive
days, thus giving time for the spores to develop into less
resistant microbes, they could be effectually destroyed. Boiled
water will not cause enteric fever or cholera, the two chief water-borne
diseases.
3. The exposure of water in divided currents to the air by
passing it through a sieve has been proposed as a means of
purifying water, but it is inefficient when trusted to alone. Plants
in reservoirs help to absorb organic matter; and fish, by destroying
small crustaceans, have been found useful. Hard waters do not
bear exposure to light, as a thick green growth of chara occurs,
which may block pipes, and give a bitter taste to the water.
4. The Addition of Chemical Substances.—(1) Clark95e’s
process consists in adding milk of lime, i.e. an emulsion of quicklime
with water, to the water in the reservoir on a large scale. By
this means calcium carbonate is precipitated, but no effect is
produced on calcium and magnesium sulphates and chlorides. The
hardness of the Thames water can thus be reduced from 16° to 3°
or 4° (Clarke’s scale). The calcium carbonate carries down with it
suspended and possibly dissolved organic matter. In the Porter-Clarke
process lime-water, i.e. milk of lime diluted, and the excess of
lime separated by settlement or filtration, is mixed with the water to
be purified, the water being freed from the precipitated calcium
carbonate either by subsidence or by being forced through a filter
of stretched canvas.
(2) Carbonate of Soda added to boiling water throws down
calcium carbonate, and possibly lead if present. Much less is
required when added to boiling than to cold water. Maignen’s
process consists in adding anti-calcaire powder, containing chiefly
carbonate of soda, lime, and alum.
(3) Aluminous salts are very effectual in removing suspended
organic matter, if the water contains calcium carbonate. On the
addition of alum, calcium sulphate and aluminium hydrate are
formed, both of which fall to the bottom, carrying with them other
impurities. The amount of alum required is about 6 grains per
gallon of water. If the water is not hard, a little calcium chloride
and carbonate of soda should be put in before the alum is added,
in order that a precipitable substance may be formed.
(4) Potassium permanganate readily removes the offensive smell
of stagnant water, but it gives a yellow tint to the water. The
addition of a little alum will help to carry down the decomposed
permanganate.
(5) Perchloride of Iron, in the proportion of 2½ grains to a
gallon of water, has been found to completely purify water from
finely suspended organic matters and clay.
(6) More recently, other substances, such as iodine and hyposulphite
of soda, have been recommended. These are supposed to
act by sterilizing the water, and iodine in suitable quantities
undoubtedly effects this.
Chemical processes for the purification of water, with the
exception of the softening process, are not to be recommended for
general use. Efficient filtration, or boiling, is safer than chemical
treatment; and it would only be justifiable to trust to the latter,
when, as in a military campaign, an attempt at purification was
necessary, and no means were available for filtering or boiling
water.
7. Filtration.—The object of filtration is to remove the
impurities of water. The most dangerous impurities are suspended
in it, especially the microbes causing infectious diseases. Hence
the most perfect filter is the one which most completely prevents
the passage through it of microbes. If the water supply is pure,
domestic filtration is not only useless, but likely to do more harm
than good. This is true for such upland surface waters as those
supplied to Liverpool, Glasgow, and Manchester; for such deep96
well-water supplies as those of Brighton (deep chalk), of Nottingham
(new red sandstone), and others, when pumped from wells
remote from inhabited houses. For upland surface waters known
to attack lead pipes, filtration through charcoal or spongy iron may
be advisable; for river water, filtration through a germ-proof filter
is best.
Filtration on a large scale is generally carried on as follows:—A
preliminary step consists in collecting the water into settling
reservoirs, wherein the more bulky substances subside. The water
is then filtered through beds of gravel and sand, containing
perforated tubular drains below, into which the filtered water flows.
The drains are covered by a bed of gravel about 3 feet deep, over
which is spread a layer of sand about 1½ to 2 feet deep. Sharp
angular particles of sand are the best; and the gravel should
gradually increase in its coarseness as it descends.
The effect of this filtration is chiefly mechanical; it separates any
suspended matter, whether organic or inorganic. A certain amount of
biological action possibly also takes place. Piefke found that a perfectly
cleaned and sterilised filter when first used, increases the microbes
in water, instead of decreasing them. Gradually a gelatinous
layer of slimy matter is formed on the top of the sand; the
water now filters through much more slowly, but it gradually
becomes freer from microbes, these being intercepted by the slimy
layer. It is important that this layer should not be disturbed by
too rapid or forced filtration, and that when the surface layer
requires to be removed, because the filter has become impervious,
time should be allowed for another thin film to form before the
filtered water is again utilised. Koch concluded that the rapidity
of filtration should never be allowed to exceed 100 millimetres
(about 4 inches) per hour; and that the number of microbes per
c.c. in the filtered water should never exceed 100. Some oxidation
of organic matter, as well as detention of microbes, may take place
during the filtration of water, nitrates being formed by the vital
activity of certain “nitrifying” microbes in the filter. (On nitrification,
see pages 195 and 274.) P. Frankland’s observations show
that the number of microbes in Thames water is reduced by filtration
through sand and gravel beds, as practised by the London
Water Companies, so that only 3·4 per cent. of those originally
present remained. He also concludes that the majority of the
microbes present in filtered water are derived from post-filtration
sources. Thus the number is greater in tap-water than in water
derived from near the reservoirs.
Other materials besides sand have been used for filtration on a
large scale, but none with proved success.
Domestic Filtration ought, as already explained, not to be
needed, but circumstances often arise in which the public supply is
open to suspicion, and a second domestic line of defence against
infection through the water supply is desirable. When this is so,
the form of filter which will best protect the household is one
attached to the house-tap, so that all drinking-water is perforce
filtered. When filtering involves the transfer of water from the97
tap to the interior of the filter, opportunity is left for carelessness or
forgetfulness. The one essential point of a domestic filter is that
it will prevent the passage through it of microbes. Every filter
must be tested from this standpoint.
On this point the experiments of Woodhead and Cartwright
Wood are conclusive. They first of all experimented on various
filters with fine artificial ultramarine containing particles 16 µ to
0·6 µ or even less in diameter in suspension; and milk containing
granules and globules of fat 0·5 µ to 30 µ or more in diameter,
freely diluted with water.
|
TIME IN MINUTES
REQUIRED FOR
FILTRATION OF 1
PINT OF WATER. |
PRESENCE OR
ABSENCE OF
ULTRAMARINE IN
FILTRATE. |
PRESENCE OR
ABSENCE OF MILK
IN FILTRATE |
| Silicated carbon filter |
68 |
++ |
+++ |
| Carbon filter |
18 |
+ |
+++ |
| Maignen’s Filtre Rapide |
4 |
0 |
++ |
| Spongy iron filter |
14 |
0 |
+++ |
| Pasteur-Chamberland filter |
420 |
0 |
0 |
| Berkefeld filter |
140 |
0 |
0 |
The number + indicates the relative amount of the experimental
substances that made their way through the filtering medium.
Experiments were then made with the actual microbes of
certain infectious diseases, and it was found that certain filters allow
these to pass. Thus a silicated carbon filter allowed 1,000 out of
15,000 typhoid bacilli suspended in water to pass through its
substance; a manganous carbon filter allowed 600 to 800 out of
10,000 cholera vibrios to pass through; Maignen’s filter on the
second day of experiment allowed 150 out of 5,000 cholera vibrios
to pass through; Lipscombe’s charcoal filter experimentally only
reduced typhoid bacilli from 20,000 to 5,000; the magnetic carbide
filter only reduced them from 20,000 to 10,000; the spongy iron
filter from 20,000 to 3,000; while, on the contrary, the Pasteur-Chamberland
and the Berkefeld filter completely stopped all
microbes and produced a sterile water. (As to these two, see
page 98.)
Of the materials enumerated animal charcoal was formerly
regarded as an excellent filtering medium. It is capable of
oxidising organic matter dissolved in water, but so far from
sterilizing water, it favours the growth of microbes in it. Water
filtered through charcoal, after the first few days of use of the
charcoal, deteriorates, as the charcoal yields up impurities to it.
Manganous Carbon consists of animal charcoal and black
oxide of manganese mixed with oil, and heated strongly together
out of contact with the air. The oxidising power of the carbon is
said to be thus greatly increased. It shares the objections to
carbon.
Silicated Carbon consists of 75 per cent. of charcoal and
22 per cent. of silica, with a little oxide of iron and alumina.
It is not an efficient filtering medium.
98
Spongy iron is prepared by the reduction of hæmatite ore with
fusion, so that the iron is obtained in a porous and finely-divided
condition. The Rivers Pollution Commissioners found spongy iron
to be “a very active agent, not
only in removing organic matter
from water, but also in materially
reducing its hardness, and otherwise
altering its character.” It
is a powerful oxidising agent, some
of the water being decomposed,
and hydrogen set free, and the
oxygen acting upon any organic
matter present. It also removes
lead from water. As already seen,
it does not, however, fulfil the
primary object of water, by depriving
it of any microbes contained
in it.
Magnetic carbide of iron is
obtained by heating hæmatite ore
with sawdust. Its action is
similar to that of spongy iron.
The Pasteur-Chamberland
filter consists of a cylinder of
unglazed fine porcelain made from
a well-baked Kaolin of a certain
degree of porosity and hardness.
(Fig. 10.)
The water passes through the
porcelain from without inwards,
and with the pressure of 1½ to
2½ atmospheres which is usually
present in the pipes of a water-service,
passes through at the rate
of about three quarts per hour.
The filter can easily be cleaned by
brushing it in a stream of hot
water, or by subjecting to the
heat of a Bunsen burner. The
filtration is entirely mechanical,
the filtered water being quite freed
of microbes. No chemical action
takes place.
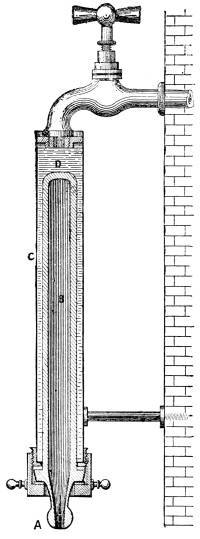
Fig. 10.
Pasteur-Chamberland Filter.
A.—Outlet of filtered water. B.—Pasteur
tube. C.—Metal tube containing unfiltered
water. D.—Unfiltered water
delivered through tap.
The Berkefeld filter is cylindrical
like the Pasteur-Chamberland
filter, and is used in the
same way. It is made of infusorial
earth, which is soft and friable
and liable to break. The cylinder
becomes gradually worn thin by
cleaning, and it then ceases to filter efficiently. Its sole advantage99
over the Pasteur-Chamberland filter is the more rapid rate of
filtration; and against this is to be set the greater liability to
fracture and the lack of continuance of efficient filtration. Woodhead
and Wood in the report already quoted, state: “The Berkefeld
filter appears to have the largest pores among the efficient filters,
as is evidenced by the fact that the water organisms were not
apparently weakened, that more species of organisms appeared in
its filtrate, and that lowering the temperature to 11° C. did not
prevent their appearance. The Pasteur-Chamberland filter, on the
other hand, at 11° C. was able to give an apparently sterile filtrate
for a prolonged period.” More recent experiments have shewn
that pathogenic (disease-producing) microbes contained in water
after awhile grow through the substance of a Berkefeld filter,
and that this does not happen with a Pasteur-Chamberland filter.
The latter is therefore preferable.
In determining the number of bougies required for any filter
to secure a given amount of pure water, it is necessary to
calculate on the basis of the output after several weeks’ use, not
on the original output. If this is done, pure water will be secured
without disappointment as to the amount supplied.
100
CHAPTER XIV.
COMPOSITION AND PROPERTIES OF AIR.
An abundant supply of fresh air is necessary at all times.
And yet its importance is commonly ignored in practical life.
Strenuous efforts are made to ensure a supply of food, and
water is commonly filtered or otherwise purified before drinking;
but many are content to live in an impure atmosphere, which
hardly suffices for the preservation of life, and certainly not of
health. Deprivation of food, or even of water, only kills after
several days or weeks; deprivation of air kills in a few minutes.
Only about three pints of water are required daily, while at least
1,500 gallons of air are necessary every day for carrying on the vital
functions.
Composition of Air.—The air constitutes a gaseous ocean in
which we live, as fishes live in water. In virtue of its weight, it
exerts a pressure of about 15 lbs. on every square inch. This
pressure is usually measured by the barometer, and is equivalent on
an average to that of a column of 30 inches of quicksilver. (See
page 331).
Chemically, air consists of a mixture of various gases and
vapours. These are chiefly Oxygen and Nitrogen; but in addition,
there are minute quantities of carbonic acid, argon, hydrogen, water
vapour, ammonia, ozone, and suspended matters.
The oxygen and nitrogen exist, in the proportion by volume of
20·9 of oxygen to 79·1 of nitrogen, or of 23·16 grains of oxygen to
76·84 of nitrogen, by weight.
These two gases do not exist in chemical combination, but
mechanically mixed. This is proved by the fact, that they do not
exist in air in the proportion of their combining weights, or any
multiple of these; that the proportion varies slightly at different
parts; and that the air which is dissolved in water does not contain
the nitrogen and oxygen in the proportion 4 to 1 (as in the atmosphere),
but in the proportion 1·87 to 1. This means that oxygen,
being more soluble in water than nitrogen, has dissolved in a larger
proportion; as it certainly would not have done, had the oxygen
and nitrogen been chemically combined. The oxygen dissolved in
water supplies fishes with the necessary oxygen for their respiratory
processes. Similarly the oxygen in the atmosphere is its most
essential constituent, being required in all processes of oxidation
(i.e., combustion), whether in living organisms or in the inanimate
world. Nitrogen serves as a diluting agent. It is incapable of101
supporting life alone; and many of the fatal accidents which have
occurred through men descending deep wells without first testing,
by means of a lit candle held well below them, the quality of the
air near the bottom, have been due to an accumulation of nitrogen
in the well.
Ozone is a condensed form of oxygen, which is present in
minute quantities in pure air, and especially during a thunder-storm
or after a fall of snow, and in the air near the sea. In it three
volumes of oxygen are condensed so as to occupy two volumes.
In this condensed condition it has powerful chemical affinities;
often oxidising substances which oxygen cannot attack. It is
generally absent from the close air of towns and dwelling houses,
having been used up to oxidise the organic matter present in these
places. Air without it is said to be “devitalised”; and ozone has
been described as the scavenger of the air.
Ozone can be produced by hanging a piece of moist phosphorus
in a room; and it is stated by Dr. Daubeny, that part of the oxygen
given out by plants, especially by scented flowering plants, is in the
condition of ozone. A small quantity is produced when an
electrical machine is worked; its presence is evidenced by a
peculiar smell (the name ozone is derived from the Greek word for
smell).
Test of Ozone in Air.—Traces of ozone in air are detected by exposing
strips of blotting paper moistened with a mixture of a solution of potassic iodide
and starch. If ozone is present, the paper assumes a blue tint, due to the
liberation of iodine, and its combination with the starch. Other acid gases may,
however, produce the same effect. A second test should, therefore, be tried.
Soak red litmus paper with a very dilute solution of potassic iodide, and expose
as before. Potassic oxide is produced if ozone is present, and this turns the
litmus blue.
Aqueous Vapour is always present in air, though the amount
varies greatly. It is invisible in the ordinary condition, but by
condensation becomes cloud or fog, rain, snow, or hail. The
quantity of moisture present varies with the temperature of the
air; the higher the temperature, the more water can be vaporised,
without the point of saturation being reached. An increase of
27° Fahr. doubles the capacity of air for moisture. The amount of
moisture that would saturate air at 50° Fahr. only gives 71 per
cent. of the saturation amount at 60° Fahr. The amount of
moisture is estimated by the hygrometer (page 240).
Air saturated with moisture at 32° Fahr., holds vapour equal
to 1 ∕ 160 of its weight; at 59° it holds 1 ∕ 80, at 86° 1 ∕ 40, at 113° 1 ∕ 20, and at
140° 1 ∕ 10.
Ammonia in normal air does not exceed one part in a million
of air; but it is always present—either as free ammonia or as
sulphate, chloride, carbonate, or sulphide of ammonia. From this
source, plants derive some of the nitrogen they require as food;
some also from the free nitrogen, which is fixed by certain
microbes, growing in the nodules connected with the roots of peas,
lentils, and other plants (page 274).
102
Traces of nitrous and nitric acid are also present in the air,
produced by the direct combination of nitrogen and oxygen
occurring as the result of the electric spark during lightning.
Carbonic Acid or carbon dioxide is always present in air, in
the proportion of 3·36 to 4 parts in 10,000; but in impure air may
be present in much larger amount. It is a heavy gas, incapable of
supporting combustion, and therefore of supporting animal life.
Being a heavy gas, it tends to accumulate where it is produced, as
about lime-kilns by the heating of chalk. Thus CaCO₃ (chalk) (heated) =
CaO (lime) + CO₂ (carbonic acid). Tramps have occasionally died of carbonic acid
poisoning through sleeping near lime-kilns.
It is produced by the oxidation of carbonaceous matters, hence
in all ordinary combustion, in many cases of putrefaction and
fermentation, and in the respiratory processes of all animals.
Plants diminish the amount of carbonic acid in the atmosphere.
Two processes occur in most plants: a process of
respiration, as in animals; and a process of assimilation, by which
the leaves and all other green parts of a plant under the influence of
sunlight decompose the carbonic acid of the atmosphere, fixing its
carbon and liberating its oxygen. Plants such as fungi, which are
destitute of green colouring matter, cannot decompose carbonic acid;
nor can any plants during the night. During the day green plants
are air purifiers; during the night all plants vitiate the air to a
slight extent.
The Air in Relation to Respiration.—The oxygen of air
is absolutely essential for the continuance of life. In every
organised animal, lungs or analogous organs are provided, in order
to supply the necessary oxygen to the system, and to remove the
impure air from it.
The act of breathing occurs in man about seventeen times per
minute. While the inspired air is in contact with the interior of the
lungs, it undergoes important alterations. It comes into contact
with the five or six millions air-vesicles which form the minute
dilated terminations of the windpipe, and have an aggregate area of
ten to twenty square feet. Each of the air-vesicles has extremely
thin walls; and outside these delicate walls lie capillary blood-vessels,
full of impure blood. An active interchange now
occurs between the air and the gases dissolved in the blood.
Oxygen passes through the intervening membrane into the blood,
while carbonic acid and other impurities of the blood pass
into the air-vesicle. The consequence of this is that the impure
dark-coloured blood becomes bright scarlet and pure. This
purification is not confined to any one portion of the blood; for the
heart contracting 60 or 70 times per minute, pours successive
portions of blood into the capillaries surrounding the air-vesicles;
while at the same time, pure air is brought into the air-vesicles
seventeen times per minute, and so the interchange is constantly
kept up.
In view of the incessant character of respiration and circulation,103
it is clear that all the blood will be purified if the external air is
pure; and that if there is any detrimental matter in the air, it
probably will come into contact with the blood in the lungs.
The amount of air taken in with each inspiration is about thirty
cubic inches. This is called the tidal air, as it is constantly ebbing
and flowing from and to the lungs. By means of a very forced
inspiration, about 100 cubic inches of additional air can be inspired;
and similarly after an ordinary inspiration, one can expire forcibly
an additional 100 cubic inches, though there will still be left in the
lungs another 100 cubic inches of air. Thus:—
| Tidal air |
30 |
cub. in. |
| Complemental air |
100 |
„ |
| Supplemental air |
100 |
„ |
| Residual air |
100 |
„ |
| —— |
| Total capacity of lungs |
330 |
„ |
Corresponding to the respiratory changes in the lungs, there
are changes in the tissues throughout the body. The pure and
oxygenated blood leaving the lungs, is carried to all parts of the
system. Oxidation and allied processes are actively carried on, the
result of which is the formation of urea, carbonic acid, and smaller
quantities of other effete matters. These are then carried by the
blood to the excretory organs, urea being chiefly eliminated by the
kidneys, and carbonic acid by the lungs.
Examination of Expired Air shows that—1. It is heated; in
its passage through the nose and deeper respiratory passages it has
acquired a temperature approaching that of the blood.
2. Its moisture is increased. By the skin and lungs from 25 to
40 ounces of water pass off in the twenty-four hours; the relative
amount varies somewhat.
3. It contains 4 to 5 per cent. less oxygen, and 4 per cent. more
carbonic acid than inspired air. The carbonic acid, instead of being
4 parts in 10,000 of air, becomes over 400 in 10,000, while the
oxygen is diminished in a somewhat larger proportion. Thus:—
|
OXYGEN. |
NITROGEN. |
CARBONIC
ACID. |
| Inspired air contains |
20·81 |
79·15 |
·04 |
| Expired air contains |
16·033 |
79·557 |
4·38 |
The amount of carbonic acid expired varies under different
circumstances. It is increased by active work, by an increase of
food, by a diminution of the external temperature; it is greater
when the surrounding air is pure, and when it is moist; and it varies
with the season, being greatest in spring, and least in autumn.
Children require more oxygen, and expire more carbonic acid
than adults, weight for weight. A child six or seven years old
requires nearly as much oxygen as one twice that age. Boys
usually require more air than girls, as they are more active and
exhale a larger amount of carbonic acid and other impurities.
104
The average amount of carbonic acid eliminated by a healthy
adult is at least 0·6 cubic foot per hour, or 14·4 cubic feet per day.
This reckoned as carbon is equivalent to 160 grains per hour, or
half a pound of carbon in the twenty-four hours. Liebig gives the
amount of carbonic acid expired as 0·79 cubic foot per hour, or
19 cubic feet per day.
4. It contains organic impurities. These are chiefly gaseous,
solid particles only being expired during coughing, or possibly
during conversation. The danger from the “breath” of patients in
infectious diseases is really associated rather with the dried
discharges on handkerchiefs, etc., than from the “breath” itself;
unless droplets of saliva discharged during speaking, or mucus
during coughing, are directly inhaled.
105
CHAPTER XV.
SUSPENDED IMPURITIES OF AIR.
Pure air being essential to life and health, it is important to
ascertain the character and origin of the impurities of air.
Innumerable substance—in the condition of gases, vapours, or
solid particles—constantly pass into it, and deteriorate its quality.
To counteract this, certain purifying agencies are at work, the
mechanism of which will be considered hereafter.
Impurities are much commoner and more abundant in the air
of enclosed spaces than in the external air, as the natural processes
of purification cannot be brought to bear so efficiently in the former
case. In sick rooms, hospitals, etc., impurities arise, which are
not present where only healthy people are collected. The most
important impurities are derived from the respiration of animals,
and the combustion of gases, candles, or lamps in rooms, from
sewage emanations, from various occupations, and the air of
marshes, mines, church-yards, etc. These may be classed under
two heads—solid and gaseous; the solid being simply suspended in
the air in a finely divided condition, or floated about in a coarser
condition by currents of air. They are revealed in an atmosphere
in which one did not previously suspect their existence, by the
passage of a beam of sunlight. Light itself is invisible, but its
course is rendered visible by the particles from which its rays are
reflected. Tyndall demonstrated the presence of minute particulate
matter in the air of all ordinary situations, and showed that a large
proportion of this matter consists of germs (microbes). In his
experiments with vapours in closed tubes, floating matter was
always revealed by a concentrated beam of light, even though the
air entering the tube had been first drawn through sulphuric acid
and through a strong solution of caustic potash. If this air was
then passed through a red-hot platinum tube and across folds of red-hot
platinum gauze, it became optically empty; the floating matter
had been burnt, and disappeared. It was therefore organic. In
subsequent experiments, he took organic solutions, as of meat,
turnip, and the like, and rendered them sterile by repeated boiling.
They remained sterile when kept in air-tight vessels or in vessels
covered with a thick layer of cotton-wool, which would efficiently
filter any entering air; but when exposed to the air, they invariably
became turbid, owing to an enormous multiplication of germs.
Clearly, therefore, air contains organic, matter, and much of this
organic matter consists of living germs. Most of these germs are
comparatively harmless under ordinary conditions. They are,106
however, the causes of fermentation, putrefaction, and all the
processes of decomposition which occur in organic substances.
The importance of the exclusion of the dust of air has received an
important application in Lister’s antiseptic and in the aseptic system of
treatment of wounds. Formerly accidents and operations were
frequently fatal; now vast numbers of lives are saved by improved
surgical methods. The original antiseptic method acted on the
supposition that some germicidal application to the wounds was
necessary; now it is realized that if, during the operation, germs
are not allowed to remain in the wound, all that is afterwards
necessary to insure rapid recovery is that they shall be prevented
from entering the wound from the external air during its process of
recovery. By the adoption of such means, large wounds can be
made to heal, without the formation of a drop of “pus” or
“matter.” (See also page 110.)
Suspended Matters are mineral or organic, the two being
commonly associated together. The mineral matters consist largely
of fine particles of common salt, silica, clay, iron rust, dried mud,
chalk, coal, soot, and similar substances. Not uncommonly the
mineral particles are coated by, or mixed with, organic matter, the
comparative lightness of the organic matter enabling the mineral
matter to float about more easily. The objection to dust is thus
intensified, for not only is it irritating to the respiratory passages
and generally disagreeable, but it carries with it putrescent and
possibly morbific particles. The prevention of infectious diseases
resolves itself largely into means for preventing the inhalation of
dust.
Organic Suspended Matters in the open air are, most
commonly, minute fragments of wood and straw, dried horse litter,
fragments of insects, the spores and pollen of plants, and microscopic
plants and animals. In addition, there is the putrescent organic
matter resulting from respiration and other organic functions.
Indoors, the air commonly contains, in addition, fragments of
cotton, linen, silk, or other fibres, fragments of vegetables, starch
cells, soot, charred wood, splinters from floors, etc.
In Sick Rooms, products of the morbid conditions may be
evolved; thus, pus-cells, particles from the expectoration, blood
cells, fat particles, epithelium, or the special germs or microbes to
which infectious diseases are due. These are disturbed by the
movements of persons, causing the dust to rise; and thus the
infection of consumption, and of the acute infectious diseases, is
frequently spread.
Flies and other winged insects are important auxiliaries in the
diffusion of disease-carrying particles. Receiving some morbid
secretions on their limbs, or other parts of their bodies, they have
occasionally been the means of spreading erysipelas in hospitals,
and glanders in veterinary stables. The specific contagia of
cholera, enteric fever, and summer diarrhœa are occasionally
conveyed to food by flies which have previously alighted on latrines
or privies or other places where the stools of such patients have
been deposited (page 281). The excreta of flies, which are not107
uncommonly deposited on food, or on articles of furniture, have
occasionally being found to contain the minute ova of intestinal
worms.
Effects of Suspended Matters.—The inhalation of dust is
followed by deleterious effects. We may divide the solid substances
inhaled as dust into three kinds:—dead substances, living substances,
and the contagia (microbes or germs) of various diseases.
1. Dead Substances inhaled for a prolonged period in various
occupations are a common cause of premature death. The potter
draws into his lungs a fine silicious dust, which irritates his lungs,
and finally produces a fatal disease, known as potter’s asthma.
Mill-stone Cutters and Stone Masons inhale the fine particles
of stone given off from the material which is being chiselled. These
produce serious disease of the lungs.
Pearl Cutters inhale fine particles of pearl-dust, and as they
generally work in close rooms, and the dust is light and tasteless,
serious disease of the lungs results.
Sand-paper Makers inhale minute portions of glass and sand;
and needle and knife grinders are exposed to similar dangers, and
at one time the mortality among them was frightful. It has greatly
diminished since the introduction of wet grinding, the use of steam
fans, and wearing of respirators.
Hemp and Flax Dressers inhale a dust which is peculiarly
irritating. Workers in rags and in wool suffer in like manner
from dust. The dust from fleeces of wool, and especially from the
alpaca fleece, has produced in many cases (in the neighbourhood of
Bradford and elsewhere) an acute disease (anthrax) proving fatal in
a few days. The spores of this disease are very persistent of life
(page 274), and remain active for mischief for months after the
death of the animal which had suffered from it. The fleece can be
disinfected by steam; and the use of fans for diverting the dust
created during “sorting” minimises the danger from it.
The miller commonly suffers from a form of asthma, not so
severe as potter’s asthma, as the particles in this case are not equally
irritating. The hairdresser is liable to inhale the short fragments
of hair cut by the scissors, and the mortality of this class of workers
is high. Miners in coal have a surprisingly low mortality, when
accidents are excluded from the calculation; except in South Wales,
where it is slightly higher than for all males in the same district.
Coal dust is relatively free from sharp angles, and is therefore not
so irritating to the lungs as metallic dust. Consumption is
relatively rare among miners.
The Fur-dyer is very prone to suffer from the dust of the dyed
furs, great irritation and disease resulting in many cases.
Artificial Flower-makers, and those engaged in colouring
arsenical wall-papers, suffer from the inhalation of arsenical vapours,
as well as from the irritating effects of its absorption by the skin.
These are now seldom seen, owing to the almost complete abandonment
of the use of arsenic for wall-pigments.
Cigar-makers are liable to have their lungs irritated by108
inhalation of the dust of the tobacco-leaf; and may suffer from
tobacco-poisoning.
Workers in Lead are very liable to be poisoned by the metal,
e.g., house painters, potters engaged in the glazing process, in which
the ware is dipped into a solution containing lead, manufacturers of
white lead, and others. The lead is partly absorbed by the skin;
in some cases it is inhaled as dust; and more often it is swallowed,
when the workman eats his meals with unwashed hands. Of the
symptoms “painter’s colic” and “drop-wrist” are the two most
important, though, in some cases, lead shews its effects more
insidiously, leading to gout and chronic renal disease. It is now
compulsory on employers to provide in the workshop, complete
washing arrangements for the use of workers in lead. Every
doctor called to attend a case of lead or phosphorus or arsenic
poisoning or anthrax, which has been acquired in an industrial
occupation, must notify the same to H.M. Inspector of Factories.
This implies inspection of the factory or workshop and the subsequent
adoption of further measures of precaution.
Brass-founders occasionally inhale the fumes of oxide of zinc;
and diarrhœa, cramp, waterbrash, and other troubles are the result.
Those engaged in the manufacture of bichromate of potass, are
liable to partial destruction of the mucous membrane of the nose,
and to irritation of the skin, with the formation, in some cases, of
small ulcers.
Workers with Phosphorus, as those engaged in the making
of phosphorus matches, not uncommonly suffer from a gradual
necrosis (death) of the jaw-bone. Those having carious teeth are
especially attacked by this disease, which is due to the fumes of
oxide of phosphorus, attacking the jaw. Improved ventilation of
workshops, careful attention to the teeth, and other measures, have
greatly diminished this disease; and it has disappeared where safety
matches made from red non-volatile phosphorus, have replaced
matches made from the yellow variety.
Chimney Sweeps occasionally suffer from irritative skin
diseases, as well as bronchitis. In some cases the chronic irritation
of the soot has produced cancer of the skin.
The effect of dust on workers can be seen in the mortality returns: Among
men aged 25 to 65 years in 1881-90, the comparative mortality figure in England
and Wales was as follows, all males throughout the country being taken as a
standard and given as 1,000:—
Comparative Mortality Figures.
| All males |
1000 |
| OCCUPATIONS WITH NO DUST. |
DUSTY OCCUPATIONS. |
| Clergyman |
533 |
Coal miner (Derby and Notts.) |
727 |
| Gardener |
553 |
Carpenter |
783 |
| Farmer |
563 |
Bricklayer, mason |
1,001 |
| Teacher |
604 |
Coal miner (Lanc.) |
1,069 |
|
|
Tool and scissors maker |
1,412 |
|
|
Potter |
1,706 |
|
|
File-maker |
1,810 |
Remedial Measures.—Means have been taken to diminish109
the prevalence of the above dust diseases, in several cases with
remarkable success. In the case of steel-grinding, for instance, the
mortality is greatest with dry grinding, and least with wet grinding.
Wet processes have been applied to others of the industries named,
with a like success. Where the dust cannot be avoided, the use of
steam or electric fans, to deflect the dust away from the workman,
has been found successful; and in many cases, free ventilation of
the workshops has greatly diminished the mortality. Where none
of the above measures suffice, the use of respirators ought to be
insisted on. Breathing through the nostrils ought to be carefully
maintained, as thus the dust is to a large extent stopped before
reaching the lungs.
The dangers of lead poisoning may be avoided by absolute
cleanliness, the hands being always washed before taking meals,
and the nail-brush used to secure complete cleanliness beneath the
nails.
2. Living Substances.—The pollen of plants in some persons
produces a distressing form of disease, called hay-asthma, which is
apt to recur each year, and is sometimes only curable by living in
a town or removing to the sea-coast. The amount of pollen
floating about in the atmosphere is considerable; 95 per cent. of it
is grass-pollen, and this form and the pollen from pine-trees appear
to be the most powerful in inducing hay-asthma. According to
some authorities, hay-asthma is rather due to the minute particles
constituting the scent of various flowers, than to the pollen; but
that is probably not the usual mode of origin of the disease,
though it may be in some cases. In some cases, true asthma
results from smelling particular plants. Here as in the case of
hay-asthma a peculiar idiosyncrasy is involved, only a very small
proportion of those exposed to the minute particles suffering from
asthma.
The spores of many fungi and of other living organisms are
constantly being floated about in the air, until they find a suitable
resting place, when they settle and proceed to grow and multiply.
The souring of milk, the fermentation of a saccharine solution, the
moulding of bread, the presence of mildew, the blighting of corn,
and numerous other phenomena are due to the growth of organisms
carried by the atmosphere from one part to another.
3. The Contagia (microbes or germs) of the acute infectious
diseases are minute living organisms, known as bacteria. Hence these
diseases may be carried about by currents of air, some much more
easily than others. Some of the contagia have a persistent
vitality. Thus the contagia of scarlet fever, diphtheria, or small-pox
may infect a room for months, causing the disease in question,
when infected articles in the room are disturbed. The contagia
of typhus fever and of measles, on the other hand, are short-lived,
and do not usually resist free ventilation and exposure to sunlight.
Besides the contagia of the acute fevers, septic organisms may
be carried by the atmosphere. Formerly, blood-poisoning from
operation and other wounds was common; but Lister, by insisting
on absolute cleanliness of wounds, and only allowing air to have110
access to the wound which had been filtered through layers of
gauze and deprived of its septic germs, has secured that wounds
can now be kept perfectly “sweet,” the suppuration in them
reduced to a minimum, and the danger of blood-poisoning almost
annihilated (page 106). It had often been noticed that recovery
from even very severe injuries was common, if only the skin
remained unbroken; while the same injuries, with the addition of
a rupture of the skin, and consequent access of air, were rapidly
fatal. But to Lister is due the great honour of proving that it was
not the air which produced the mischief, but the germs it contained,
and that filtered air might be admitted with impunity.
Erysipelas and hospital gangrene have occasionally been carried
about in hospital wards by dirty sponges and dressings; and if the
ventilation is not perfect, particles of epithelium and pus from
diseased persons may be carried to other patients at a distance.
Some forms of purulent disease of the eyes are transferable from
patient to patient, and in children some forms of eczema are also
contagious.
111
CHAPTER XVI.
GASEOUS AND OTHER IMPURITIES OF AIR.
Gaseous impurities of the air are very commonly associated
with suspended matters, and it is often difficult to separate the
effects of the two.
Different gases are also often associated, and so produce
modified results. It will be convenient to consider, first of all,
certain well-marked gaseous impurities, and then others in which
there is a mixture of several gases, or of these with suspended
solid particles.
Under the first head the most important impurity is—
(1) Carbonic Acid.—This is reckoned an impurity if
amounting to more than 5 parts in 10,000 of air. Owing to the
large amount produced in the respiration of animals, in the
combustion of fires, gas, lamps, etc., and in other natural
processes, it would be much greater in populous parts, were it
not for the rapid diffusion occurring in the air, and the purifying
action of plants. The following analyses (Angus Smith) illustrate
the facts that in towns the amount rises, and is greatest in the
most populous parts, while during fogs it is greatly increased.
| VOLUMES OF CARBONIC ACID IN 10,000 VOLUMES OF AIR. |
| On the mountains and moors of Scotland—mean of 57 analyses |
3·36 |
| In the streets of Glasgow—mean of 42 analyses |
5·02 |
| London, N., N.E., and N.W. postal districts—mean of 30 analyses |
4·384 |
| London, E. and E.C.—mean of 12 analyses |
4·745 |
| Manchester streets, ordinary weather |
4·03 |
| During fogs in Manchester |
6·79 |
The effects of carbonic acid gas alone must be carefully
distinguished from those of the same gas plus the organic impurities
from respiration, with which it is commonly associated. Dr.
Angus Smith found that air containing 3 per cent. of carbonic
acid produced difficulty of breathing, but he was able to breathe
comfortably an atmosphere containing 0·2 per cent. carbonic acid.
Other observers have found they could breathe without discomfort
air containing 1 per cent. carbonic acid. When the carbonic acid
is derived from respiration, headache and giddiness are produced
in many persons when the carbonic acid amounts to 0·15 per cent.112
A fatal result has occasionally occurred from the inhalation of the
carbonic acid at the bottom of brewing vats, or about lime-kilns.
The gaseous impurity at the bottom of wells is more commonly
nitrogen than carbonic acid (page 102).
The presence of an excess of carbonic acid diminishes the
elimination of carbonic acid from the lungs, nutrition and muscular
energy being consequently impaired. This is seen in workshops
where the air is confined and gaslight is commonly employed;
though the air here also contains carbonic oxide, sulphurous acid,
and organic impurities, and these probably have a large share in
producing the evil results.
(2) Carbonic Oxide in the proportion of more than 1 per
cent. is rapidly fatal, and has poisoned when under ½ per cent.
Poisoning by its means occurs where charcoal stoves are used, and
especially when the charcoal is burnt in rooms with no chimney
flue. This is an occasional mode of suicide on the continent.
Carbonic oxide is a much more deadly poison than the dioxide
(carbonic acid); it forms a stable compound with the hæmoglobin
of the red blood-corpuscles, displacing oxygen from them, and is
got rid of with great difficulty. Lace-frame makers place a coke
stove under their work, and thus inhale the invisible gas. Headache,
giddiness, irregular action of the heart, and depression of
the general health result. Carbonic oxide is the most poisonous
constituent of coal-gas, and is present in much larger quantity in
carburetted water-gas with which coal-gas is now commonly mixed,
than in pure coal-gas (page 115).
(3) The inhalation of Sulphuretted Hydrogen produces
headache, nausea, and diarrhœa; but in manufactures involving
the inhalation of a small proportion of this gas the symptoms are
much slighter.
(4) Sulphurous Acid is always present in small quantities
in the air of towns, derived from the combustion of coal and
coal-gas. Straw-bleachers and the bleachers in cotton and worsted
manufactories, often suffer from severe cough and bronchitis due
to inhaling its irritating vapours.
(5) Carbon Disulphide when vaporised and inhaled produces
headache, general muscular pains, and nervous depression. It is
used in the manufacture of waterproof coats, toy balloons, etc.
(6) Ammonia produces irritation of the eyes and bronchial
irritation. Hat-makers commonly suffer from its effects, being
generally pale and feeble. It is difficult to say how much is due
to the ammonia, and how much to the high temperature at which
they work.
(7) Acid Fumes are very irritating to the lungs, and in the
case of alkali manufactures, they destroy all vegetation for
considerable distances. Hydrochloric acid produces great irritation,
and chlorine even more so. The fur-dyer is not only subject to
the dangers of dust, but also of the fumes of nitric acid, used to
remove fat and give certain shades of colour to the fur.
(8) Other Vapours evolved in various processes produce113
special symptoms. House-painters suffer from the inhalation of
turpentine vapour, headache and loss of appetite commonly resulting.
The symptoms from the commonly coexistent lead-poisoning are
distinct. Brush-makers have a persistent cough, due to the
inhalation of resinous fumes, evolved in making brushes.
Workers in paraffin are liable to an irritative disease of the
hair-follicles of the body, followed by the formation of scars,
almost like small-pox marks.
Workers in quicksilver, as those engaged in making mirrors or
thermometers, are prone to suffer from mercurial poisoning. The
gums become spongy, and there is profuse salivation, also generally
alimentary disturbance; and in some cases nervous affections,
resulting in persistent muscular tremblings, etc.
Under the second head—cases of inhalation of mixed gaseous
and particulate contamination—we must consider
(1) The Effects of Air Rendered Impure by Respiration.—It
has been already stated that an amount of carbonic acid which
could easily be borne alone, is intolerable when other products of
respiration are mixed with it. These are chiefly organic gases and
solids, which (unless removed quickly) render the atmosphere close
and “stuffy”—an effect which is readily perceptible by the sense
of smell of those entering an occupied room immediately from
the outer air. When such a room is inhabited for a few hours,
headache, langour, drowsiness, and yawning (which is really a cry
for purer air) result. The soporific effects so commonly produced
in churches, etc., are commonly due to the vitiated atmosphere,
rather than as is supposed to the soothing effects of the sermon.
When the exposure to foul air is more chronic, and occurs day
after day, there results a general lowering of strength and vigour—both
bodily and mental—even where no actual disease is set up.
Oxidation processes are retarded; the consequence is an anæmic
sallow complexion, which compares badly with the ruddy
complexion of those spending a great part of the day out of doors.
The prolonged breathing of air, foul from the products of
respiration, is perhaps more common in workshops and schools than
in private houses; but in both, a faint smell is commonly perceptible
on entering from the open air, indicating imperfect ventilation and
accumulation of organic putrescible matter. The preceding remarks
are left as in the last edition. It must be noted, however, that
recent research attaches more importance to the particulate matter
(dust) in the atmosphere than to the amount of gaseous impurity,
though the latter remains a convenient index of impurity.
Experiments made by Drs. Haldane and L. Smith on themselves
negative the older conclusion that a special organic poison exists in
expired air. They were able without any appreciable effect on
themselves to breathe air which was vitiated to such an extent as to
completely prevent a match from burning; and they conclude that
excess of carbonic acid and deficiency of oxygen are the sole cause
of danger from breathing air highly vitiated by respiration. This114
conclusion may be accepted under the conditions of these
experiments. Under ordinary conditions, however, the evil effects
produced by breathing the air of crowded rooms, are due not only
to the excess of carbonic acid and deficiency of oxygen, but also to
the dust which is usually associated with them. This dust, which
may be derived from handkerchiefs of patients suffering from
influenza, consumption, sore throat, &c., or from other sources, is
apt to be inhaled by the persons occupying such rooms.
The tendency to catarrhs is greatly increased by living in a
vitiated atmosphere. In the causation of “colds” two elements are
concerned, the infective agent, and the condition of the patients.
“Colds” are caused primarily by infection from previous patients.
The nasal discharge of a “cold in the head” contains the contagium.
This is dried on handkerchiefs, and is subsequently scattered as
dust, and thus conveyed to others. Ordinarily there is considerable
resisting power against such catarrhs. When, however, the general
vitality or the local vitality of the mucous membrane of the nose,
throat, and lungs is impaired by the breathing of impure air or by
sitting in wet clothes after exposure to wet and cold, a catarrh is
produced.
The close connection of phthisis (consumption) with overcrowding
and the breathing of a vitiated atmosphere will be
discussed hereafter (page 313). The polluted air acts in producing
consumption by depressing vital functions, and diminishing the
powers of resistance against the actual contagium of the disease,
which is inhaled as dust, produced by the drying of the expectoration
of consumptive patients.
The germs of infectious diseases are propagated very rapidly in
an impure atmosphere; and typhus fever occurs almost solely in
conditions of overcrowding.
In the cattle-plague of 1866, it was found that nearly all the
cows died when crowded together in unventilated sheds, while only
a third died when there was free ventilation.
The effects of air containing the products of respiration in a
concentrated condition, and of a deficient supply of air, have been
shown only too well in the oft-quoted case of the Black Hole
of Calcutta. In this case, 146 persons were confined in a space
eighteen feet every way, with two small windows on one side.
Next morning 123 were found dead, and the remaining 23 were
very ill.
In the experience of this country, the highest death-rates are in
the most densely populated districts. The death-rate from phthisis,
childbirth, and typhus fever for instance, is far higher in cities than
in country-places. The fact may be explained in various ways.
Density of population commonly implies insufficient or unwholesome
food, unhealthy work, and poverty; but especially impurity of the
air, uncleanliness, and imperfect removal of excreta. Of these
factors, the vitiated air is probably the most powerful for evil.
Children suffer more than adults from close aggregation of115
population, largely owing to the greater ease with which infectious
diseases spread in towns.
(2) Coal-gas is obtained by the destructive distillation of coal,
free from access of air. The average composition of London coal-gas
is hydrogen 50 to 53, saturated hydrocarbons 33 to 66,
unsaturated hydrocarbons 3·5 to 3·6, carbonic oxide 5·7 to 7·1,
carbonic acid 0 to 0·6, nitrogen 2·5 to 4·1, and oxygen 0·2 to 0·3 per
cent. Of these the illuminants are olefiant gas (C₂H₄) and the
higher hydrocarbons. Sulphuretted hydrogen and other sulphur
compounds are present in small quantities, averaging 12 grains of
sulphur per 100 cubic feet of London gas.
The inhalation of coal-gas, even in small quantities, is liable to
produce headache, and may lead to chronic poisoning if allowed to
continue. Where the escape of gas is more extensive, as when a
tap is left turned on accidentally during the night, two dangers may
arise. If a light is struck in the room an explosion occurs; or
persons may be poisoned in their sleep by inhalation of the gas.
The most poisonous gas in coal-gas is the carbonic oxide. The
chief product of the combustion of coal-gas is carbonic acid. Some
sulphurous acid is also produced, which is irritating to breathe,
and injurious to bookbindings, picture-frames, etc. If the flame is
imperfect, as when the pressure of gas is too great, some carbonic
oxide may also escape.
In recent years Carburetted water-gas has been largely
mixed with coal-gas in certain districts. This is made by passing
steam over heated coke. Thus
| C |
+ |
H₂O |
= |
CO |
+ |
H₂ |
| (coke) |
|
(steam) |
|
(carbonic oxide) |
|
(hydrogen) |
The product is water-gas which burns with a non-luminous flame
and has no smell. For illuminating purposes it is enriched with
vaporised paraffin oil, which gives it a high illuminating power, and
a smell rather like that of coal-gas. In some towns as much as 60
per cent. of this carburetted water-gas is mixed with 40 per cent. of
coal-gas. Now as the former contains about 30 per cent. of
carbonic oxide, and the latter only 7 per cent., a mixture of equal
parts of the two gases would contain 18·5 per cent. of carbonic
oxide, and would therefore be much more dangerous than coal-gas.
This has been found to be so in actual experience of escapes of
gas.
In speaking of these products of different illuminants, it is
necessary to adopt a standard of light. In this country the
standard has hitherto been a light known as “one-candle power”
which is given by a sperm candle burning 120 grains per hour, or
in V. Harcourt’s standard flame by a mixture of air and pentane
(C₅H12). A good fish-tail or bat’s wing burner for coal-gas gives an
illuminating power equal to 16 candles, and burns from 4 to 5 cubic
feet of gas per hour. Most flat flame burners known as 4 or 5, and
supposed to burn that number of cubic feet of gas per hour, really
consume nearly double this amount of gas. In the following table
the amount of various products produced and of vitiation of air116
caused by various forms of illuminants is compared, when an
illumination equal to 16 candles is produced in each instance:—
|
AMOUNT BURNT. |
CARBONIC ACID PRODUCED. |
MOISTURE PRODUCED. |
OXYGEN REMOVED. |
AMOUNT OF VITIATION PRODUCED STATED IN TERMS OF THE
NUMBER OF ADULTS WHO WOULD CAUSE AN EQUAL VITIATION. |
| Sperm candles |
1740 grains |
6·6 c.ft. |
6·6 c.ft. |
9·6 c.ft. |
11 |
| Paraffin oil |
992 „ |
4·5 „ |
3·5 „ |
6·2 „ |
7 |
| Coal gas burned in Argand burner |
4·8 c.ft. |
2·5 „ |
6·4 „ |
5·8 „ |
4 |
| Flat-flame burner |
5·5 „ |
3·5 „ |
7·4 „ |
6·5 „ |
6 |
Thus as an adult expires 0·6 cubic feet of carbonic acid per
hour, it follows that the amount of carbonic acid produced in one
hour by the various illuminants named in the above table, burning
so as to give a light equal to 16 standard candles, varies from 4 to
11 times the amount produced by the adult. Candle and oils possess
the advantage over coal-gas that no sulphurous acid is produced in
combustion. If the pressure in the mains is excessive, some gas may
escape through the burner unburnt or carbonic oxide may escape.
In England the flashing-point of mineral oils has been fixed at
73° Fahr. The material of which the reservoirs of lamps are composed
should not be glass or other breakable material, and the wick
should be contained in a small wick chamber extending nearly
to the bottom of the reservoir. Only a tight fitting wick must
be used.
The best illuminant for domestic purposes is incandescent
electricity, in which no products of combustion are formed, and
only a comparative small amount of heat is produced. Electrical
illumination possesses the further advantages that there is no
blackening of ceilings and no damaging of other decorations as in
illumination by gas.
(3) Air Rendered Impure by Exhalations from the Sick.
In addition to the ordinary impurities of occupied rooms, special
impurities are produced, varying with the character of the disease.
They may include infectious particles from the sick. In wards for
consumptives and for diphtheria, dust in the room has been found
to contain the special microbes of these diseases. Making beds,
sweeping floors, &c. may help to scatter infectious dust; hence the
importance of adopting means of cleansing which will not scatter
dust, and of keeping sick-rooms spotlessly clean. In many diseases
e.g. consumption, a patient may re-infect himself with such
infectious dust, and thus diminish his own chance of recovery
(see page 311). Hospital wards can scarcely be too freely ventilated;
but even more important than ventilation is the strictest cleanliness
in every minute detail.
(4) The Air of Sewers, Cesspools, etc., may contain the117
products of decomposition of sewage, such as volatile fœtid organic
matter, carbo-ammoniacal substances, sulphuretted hydrogen,
carbonic acid, etc. The amount of these various products varies
greatly under different circumstances, such as dilution of the
sewage, ventilation of sewers, temperature, etc. The effluvia from
cesspools are usually more concentrated than those from sewers. It
appears fairly certain that the emanations from sewers or drains
may give rise to diarrhœa and gastric disturbances, and to certain
forms of sore throat, which favour the production of diphtheria.
On the other hand, there is much evidence showing that the danger
from sewer-emanations has been exaggerated. Carnelley and
Haldane found that the air of the sewers of the Houses of
Parliament and of certain sewers of Dundee was not very impure,
containing a smaller number of bacteria than external air. There
is reason to believe that the emanations from well-ventilated sewers,
possessing a good gradient, so that the contents of the sewers are
hurried away to the outfall, are free from danger. The chief source
of possible danger would be the escape of the bacteria of such
diseases as enteric fever or diphtheria, which had been discharged
into the sewer from patients suffering from these diseases. But, in
the absence of splashing, these bacteria could not escape from a
liquid medium. Their escape could only occur when the sewer
became dry, and the dust was carried up by rapid currents of air, a
very improbable occurrence in sewers. Hence in the majority of
instances sewer emanations must be freed from the accusation of
producing infectious diseases. Sewer-men usually enjoy good
health, and there is no excess of infectious diseases among them.
The emanations from obstructed drains or sewers may cause
serious mischief, similarly to that occasionally produced by the
emanations from cesspools. Under such conditions, sulphuretted
hydrogen, carburetted hydrogen, and other gases are evolved, and
fatal asphyxia has been caused by these. In other instances acute
sewer-gas poisoning, without pneumonia, has followed.
The exhalations from cesspools or privies while cleaning them
out, may produce severe disorders, which are sometimes fatal.
When a drain is newly opened or sewer gas gets into a house,
a less marked form of poisoning sometimes occurs, chiefly
characterised by languor, headache, vomiting, and diarrhœa. In
some cases there may be febrile attacks lasting a few days. Children
are especially sensitive to such conditions and quickly fall into ill
health.
The direct origin of acute infectious diseases from the effluvia
from drains or cesspools has occasionally occurred. Leaky and
choked drains under a house are especially dangerous. The subsoil
becomes contaminated more and more as time goes on; foul
gases are aspirated into the house, owing to its interior being
warmer than the subsoil; and finally infectious matter may find
its way into the house, or carried by insects or vermin, through
cracks in the earth.
Diphtheria has been ascribed to emanations from drains and
sewers. There is reason to believe that a non-specific form of sore118
throat may originate in this way; but diphtheria is generally, if
not always, spread by personal infection. Diarrhœa has been
occasionally ascribed to sewer-emanations. It chiefly occurs in hot
weather, and is usually associated with a foul condition of the
surface soil, and speedily ceases after this has been scoured by
copious rain.
Enteric or typhoid fever, has been frequently ascribed to drain
and sewer effluvia. It was formerly thought that putrefactive
changes alone, under certain conditions of temperature, etc., would
produce it, and Dr. Murchison, one of the greatest authorities on
the subject, who adopted this view, proposed for enteric fever the
name “pythogenic fever” (i.e. filth-produced). Isolated cases of
enteric fever, occurring where there is no system of drainage,
support the same view, as does also the fact that, with the adoption
of drainage, the enteric mortality has steadily diminished. On the
other hand, numerous cases can be quoted to show that emanations
from excreta have been breathed, and sewage-contaminated water
drunk, for years, without the production of a single case of enteric
fever—until a case is accidentally imported. The weight of
evidence is clearly on the side of the view that only emanations from
the liquid or solid dejecta of previous enteric patients will produce
enteric fever, and that it is the solid particles of the urine or fæces,
either inhaled as dust or carried on to food by flies, &c., or mixed
with food by contaminated water, &c., which cause infection.
Furthermore, modern investigation shows that infection by dust
is the exception in England; and that the enteric infection is
usually swallowed and not inhaled, being taken in infected water or
milk or other food.
(5) Effluvia from Decomposing Organic Matter.—(a)
The air of marshes contains an excess of carbonic acid, marsh
gas, etc., in addition to other organic matters. Malarial diseases
are commonly ascribed to the inhalation of the marsh effluvia under
certain conditions, though the recent proof of the part played by
the mosquito in spreading malaria, puts the inhalation of such
effluvia in the background as cause of this disease (page 307). Some
forms of diarrhœa and dysentery have been ascribed, with a less
degree of probability, to the same cause. In this case, as in that of
emanations from other organic sources, the impurities received by
the air are both gaseous and particulate.
(b) The Air of Graveyards contains an excess of carbonic
acid. The older intramural graveyards appear to have been a
cause of illness; but modern graveyards, kept under good regulations
have never been shown to cause illness.
(c) The Effluvia from Decomposing Carcases, especially
of horses on the battle-field, have led to outbreaks of diarrhœa
and dysentery among the soldiers.
(d) The Effluvia from Manure and Similar Manufactories
do not seem to injure the workmen as a rule, but attacks of diarrhœa
have been produced in the neighbourhood when the wind has
wafted the effluvia towards any particular part. Sore throat, and119
occasionally diphtheria, have been ascribed to the inhalation of
London manure taken into Essex.
(6) The Effluvia from Certain Manufacturing Processes
seem to be rather nuisances than actually productive of ill health.
The vapours given off by tallow-making and bone-burning processes
are most disagreeable, but there is little or no positive evidence of
their direct insalubrity.
The air of brickfields and cement works is peculiarly disagreeable.
The Degree of Moisture and the Temperature of air are
of great importance in relation to health. Air which is unduly
moist or dry, hot or cold, may be injurious apart from any foreign
matters it contains.
The relative amount of moisture is of greater importance than its
actual amount. An atmosphere which contains aqueous vapour up
to the point of saturation is very oppressive; the normal evaporation
of insensible perspiration (and with it of the organic impurities
removed from the skin) is interfered with; and consequently the
“oppressiveness of the day” is complained of.
An unduly hot air is generally productive of pallor and ill
health, though it is difficult to know how much to ascribe to the
high temperature, and how much to the commonly coexistent
vitiated atmosphere. The temperature of living-rooms ought not
to be over 60° to 65° Fahr., and of bedrooms not over 60° Fahr.
The devitalising influence of extreme cold is well known. Its
effects are more particularly seen in young children and the very
old, who require to be carefully tended during severe and long-continued
cold weather. Dry, cold weather, with the temperature
near the freezing point of water, and a cutting east wind
prevailing, is not uncommonly described as “bracing.” This is so
far from being the case, that it requires all the vital powers of the
strong and healthy to resist its depressing influence, and the feeble
of both extremes of age succumb.
120
Many occupations are the source of considerable danger to the workers
engaged in them. They are chiefly injurious by the inhalation into the lungs
of some foreign agent, which produces serious local inconveniences and irritation,
and may be also absorbed into the circulation and produce more remote effects.
The injurious agents may be classified under four heads:—
(1) Insoluble particles or dust.
(2) Soluble or partially soluble substances.
(3) Injurious gases or vapours.
(4) Effluvia from offensive trades.
It is evident that, as regards the effluvia named under (4), they might
generally be included under the three previous heads, though it is convenient
for our present purpose to keep them separate.
The occupations in which dust and soluble substances are productive of
injurious effect have already been described, pages 107 to 109.
Injurious gases and vapours have received consideration on pages 111 and
112. The special offensive trades still require attention.
Offensive Trades.—The legal enactments relating to offensive trades are
contained in sect. 112 of the Public Health Act, 1875, which states, any person
who, after the passing of this Act, establishes within the district of an urban
sanitary authority, without their consent in writing, any offensive trade, that
is to say, the trade of—
- Blood boiler, or
- Bone boiler, or
- Fellmonger, or
- Soap boiler, or
- Tallow melter, or
- Tripe boiler, or
- any other noxious or offensive trade, business, or manufacture,
shall be liable to a penalty not exceeding fifty pounds in respect of the establishment
thereof, and a penalty not exceeding forty shillings for every day on which
the offence is continued.
These provisions can only be enforced in rural districts with the sanction
of the Local Government Board.
The “other noxious or offensive trades,” in order to be brought within the
operation of the section, must be analogous to those which are specially
enumerated.
The most exhaustive and authoritative report on this subject is by the
late Dr. Ballard, whose report is largely quoted in the following remarks.
We may consider (1) the extent to which the public is inconvenienced by various
effluvium nuisances. The majority of the nuisances arise from trade processes
in which animal matters are chiefly used. Among the most disgusting are
the effluvia from gut-scraping, and the preparation of sausage skins and catgut,
the preparation of artificial manures from “skutch” (the refuse matter of the
manufacture of glue), the manufacture of some kinds of artificial manures, and
the melting of some kinds of fat. Manufacturing businesses dealing with
vegetable substances are often offensive, but rarely give out disgusting effluvia.
The most offensive vegetable effluvia are probably those thrown off during the
heating of vegetable oils, as in the boiling of linseed oil, the manufacture of
palmitic acid from cotton oil or palm oil, the manufacture of some kinds of121
varnish, the drying of fabrics coated with such varnishes, and the burning of
painted articles, such as disused meat-tins.
Occasionally offensive effluvia arise in connection with industries in which
neither vegetable nor animal matters are used; as in the manufacture of sulphate
or chloride of ammonia, and some other processes in which sulphuretted
hydrogen is copiously evolved; and in the making of gas and the distillation
of tar. The fumes from the manufacture of alkali and bleaching powder are
acid and irritating, and produce very injurious effects on vegetation in the
neighbourhood.
The distances to which nuisances extend vary greatly according to circumstances—as,
for instance, the elevation at which the effluvia are discharged into
the air. Discharge from a high chimney may relieve the immediate vicinity
of the works at the partial expense of those living at a greater distance. With
a damp and comparatively stagnant atmosphere, effluvia have a much greater
tendency to cling about a neighbourhood.
(2) The industrial processes in which offensive effluvia are produced are
classified by Dr. Ballard as follows:—
1. The keeping of animals.
2. The slaughtering of animals.
3. Other branches of industry in which animal matters or substances
of animal origin are chiefly dealt with.
4. Branches of industry in which vegetable matters are chiefly dealt
with.
5. Branches of industry in which mineral matters are chiefly dealt
with.
6. Branches of industry in which matters of mixed origin (animal,
vegetable, and mineral) are dealt with.
(3) It is important to inquire to what extent offensive trade effluvia are injurious
to the public health. It is impossible to bring statistics to bear on the inquiry, as
other influences, apart from occupation, can scarcely be eliminated. The term
“injurious to health” is capable of a double interpretation. It might mean
either serious damage to health, or the mere production of bodily discomfort
or other functional disturbance by the offensive effluvia, leading by its continuance
to an appreciable impairment of vigour, though not to any actual
disease.
In the latter sense offensive effluvia have a deleterious effect on health.
Such symptoms as loss of appetite, nausea, headache, occasionally diarrhœa,
and general malaise are produced by effluvia of various kinds, but agreeing in
being all offensive. “A condition of dis-ease or mal-aise is produced.”
There is little difficulty in proving bad effects on the workmen, though
the invariable defence of manufacturers is an appeal to the condition of health
of their workmen. The workmen only remain such so long as they are healthy,
and as they become disabled they necessarily cease to rank among workmen.
The decomposition of putrefying organic matters is unquestionably dangerous.
The general doctrine of sanitation that filth is one of the chief factors in
producing disease is certainly applicable to trade effluvia as well as to general
sanitation. It has been alleged on behalf of such effluvia as chlorine sulphurous
acid and tar vapours that they are useful disinfectants; but modern research
has shown that disinfectants, in order to be of practical use, must be in such
a concentrated condition that the air containing them is irrespirable. Probably
such septic diseases as erysipelas are favoured by organic trade effluvia.
(4) The means available to prevent or minimise the nuisances arising from
trade effluvia vary with the character of the processes. The general principles
on which treatment must be founded depend, as Dr. Ballard points out, on a
recognition of the following kinds of effluvia:—
Effluvia dependent—
1. On the accumulation of filth on or about business premises, or on
its removal in an offensive condition.
2. On a generally filthy condition of the interior of buildings and
premises and utensils generally.
122
3. On an improper mode of disposal of offensive refuse, liquid or
otherwise.
4. On insufficient or careless arrangements in reception of offensive
materials of the trade, or in removal of offensive products.
5. On an improper mode of storing offensive materials or products.
6. On the escape of offensive gases or vapours given off during some
part of the trade processes.
It is evident that under the first two headings the proper remedy is
cleanliness. Filth should be removed in impervious covered vessels, at regular
intervals. Structural arrangements should be made, which will facilitate
cleansing operations. Solid refuse should, as far as possible, be separated
from liquid refuse, as thus putrefaction is retarded.
Under the last head important remedies are applicable. In many cases a
careful selection of the materials of manufacture will form an effective remedy.
Thus much of the nuisance connected with soap or candle works arises from
the putrid condition of the fat collected from butchers and marine store dealers,
and might be obviated by more regular and more frequent collection of the
materials of manufacture. The offensive vapours arising during processes of
manufacture may be intercepted before reaching the external air, and so
treated that they lose their obnoxious character. Various methods of interception
are adopted, according to the processes involved. Occasionally it is
necessary to have the air of the entire workshop drawn by means of artificial
ventilation in a special direction; usually the interception of air from special
chambers suffices. When thus collected, the offensive air may be dealt with
by (1) passing it through water or some other liquid capable of absorbing the
offensive materials; or (2) passing it through some powder with which it has
chemical affinity; or (3) if its offensive materials are capable of condensation
by cold, passing them through an appropriate condensing apparatus; or (4) if
the evolved matters are organic in nature, conducting them through a fire.
(5) Occasionally it is sufficient to discharge the offensive gases into the air from
a high chimney; and this always produces a mitigation of nuisance, as compared
with discharge at a low level.
It is usually found that the adoption of one or other of these methods is
directly or indirectly profitable to the offender.
Nuisances from the Keeping of Animals.—The 47th section of the
Public Heath Act prohibits the keeping of pigs in towns so as to be a nuisance,
and, as a general rule, it is possible to obtain a magistrate’s order, entirely
prohibiting the keeping of pigs in towns. The excreta of the pig have a very
offensive and penetrating odour, and however carefully kept, pigs in towns form
an intolerable nuisance.
Not only is there nuisance from the accumulation of manure and dirtiness
of the piggeries, but also from the storage and subsequent preparation of
food. The boiling of hog-wash is often an even greater nuisance than the
filth of the styes.
Cow-keeping and horse-keeping in towns are still allowed and, as compared
with pig-keeping, form a small nuisance. Mews, if kept clean and well drained,
need not be offensive, though it is objectionable for persons to sleep over stables.
The removal of manure also constitutes a difficulty. The manure should not be
allowed to accumulate in deep wet pits, but in an iron cage-work over a cement
paving at or above the ground-level, thus allowing free drainage, and keeping the
manure dry, and reducing ammoniacal decomposition to a minimum.
Cowsheds are generally very badly ventilated, as the cowkeeper finds that
more milk is produced by the cows when the temperature of the shed is maintained
at 65° or higher; and he does not see the necessity for providing artificial
means of warmth. The grains which are used so largely for food are stored in a
wet condition, and speedily give rise to nuisance. Cowsheds and stables should
be well paved and well drained. At least 800 cubic feet should be allowed for
each cow in the shed.
Cowsheds are regulated under the Dairies’, Cowsheds’, and Milkshops’ Order
of the Local Government Board. This order provides for and insists on the
registration of cowkeepers, dairymen, and purveyors of milk, by the local123
authority. It also provides that no cowshed or dairy shall be occupied as
such, unless provision is made to the satisfaction of the local authority, for
the lighting and ventilation, including air-space, and the cleansing, drainage,
and water-supply of the same; and for the protection of the milk against
infection or contamination. With the view of preventing contamination of
milk, no person suffering from an infectious disorder, or having recently been
in contact with a person so suffering, is allowed to milk cows or take any part
in any stage of the business of a milk-seller. The milk of a cow suffering from
cattle plague, pleuro-pneumonia, or foot and mouth disease must not be mixed
with other milk, must not be sold or used for human food, nor for food for swine
or other animals, unless it has been boiled. By the order of 1899 this regulation
is made to extend to tubercular disease of the udder.
Slaughtering of Animals.—Nuisance may arise in slaughter-houses from
various causes:—(1) the uncleanly way in which animals are kept in the pound
or lair before being killed; (2) the insanitary condition, bad paving, lack of lime-whiting
of walls, etc., of the slaughter-house; (3) the accumulation of hides,
blood, fat, offal, dung, or garbage on the premises; (4) the uncleanly condition
of the blood-pits, or the receptacles for garbage; (5) the flowing of blood or offal
into the drains and thence into the public sewer.
Private slaughter-houses ought to be abolished, and all animals intended for
human food slaughtered in public abattoirs under efficient supervision. When
a large number of private slaughter-houses exist in different parts of a large
town, it is impossible for the sanitary officials to properly supervise the
slaughtering, or to ensure that diseased meat shall not enter the market. The
inspector may only have the opportunity of examining the flesh, the internal
organs which more particularly show the presence of a diseased condition
having been concealed. Such concealment and the consequent foisting of
diseased meat upon the public, can only be efficiently prevented by forbidding
the slaughtering of any animal intended for food in a private slaughter-house.
Most local authorities have bye-laws regulating the slaughtering of animals.
These provide for a cleanly condition of the lairs, and prevent keeping the
animals longer in the lairs than is necessary for the purpose of preparation
for slaughtering. They also insist on the provision of proper covered
receptacles of iron or other non-absorbent material for the reception of
garbage, and similar receptacles for blood; for cleansing of the floor, etc.
after slaughtering; for lime-whiting of the walls four times a year; and for
other matters of detail.
For knackers’ yards similar regulations are applicable. The flesh should not
be kept until it becomes putrid before being boiled, and the boiling of the flesh
and fat should be so arranged as to avoid the escape of offensive vapours into
the external air.
In smoking bacon, the singeing has formed a serious nuisance. Fish-frying in
small shops is often a most troublesome nuisance. A hopper over the pan in
which the frying is conducted has not been always successful in carrying the
fumes up the chimney. The frying should preferably be done in a closed outhouse,
close to a chimney with a good up-draught.
The fellmonger prepares skins for the leather-dresser, the chief operations
being taking off the wool, liming the skins, etc. The skins deprived of wool are
called “pelts.” The pelts are thrown into a pit containing milk of lime, and
thence sent direct to the leather-dresser. Nuisance may arise from (1) the odour
of the raw skins; (2) the ammoniacal odour from the lime-painted skins hanging
in the yard; (3) the emptying and cleansing of the “poke” or tank in which
the hides are washed; (4) the foul condition of the waste lime taken from the
exhausted lime pits; (5) the odour from the dirty unpaved yards.
The leather-dresser only deals with “pelts,” derived from sheep-skins; the
tanner with bullocks’-hides. The skins brought from the fellmonger to the
leather-dresser are first deprived of lime, and then soaked in a solution of dog’s
dung, called “pure,” until they become soft. In winter this “pure” solution
is warmed for use. The odour is very abominable, both from the “pure” tub,
and from the discharge of the exhausted “pure” liquid into the drain.
At each stage of tanning nuisance may arise unless great precautions are
taken, as when the hides are soaked in lime and water, when the hair is being
removed, when the loose inner skin of the hide is being removed, and especially124
when the hides are soaked in pits containing pigeons’ or other dung. Nuisance
may arise again during the passage of offensive hides through the street.
Cleanliness is the great rule. If every process is carried on with due precaution,
including frequent washing out of receptacles and the free use of disinfectants,
little complaint need arise.
The manufacturers of glue and size boil out the gelatine from bits of hides
and “fleshings” from leather dressers and tanners, from damaged “pelts,” ox
or calves’ feet, horns, and other similar substances. The raw material is apt
to be offensive in collection or while accumulating on the premises. The process
of boiling causes offence by the effluvia from the steam. The residue remaining
after the process is known as “scutch,” and this, unless frequently removed, is
a most serious source of nuisance.
Prussiate of Potass is manufactured by heating carbonate of potass with
refuse animal matters. In order to avoid nuisance the pot in which the boiling
is done should have a pipe to conduct away the steam, first running horizontally
and then vertically down to the back part of the fire.
Fat-melting and Dip-candle-making, as usually carried on, give rise to
nuisance. The fat which is melted down usually comes from butchers and
marine-store dealers in a rancid or even putrid condition, and it may be stored
on the premises for some time before it is boiled. The vapours from the melting-vat
are very offensive. They should be carried by means of a pipe down until
they discharge just under the boiler-fire. The residue from the fat-melting
process (known as “greaves”) requires frequent removal to avoid nuisance.
Bone-boiling, in order to extract the fat and gelatine, is most offensive, and
most difficult to deal with. After boiling, the bones are apt to give off offensive
smells. The vapours from the closed boiler should be condensed as far as
possible in a worm condenser, and the remainder passed through a furnace
fire.
In the manufacture of artificial manures nuisance is apt to arise (1) from
the reception and accumulation of the raw materials, as putrid fish, putrid
blood, scutch (the residue from the manufacture of glue), recently boiled bones,
etc.; (2) from the preparation of the raw material for use. Thus the drying
of condemned fish or meat on open kilns is very offensive; similarly the drying
of sewage sludge. (3) From the process of mixing the materials of manufacture,
irritant and offensive vapours being evolved, as for instance in the manufacture
of manure by crushing bones, and converting into super-phosphate by the
addition of sulphuric acid. (4) From the removal of the manure from the hot
den, after it has been dried. When sulphuric acid is mixed with coprolites
or other mineral phosphates, most irritant and offensive vapours are produced,
which may be perceived in some cases at the distance of a mile.
Blood-boiling is now almost obsolete, having been replaced by albumen-making
and clot-drying. Nuisance may arise from the blood collected from
slaughter-houses being in a putrid state; and from the effluvia evolved during
the drying process.
Gutscraping, gut-spinning, and the preparation of sausage-skins are very closely
akin. In gut-scraping the putrid intestines are deprived of their interior soft
parts by scraping with pieces of wood, and are then, after being cleansed,
ready for sausage-skins. In gut-spinning the prepared gut is twisted into a
cord. The small intestines of hogs and sheep are used for this purpose. The
stench from these establishments is indescribably horrible. Extreme cleanliness
is desirable. Immersion of the guts in common salt is useful; so also the use
of impervious vessels, early removal of all refuse material, etc.
Brick and ballast burning are a frequent source of complaint in the
neighbourhood of towns. Brick burning is conducted either in kilns or clamps.
When bricks are burnt in closed kilns comparatively little nuisance arises; but
when they are burnt in open clamps the effluvia are very irritating, partly owing
to the fact that very commonly house refuse, containing vegetable and animal
matters, is burnt with the bricks. Clamp burning should be absolutely
prohibited in the neighbourhood of large towns.
In Ballast burning stiff clay is converted by the agency of heat into a
brick-like material, which is of use in road-making. The clay is usually
burnt in heaps, mixed with ashes and breeze from dust-bins. The process is
offensive unless carried on with precautions similar to those for brick-burning.
125
There are various methods of ascertaining the quality of the
air in enclosed spaces, of which not the least useful is the information
furnished by the sense of smell, on entering a room from the
external air. Besides the evidence given by the senses, chemical and
microscopical examination of the air gives important information,
while the thermometer and hydrometer ascertain the temperature and
degree of moisture.
Examination by the Senses.—The dull grey haze hanging
over a town, when it is viewed from a distance, indicates comparative
impurity of its atmosphere, and the presence of a considerable
amount of suspended matter, including smoke.
The smell of a stagnant atmosphere is a good preliminary guide
to its condition. The fact that a room has been occupied for some
time without efficient ventilation can be at once detected on entering
a room from the external air. The sense of smell is extremely
delicate; it has been estimated that the 3 ∕ 100,000,000 part of a grain of
musk can be apprehended by it. But nothing is so soon dulled as
the sense of smell. An atmosphere which did not appear to be
unpleasant while remaining in a room, is intolerable when one
returns to it after a few minutes in the open air. It is important not
to confound the “closeness” perceived by the sense of smell, with the
oppression due to the high temperature of a room. The two are
easily distinguished (unless the two co-exist) by a reference to the
thermometer, which ought always to be placed in rooms inhabited
during the evening. The remedy for a close room is to allow free
entry of fresh air, and not allow the fire to go down, as is so
commonly done, under the impression that the closeness is due
to heat.
De Chaumont has made many experiments, shewing how
accurate is the information given by an acute sense of smell. Carbonic
acid is destitute of odour, but as its amount is usually
proportionate to that of the organic matter producing closeness, it
may be taken as an index of the amount of impurity present in living
rooms. De Chaumont found that the limit of smell is reached when
carbonic acid amounts to 6 parts in 10,000 of air, or half as much
again as in the external air. In the following extracts from his126
experiments, there was a close accordance between the evidence of
his sense of smell and the amount of carbonic acid:—
| At |
14·80 |
per 10,000 |
Extremely close and unpleasant. |
| „ |
10·90 |
„ |
Extremely close. |
| „ |
9·62 |
„ |
Very close. |
| „ |
9·21 |
„ |
Close. |
| „ |
8·43 |
„ |
Not very foul. |
| „ |
8·04 |
„ |
Close. |
| „ |
6·58 |
„ |
Not very close. |
| „ |
5·68 |
„ |
Not close. |
He also found that humidity of the air had marked influence
in rendering the smell of organic matter perceptible, even more
than a rise of temperature. The sense of smell is doubtless aided
in detecting impurities in the air, by the besoin de respirer, a feeling
of oppression caused by the deficiency of interchange between the
blood and air. The state of cleanliness of the room as well as of
the persons in it influences smell; hence there may not be in
particular instances exact correspondence between excess of
carbonic acid and of organic matter.
Chemical Examination.—The estimation of nitrogen and
oxygen in air is usually unnecessary, as these vary but little. The
oxygen is, however, reduced in frequently re-breathed air. The ill
effects of an often-breathed atmosphere are due not only to deficiency
of oxygen, but also to the addition of carbonic acid and organic
matters, rendering difficult the interchange between oxygen and the
blood.
The Estimation of Carbonic Acid is of great importance, as
under ordinary circumstances, its amount is a fairly exact indication
of the amount of contamination in the air.
Pettenkofer’s Method.—A carefully dried glass vessel containing a
gallon of water is filled with the air to be examined, by emptying the water in
the room, the air of which is to be examined. Fifty cubic centimetres of clear
freshly prepared baryta water are then added, and the stopper of the bottle
then replaced. It is then well shaken, and afterwards allowed to stand for an
hour. The carbonic acid combines with part of the baryta to form barium
carbonate; and the baryta water remaining is consequently diminished in
alkalinity. Given the alkalinity of the baryta water before and after the
experiment, and the difference will give the amount of baryta which has
combined with carbonic acid.
The alkalinity of the baryta is estimated by a standard solution of oxalic
acid, of such a strength that 1 c.c. is the equivalent of 0·5 c.c. of CO₂. The
indicator used in making this test is phenolphthalein, which colours baryta
water red, but its colour disappears when neutralization is reached.
The following example is taken from “Pakes’ Laboratory Text Book of
Hygiene,” p. 292:—
The jar is found to contain 3,950 c.c.
As 50 c.c. baryta water were run into the jar, the air experimented on =
3,950-50 = 3,900 c.c.
On titrating 25 c.c. of the original baryta water, 22·50 c.c. standard acid
solution were required to neutralise it.
The baryta water in the jar required 19·35 c.c.
22·50-19·35 = 3·15 c.c. = difference of acid used.
But 1 c.c. acid = 0·5 c.c. CO₂ at 0° C. and 760 mm. of mercury.
Therefore CO₂ taken up by 25 c.c. of baryta = 3·15∕ 2 = 1·575 c.c.
As 50 c.c. were used the CO₂ absorbed by the baryta = 3·15 c.c.
This was present in 3,900 c.c. of air. Therefore the CO₂ = 0·80 per cent.
127
Correction may be required for variations from the normal pressure of 760
mm. and normal temperature of 0° C., in accordance with ordinary rules.
In Lunge and Zeckendorf’s Method, the air to be examined is
pumped through a glass bottle in which is 10 c.c. of a N ∕ 500 solution of Na₂CO₃
containing phenolphthalein as an indicator. The air is pumped by a hand
pump through this solution until the phenolphthalein is decolourized. The
number of times the ball of the pump has been squeezed indicates the amount
of CO₂ present in accordance with a table prepared from separate experiments
by Pettenkofer’s method.
Dr. Angus Smith’s plan for the estimation of carbonic acid in air is similar
in principle to the last calculations. It is based on the fact that the amount of
carbonic acid in a given volume of air will not render turbid a given amount
of lime water, unless the carbonic acid is in excess.
Table.—To be used when the point of observation is “No precipitate.” Half an
ounce of lime water containing ·0195 gramme lime.
Air at 0° C. and 760 M. M. Barometric pressure.
| CARBONIC ACID IN THE AIR PER CENT. |
VOLUME OF AIR IN CUBIC CENTIMETRES |
SIZE OF BOTTLE IN CUBIC CENTIMETRES |
SIZE OF BOTTLE IN OUNCES AVOIRDUPOIS. |
| ·03 |
571 |
584 |
20·63 |
| ·04 |
428 |
443 |
15·60 |
| ·05 |
342 |
356 |
12·58 |
| ·06 |
285 |
299 |
10·57 |
| ·07 |
245 |
259 |
9·13 |
| ·08 |
214 |
228 |
8·05 |
| ·09 |
190 |
204 |
7·21 |
| ·10 |
171 |
185 |
6·54 |
| ·11 |
156 |
170 |
6·00 |
| ·12 |
153 |
157 |
5·53 |
| ·13 |
132 |
146 |
5·15 |
| ·14 |
123 |
137 |
4·82 |
| ·15 |
114 |
128 |
4·53 |
| ·20 |
86 |
100 |
3·52 |
| ·25 |
69 |
83 |
2·92 |
| ·30 |
57 |
71 |
2·51 |
The foregoing table shows how to apply this method. The first and second
columns state the ratio of carbonic acid in a quantity of air which will give no
turbidity or precipitate in half an ounce of lime water; the third column gives
the corresponding size of the bottle in cubic centimetres; and the fourth column
gives the same in ounces. Thus different sized bottles, each containing half an
ounce of lime water, will indicate with a fair degree of accuracy the ratio of
carbonic acid in the air containing them, by giving no precipitate when the
bottle is well shaken. For instance, if a pint bottle is used and there is no
precipitate with half an ounce of lime water, it indicates that the ratio of
carbonic acid does not amount to ·03 per cent.; if an eight-ounce bottle be used,
and there is no precipitate, it indicates that the ratio does not amount to ·08 per
cent., and so on. The air of a room ought never to contain more than six
parts of carbonic acid in 10,000 of air, or ·06 per cent., i.e. a 10½ ounce bottle
full of the air shaken up with half an ounce of clear lime water ought to give
no precipitate.
Dr. Haldane has recently described (Journal of Hygiene, No. 1, 1901) a
method of estimating CO2, which, although it appears complicated, is really
both simple and convenient. For particulars, see the above Journal.
The Estimation of Organic Impurities may be accomplished approximately
by drawing a definite amount of air by means of an aspirator, through a128
dilute solution of permanganate of potassium of known strength. The result is
stated by giving the number of cubic feet of air required to decolourise .001
gramme of the permanganate in solution. Sulphuretted hydrogen, sulphurous
acid, and other substances in air likewise decolourise the permanganate; these
ought to be separately tested for, and allowance made.
The Estimation of Ammonia, whether free or derived from albuminoid
impurities, is a matter requiring very delicate processes. It is accomplished in
the same way as the estimation of ammonia in water, the air being drawn
through perfectly pure distilled water, and then the analysis proceeded with as
a water analysis. The mere presence of free ammonia may be determined by
exposing to the air strips of filtering paper dipped in Nessler’s solution, which
become brown if there is any ammonia in the air.
Microscopical Examination is required for the detection of
suspended matters. These are the most potent for harm, containing
sometimes the germs of infectious diseases. The suspended
matters scattered throughout the air may be collected by Pouchet’s
aeroscope. This consists of a small funnel drawn out to a fine
point, under which a slip of glass is placed moistened with glycerine.
Both funnel and glass are enclosed in an air-tight chamber, connected
by tubing with an aspirator, by means of which when water
is allowed to escape from it, air is drawn through the funnel
and its particles impinging on the glycerine are there arrested.
Glycerine may be objectionable from the foreign particles previously
contained in it. Various other plans have been devised, one of
which is to draw the air through a small quantity of pure distilled
water and then examine a drop of it. By microscopic examination
large particles can be detected. For the detection of bacteria and
their spores more delicate methods are required.
The Bacteriological Examination of air is usually conducted
as follows. Air is drawn through a wide glass tube (Hesse’s tube),
which has been previously sterilised, and on the inner side of which
liquid gelatine has been allowed to solidify. The air as it passes
over the gelatine deposits any germs present in it. The entrance of
any further germs is prevented by closing the tube, and it is then
left to stand for two or three days. Moulds and colonies of bacteria
will develop in the gelatine, and these can be counted and
differentiated by their appearance and by further tests. In closed
rooms the number of microbes (i.e., bacteria and moulds) ought not
to be more than 20 per litre of air in excess of those in the outside
air; and the ratio of bacteria to moulds ought not to exceed 30 to 1.
Examination of Temperature and Moisture.—The temperature
should be observed at the point most remote from an open
fire-place, and compared with the external temperature. For
methods of estimating moisture, see page 240.
It may be useful to recapitulate at this point the desiderata in
an inhabited room. The temperature should be 60-62° Fahr.,
the amount of carbonic acid should not exceed ·06 per cent. and
the humidity should range between 73 and 75 per cent. of the
amount required to produce saturation. The dry bulb thermometer
should read 63-65° Fahr., the wet bulb 58°-61° Fahr., and the
difference between the two should not be less than 4° or more
than 8°.
129
CHAPTER XIX.
THE PURIFICATION OF AIR.
In addition to the artificial measures which will be discussed in the
next chapter, various natural agencies are constantly at work for
the removal of the impurities discussed in preceding chapters. Of
these, the most important are the action of plants, the fall of rain,
natural methods of ventilation, and certain natural constituents of
the atmosphere.
1. Plants, by virtue of the chlorophyll contained in their green
parts, absorb carbonic acid from the atmosphere, liberating oxygen
in an active condition. In addition, ammonia and nitrous and
nitric acids are dissolved from the air by rain-water, and assimilated
by plants. During the night plants only give off carbonic
acid.
2. The Fall of Rain clears the atmosphere of any solid particles
contained in it, the impurities being transferred to rain-water
which generally contains an appreciable amount of ammonia as
well as other impurities. Rain not only washes and purifies the
air, but by washing the ground, diminishes dust, and prevents its
escape into the air. It is the great natural scavenger.
3. Ventilation—that is, the interchange of pure and impure
air, is constantly being effected. Before entering on the details of
ventilation, we must consider the physical causes at work which
tend to purify the air, apart from all artificial contrivances. These
are three in number—namely, diffusion, winds, and differences of
temperature of masses of air.
(1) Diffusion causes the rapid mixture of gases placed together.
Every gas diffuses at a certain rate—namely, inversely as the square
root of its density. In any room which is not air-tight, diffusion
is constantly occurring, air passing in and out at every possible
point. Through chinks and openings in the carpentry-work of a
room, the air diffuses rapidly. Bricks and stone commonly allow
air to pass through them; diffusion occurs to a slight extent even if
the wall is plastered, but very little through paper. Diffusion
alone is quite insufficient to purify a room under ordinary circumstances;
and solid particles including the organic matter evolved
from the skin and lungs, not being gaseous, are unaffected by it.
To remove these, the room must be periodically flushed with air,
and washing of all dirty surfaces must be carried out.
Diffusion sometimes produces evil results, when the sanitary
arrangements of a house are bad. If there is a leakage of sewage130
under the kitchen floor, the foul gases from it diffuse upwards;
occasionally foul air diffuses from the dust-bin through the wall
into the rooms of a house. These results are helped by the fact
that the internal temperature of a house is commonly higher than
the external.
(2) Differences of Temperature cause active movements of
air. In fact winds are caused by movements between large masses
of air of unequal temperature and consequently of unequal density.
Light gases ascend, as familiarly illustrated by the smell of dinner
perceived in bedrooms, or the smell of a cigar lit in the hall
perceived in the attic. In rooms differences of temperature of the
air are caused by the heat of fire, gas, and our own bodies.
Currents of air result; the warmer and lighter air ascends up the
chimney or towards the ceiling, while colder and denser air rushes
in under the door or through the floor, etc. The lighter gases carry
with them solid particles in suspension and thus tend to remove the
most important impurities. Assuming that the external air is colder,
if admitted into the lower part of a room, it produces a draught; if
admitted at the top of a room, being heavier, it falls by its own
weight on the heads of those in the room. The problem of
ventilation is to secure a sufficient interchange of air without the
production of perceptible currents.
Movements of air are constantly occurring, so long as the
temperature of the air is subject to changes. This cause alone
will suffice to ventilate all rooms in which the air is hotter than the
external air. It may thus happen that a room with windows and
doors closed in winter, may possess purer air than the same room
in summer with these thrown widely open. The value of diffusion
of air through the walls, and the influence of temperature on this
diffusion are well illustrated by some experiments of Pettenkofer.
When the difference between the outside and inside temperatures was 34°
Fahr. (66° inside and 32° outside), and the doors and windows were shut, an
ordinary room in his house, of the capacity of 2,650 cubic feet, which was built
of brick, and furnished with a German stove instead of an open fire-place, had
its entire atmosphere changed once in an hour. With the same difference of
temperature, but with the addition of a good fire in the stove, the change of air
rose to 3,320 cubic feet per hour. On lessening the difference between the
external and internal temperature to 7° Fahr. (64° and 71°), the change of air
was reduced to only 780 cubic feet per hour. In these experiments, all crevices
and openings in doors and windows were pasted up.
It is instructive to note the greater amount of ventilation
effected through the walls, etc., than by the draught of the stove.
The amount of ventilation through walls varies with the
material of which they are built. Mortar is exceedingly porous
when dry; sandstones and bricks are easily permeated by both
water and air. Limestone is almost impervious to air, but requires
much mortar in building, which effects a partial compensation
(see page 206).
The rise of temperature caused by the bodily heat and by the
combustion of illuminating agents, is well shown by some figures
of Dr. Angus Smith. He found that the rise of temperature of 170131
cubic feet of air in one hour, produced by the bodily heat of one man
was 5°·6 Fahr.; by the combustion of a candle 3°·8 Fahr. Thus, in
a room 8 feet high, 4 feet broad, and 6 feet long, a man burning a
candle would in an hour raise the temperature from 60° to 70°
Fahr. This rise in temperature would not only cause currents of
hot air towards the upper part of the room, but would probably
make the room uncomfortable, and so lead to the opening of a
door, etc.
(3) Winds are of great value in flushing rooms with fresh air.
They ought to be utilised as often as possible, by throwing windows
widely open; without, however, taking the place of constant
ventilation in the intervals. They are especially valuable in getting
rid of organic matters which are unaffected by diffusion.
The wind will pass through wood, and even brick and stone
walls. When it is allowed to pass directly through a room, as
from window to door, it produces a more powerful effect than can
be produced in any other way. The average rate of movement of
winds in this country is 10 feet per second, or about 7 miles an
hour. If the surface which a man exposes to this average wind
= 6’ × 1½’ = 9 square feet, then 90 cubic feet of air flows over
him in one second, and 324,000 in an hour. If 3,000 cubic feet
were the allowance for each person indoors—a much greater
allowance than is usually given—he only receives 1 ∕ 108 of the air with
which he is supplied in the open.
Winds act as a ventilating agent in two ways—directly by
perflation, driving impure air before them, or freely mixing with
it; and indirectly by aspiration, drawing the impure air along
with them. In the last case, the wind causes a partial vacuum
on each side of its path, towards which all the air in its vicinity
flows. Thus, the wind blowing over the top of a chimney
causes a current at right angles to itself up the chimney. In a
spray-producing apparatus we have a familiar instance of the
same principle, the current of air or steam along the horizontal
tube causing the fluid to rise in the vertical tube till it is scattered
in spray. In Sylvester’s plan of ventilation, both these forces are
used (see page 150).
4. Certain Constituents of the Atmosphere have an
important purifying effect. Of these oxygen is by far the most
important. By its means organic impurities become oxidised, and
thus rendered harmless. It is probable that much of this oxidation
is effected by means of ozone—a peculiarly active and concentrated
form of oxygen. A large part of this ozone is probably produced
during thunderstorms and similar electrical disturbances of the
atmosphere. The ammonia and organic impurities in air become
changed into nitrites and nitrates—chiefly of ammonium—and
being washed down by rain, form an important part of the food of
plants.
5. For Chemical Measures of purification of the atmosphere
see page 324.
132
CHAPTER XX.
GENERAL PRINCIPLES OF VENTILATION.
The Amount of Air required.—Ventilation is chiefly
concerned with the removal of the products of respiration, just as
sewage is chiefly concerned with the removal of the solid and liquid
excreta.
In a less degree it is required for removing the impurities
produced by the burning of gas, candles, and lamps. The main
problem, however, is the removal of the respiratory products.
The amount of carbonic acid in air is usually fairly proportional
to that of the other respiratory products. It may
therefore be taken as a measure of the impurity of the air.
There are, however, certain fallacies in this test. In soda water
manufactory, for instance, there would be a comparatively harmless
excess of carbonic acid. In dirty rooms, and in hospitals and
other institutions where rooms are not vacated for a considerable
period, the amount of organic matter present is often in excess of
what would have been anticipated, judging by an estimation of the
carbonic acid. This is strikingly shown by some valuable
researches at Dundee, which are summarised in the following table.
If we take the average amount (in excess of outside air) of carbonic
acid, organic matter, and micro-organisms respectively in houses of
four or more rooms as unity, then in one or two-roomed houses or
tenements we have as follows:—
|
HOUSES OF FOUR
ROOMS AND UPWARDS. |
TWO-ROOMED HOUSES. |
ONE-ROOMED HOUSES. |
| Carbonic acid |
1 |
1·5 |
2·0 |
| Organic matter |
1 |
1·6 |
4·4 |
| Micro-organisms |
1 |
5·1 |
6·7 |
It is evident that in these cases the carbonic acid did not
increase in the same proportion as the organic matter and
micro-organisms, and that it alone does not form a sufficient test
of the impurity of any given atmosphere. The amount of
carbonic acid, however, is a valuable and convenient test of
the condition of the air of a room, and the problems of ventilation,
of which examples are given on page 137, are based on its amount.
The Standard of purity is somewhat difficult to fix. The
external air ought only to contain 4 parts of carbonic acid to 10,000133
parts; but it is almost impossible to maintain this degree of purity
in inhabited rooms. The experiments made by Drs. Parkes and
De Chaumont showed that when the carbonic acid is ·06 per cent.,
or in the proportion of 6 parts in 10,000 of air, the air begins to be
perceptibly stuffy (page 125); this may therefore be taken as the
limit of impurity. Pettenkofer has adopted the limit of ·07 per
cent.7
The problem then is to discover the amount of pure external
air (containing ·04 per cent. of carbonic acid) that will be required to
pass hourly through a room, for every person in that room, in order
to keep the carbonic acid at the ratio of ·06 per cent.
This may be ascertained by actual observation of the air of
rooms in which a given number of persons are placed; or by
calculations from physiological data.
As the result of numerous experiments on the atmosphere of
prisons, barracks, etc., where the amount of fresh air supplied per
hour is exactly known, it is found that in order to keep the carbonic
acid at ·06 per cent., 3,000 cubic feet of pure air are required per
head per hour; 2,000 cubic feet keep the carbonic acid at ·07 per
cent.; 1,500 cubic feet at ·08 per cent.; and 1,200 cubic feet at ·09
per cent.
For the removal of the products of combustion of gas, an
additional supply of air is required, for the amount of which, see
page 116.
Where a number of sick persons are collected, as in hospitals
and workhouses, a much freer supply of air is required. Much
depends, however, on the cleanliness of the wards, and on whether
the ventilation is constant in character. In St. Thomas’s Hospital,
the space allotted to each ordinary patient is 1,800 cubic feet, and
to each patient in the fever wards 2,500 cubic feet. Thus, by
changing the air of the wards twice in the hour, an abundant
supply of fresh air is ensured. The mortality after operations,
and in all fevers, is much diminished by a free supply of air.
Soldiers are allowed 600 cubic feet of space per head in their
sleeping rooms, which involves a change of the air five times per
hour, in order that the carbonic acid may be maintained at ·06 per
cent. The limit of overcrowding for lodging-houses is usually fixed
at 300 to 500 cubic feet, but this is too little.
The amount of pure air required in order to keep the carbonic
acid in a room at ·06 per cent., may also be ascertained from
physiological data.
An average adult expires 3 ∕ 5 (·6) cubic foot of carbonic acid per
hour. Now as the carbonic acid in air to be breathed must not
contain more than two parts in 10,000 (·02 per cent.) in excess of
what is present in external air (·04 per cent.), it follows that if x =134
the amount of fresh air required by an adult per hour in order to
keep the carbonic acid in the room down to .06 per cent., then:—
·02 : ·6 :: 100 : x.
x = 3,000 cubic feet.
Relation of Air Required to Cubic Space of Room.—If
we accept 3,000 cubic feet of air as the amount required per head
per hour, this may clearly be furnished by having a large room with
comparatively little circulation of air, or by having a small room
with frequent interchanges. Thus, supposing the cubic space
allowed to each individual is 1,000 cubic feet—that is, 10 feet in
every direction—the atmosphere will require changing three times
per hour.
Now, it is found that when a current of air, at the temperature
of 55°-60° Fahr., is moving at the rate of less than one mile per
hour, it is not perceptible—that is, produces no draught. The rate
of a breeze, which is just perceptible, is 18 inches per second, or
one mile per hour. As draughts are objectionable, ventilation, in the
best sense of the word, means the supplying of abundant fresh air
at a rate of less than one mile per hour, or warmed air at a higher
rate. Air moving at the rate of 2½ miles per hour, or 3½ feet per
second, is perceived as a slight draught by all, at the average
temperature of our climate (about 50° Fahr.)
Where natural ventilation is employed, the difficulties of
thoroughly ventilating a small space, without draught, are very
great.
A change of air three or four times in an hour is all that can be
borne under ordinary conditions in this country, and this necessitates
a supply of 1,000 or 750 cubic feet of space respectively for each
individual. And a change of this frequency is commonly not
effected; the ventilating apparatus may fail temporarily, or may be
wilfully stopped up, or there may be no means of ventilation; it is
essential therefore to have as large a cubic space as possible. A
large cubic space, does not obviate the necessity for efficient
circulation of air. It is, however, advantageous, not only on
account of the initial longer time before the air reaches the limit
of impurity, but also because there are less draughts, and there is a
larger wall surface and larger windows for unperceived ventilation.
Common Errors as to Ventilation.—(1) In relation to the
cubic space of a room, it is most important to note that a lofty
ceiling does not compensate for deficiencies in floor-space. One hears,
“lofty” and “airy” rooms spoken of as though the two terms were
necessarily synonymous. This is by no means the case. The
impurities produced by respiration tend to accumulate about the
persons who have evolved them, although it is true that in rooms
heated by gaslight, a large amount of hot and impure air collects
near the ceiling. The necessity of an abundant floor-space is
shown by the fact that a space enclosed by four high walls and
without a roof, will, if crowded, speedily become offensive. Twelve
feet is quite high enough for large rooms in schools, hospital wards,
etc., and nine feet suffices for the rooms of a private dwelling-house.135
There is no objection to a greater height, if it is remembered that in
reckoning the practical cubic dimensions of a room, the height should
only be reckoned as twelve feet. Supposing 500 cubic feet is the
amount allowed per individual, then the floor-space should be
forty-two square feet, which would be furnished by a room about
8½ feet long and 5½ feet wide. In barracks, soldiers are allowed
fifty square feet of floor-space. In school-rooms the Education
Code requires that at least ten square feet of floor-space, and at
least 120 cubic feet shall be allowed for each child in average
attendance.
(2) It is commonly supposed that a large room compensates for
a deficient circulation of air. The cubic space of a room is really of less
importance than the capacity for frequent interchanges of air. Even the
largest enclosed space can only supply air for a limited period, after
which the same amount of fresh air must be supplied, whether the
space be small or large. Thus, supposing that as large a space as
10,000 cubic feet per head were allowed, the limit of purity would
in the absence of ventilation be reached in three hours, and after
that time an hourly supply of 3,000 cubic feet of air would be just
as necessary as if the space were only 200 cubic feet.
(3) It must not be overlooked that the furniture in a room must
be deducted from the breathing space, as the amount of air is
diminished by the space occupied by the furniture. About 10 cubic
feet ought to be allowed for each bed, and 3 to 5 cubic feet for each
individual in a room; projecting surfaces must be allowed for by
subtraction, and recesses by addition. The deductions to be made
for furniture are not of any great consequence, if there is a free
interchange of air; as the cubic space is of less importance than
free ventilation.
General Rules respecting Ventilation.—The two great
objects in ventilating being to remove all impurities from the air,
and to avoid draughts, it is important that—
1. The entering air should be, if possible, of a temperature of
55° to 60° Fahr. Whenever the temperature of a room differs from
the external temperature by 10° Fahr., a draught is certain to
ensue. It is impossible at all times to ensure the incoming air
being of the temperature of 60°, without some artificial means of
warming it. In this country it is seldom necessary to cool the
incoming air, but this may be managed in artificial systems of
ventilation by passing the incoming air over ice, or by using
compressed air which becomes cooled on expansion, or by passing
the incoming air through subterranean tunnels.
2. The entering air should be pure. When a room is hotter than
the passages and kitchens, air from the latter, whatever may be its
character, is drawn into the room. Similarly the ground-air under
the kitchen-floor or the air from ash-pits may be drawn into the
house, when no other means of ventilation are provided; and this is
often followed by evil results.
3. No draught or current should be perceptible from the incoming
air, except when it is wished to flush the room with air, by opening136
the windows wide. It is a common complaint that a room is
draughty, and, to remedy this, keyholes are stopped up, and mats are
placed at the bottom of the door, etc. The draught can often be
remedied by increasing the size and number of the openings through
which air is admitted, so that the current of air is not concentrated
and rapid. When this does not remedy it, the incoming air should
be warmed. A feeling of draught is very often due to the radiation
to and from a window, and disappears when a curtain or screen is
placed between the radiating surface and the occupant of the room.
4. The entry of air should be constant, not intermittent. The
occasional opening of a window or door will not compensate for the
lack of a constant interchange of air, although it forms a very
valuable adjunct, especially in the removal of organic particles
which do not follow the law of diffusion.
5. An exit should be provided for impure air, as well as an entrance
for pure air. The chimney furnishes this in most living-rooms, and
diminishes the necessity for other means of exit.
If the openings in a room for entrance and exit are properly
regulated, a rate of 5 feet per second (about 3½ miles per hour) will
provide sufficient air without any unpleasant draught in a room.
For instance, if the opening measure 1 square foot, then a rate of 5
feet per second will give five cubic feet of air per second, that is,
18,000 cubic feet per hour. But as only 3,000 cubic feet are
required, it follows that an opening one-sixth this size, i.e. 24 square
inches, is sufficient for each individual. Reckoning the same
amount for means of exit, 48 square inches is the size of the
ventilating orifices required by each individual.
6. A number of small divided openings are not collectively equal in
ventilating power to one large one having the same area. Thus, when a
ventilating orifice is divided into four parts, which have the same
collective area as the original orifice, it is found that only half as
much air passes through these as through the original orifice. In
order to obtain as much air, therefore, each opening must be equal
in size to half the original opening. This is in accordance with the
rule that the friction for air passing through openings is inversely to
the diameter of these openings, i.e. inversely to the square-root of
the area of the openings.
7. The most important requirements of perfect ventilation may
be recapitulated as follows:—
1st. The maximum impurity of air vitiated by respiration
should not exceed 6 parts carbonic acid per 10,000 volumes.
2nd. To ensure the maintenance of this standard, 3,000 cubic
feet of pure air must be supplied per head per hour.
3rd. In order to supply this amount of pure air, with ordinary
means of ventilation, 1,000 cubic feet at least must be allowed per
head in buildings always occupied.
137
CHAPTER XXI.
PROBLEMS AS TO VENTILATION.
The following formula enables many problems relating to ventilation
to be solved. Let p = the amount of poison (carbonic acid)
in every cubic foot of fresh air, viz. ·0004 cubic foot. Let A = the
number of cubic feet of fresh air delivered or available, P = the
amount of carbonic acid exhaled, and x = the amount of carbonic
acid per cubic foot in the room at the end of a given time. Then—
x = p + P ∕ A, whence A = P ∕ (x - p).
If the carbonic acid in the air of a room is ·75 per 1,000 volumes (that in the
outer air being ·4 per 1,000 volumes), and there are five persons in the room, how
much air is entering the room per hour?
- Here x = ·00075.
- p = ·0004.
- P = ·6 (i.e. number of cubic feet of carbonic acid expired by each person per hour).
- Now x = p + P ∕ A.
- ·00075 = ·0004 + ·6 ∕ A.
Therefore A = about 1,700.
Thus 1,700 cubic feet are required for each individual to keep the air
within the given limit, and five times this amount will be required for five
persons = 8,500 cubic feet.
A room has been occupied for one hour, at the end of which the total carbonic
acid present was found to be 1·1 per 1,000 parts. The carbonic acid in the open air
amounting to ·0004 per cubic foot, find the quantity of air supplied per hour.
- Here x = ·0011.
- p = ·0004 and P = ·6.
- Hence ·0011 = ·0004 + ·6 ∕ A.
- Therefore A = 857 cubic feet.
If six persons are in a room containing 3,000 cubic feet, and there is a supply of
2,000 cubic feet of air per head per hour; how much carbonic acid is there in the air
of the room at the end of 4 hours?
- Here p = ·0004.
- P = ·6 × 6 × 4 = 14·4.
- A = 2,000 × 6 × 4 + 3,000 = 51,000.
- x = ·004 + 14·4 ∕ 51,000 = ·000682 = 6·82 parts CO₂ in 10,000 of air.
138
The air of a room occupied by 6 persons and containing 5,000 cubic feet of
space, yields 7·5 parts of CO₂ per 10,000 parts of air. How much air is being
supplied per hour?
A = P ∕ (x - p) = ·6 x 6/(·00075 - ·0004) = 10,280 cubic feet.
In the same room what would be the condition of the air at the end of 4 hours?
x = ·0001 + ·6 × 6 × 4/(10280 × 4 + 5,000)
= ·0004 + 14·4 ∕ 46,120 = ·000712 = 7·12 of CO₂ in 10,000 of air.
Given two sleeping rooms, Y 10 ft. by 15 ft. and 10 ft. high, Z 15 ft. by 20 ft.
and 12 ft. high, with three adults in each; how much fresh air would you supply in
each? What would be the condition of the air of each of the rooms after, ¼;, ½, 1,
and 2 hours respectively?
Amount of fresh air to be supplied in Y—
A = P ∕ (x - p) = ·6 × 3/(·0005 - ·0004) = 9,000 cubic feet per hour.
Condition of air in Y after ¼ hour—
Here p = ·0004.
P = ·6 × 3 ∕ 4= ·45.
A = 9,000 ∕ 4 + 1,500 = 3,750.
x = ·0004 + ·45 ∕ 3,750 = ·00052.
At the end of 2 hours—
x = ·0004 + 3·6∕(18,000 + 1,500) = ·000584.
And similarly for Z.
Suppose two rooms, one 10 feet cube, the other 50 feet by 20 feet and 15 feet
high, have continuously admitted into each of them a volume of fresh air containing ·04
parts carbonic acid per 100 parts, amounting to 2,000 cubic feet per hour, so as to
replace to that extent the air of the room; suppose also that an average adult be
placed in each room: show by detailed calculation what would be the condition of
impurity of air in each room, as measured by carbonic acid, at the end of 4 hours
and 12 hours respectively.
In the case of the first room—
- P = ·6 × 4 = 2·4.
- A = 2,000 × 4 + 1,000 = 9,000.
- p = ·0004.
- x = ·0004 + 2·4 ∕ 9,000 = ·000667.
The amount of impurity at the end of 12 hours, and in the second room
may be similarly ascertained.
Ventilation in relation to Temperature.—The temperature
of a given atmosphere is a most important factor in determining
the ease with which it is replenished from the external air. Speaking
generally, the greater the difference between the temperature of
two masses of air the more rapidly an interchange occurs.
Air has weight. A column of it one inch square and extending
to the uppermost limit of the atmosphere weighs about 14·6 lbs.,139
and exerts this pressure on all substances at the surface of the
earth. This pressure is exerted uniformly in all directions; but for
this fact our chests would be crushed in by the external pressure on
them, which amounts to over four tons. If the atmospheric pressure
is diminished at any point, it is evident that the surrounding air will
tend to press in this direction. Now, when air is heated it expands,
and consequently the heavier fresh air flows in from all sides and
pushes the lighter air upwards.
The expansion of air for every increase of 1° Cent. is ·003665 (1 ∕ 273), for
every increase of 1° Fahr. is ·00203 (1 ∕ 492). Thus if the air in a room is 20° F.
warmer than that outside, it will be expanded to 1 ∕ 25 additional bulk.
Thus if M = volume of a given air at 32°, with the barometer at 30
inches, and
M1 = volume at temperature t° above 32°, while a = co-efficient
of expansion for each degree of elevation of temperature, then the dilatation
effected by heat will be expressed by the formula—
M1 = M (1 + at).
When the temperature is decreasing
M1 = M (1-at).
If the air in a chimney flue is cooler than the air of the room with which
it communicates, it will flow down into the room. It is the object of an
economical fire-place to cause the chimney to act as an outlet for the products of
combustion and for the impurities of the air of the room with the smallest
possible waste of heat. Short of producing a down draught of cold air and
smoke, the smaller the difference between the temperature of the air of a room
and of the air escaping near the top of the chimney, the greater the economy of
fuel.
The movement of air in flues and other outlets is governed by
general laws, like those governing the general movements of fluids, but allowances
require to be made for friction in the channels of entrance and outlet.
The theoretical velocity, when friction is not taken into account, may be
calculated by a formula based on what is known as the law of Montgolfier, or the
law of spouting fluids. According to this law, fluids pass through an
opening in a partition with the same velocity as a body would attain in
falling through a height equal to the difference in depth of the fluid on
the two sides of the partition, i.e. to the difference of pressure on the
two sides. Thus, if AB equals the height of a column of air at, say,
50° F., and AC is the height of the same quantity of air heated to 60°,
then the velocity with which the warmer air ascends will be that which
a body would acquire in falling from C to B.
Now the velocity in feet per second of falling bodies is about eight
times the square root of the height from which they have fallen; and
the formula for determining this is—
v = c √(2gh) = 8·2c √h.
- g = 32·17 feet per second;
- h = distance fallen through by the body;
- c = a constant determined by experiment, and expressing
the proportion of the actual to the theoretical velocity.
Adapting this formula to the special circumstances under which Montgolfier’s
formula holds, we find that the force which drives the warm air up the flue is
the force of gravity, i.e. of the excess of the weight of a column of cold air over
the weight of a column of warm air of exactly the same size (represented by BC
in the preceding diagram). The difference of the two weights or pressures is
found by multiplying the distance from the point of escape of heated air out of
the room (fire-place or elsewhere) to the point of escape into the outer air (top of140
chimney or other point of exit), by the difference in temperature inside and
outside, and again multiplying this product by 1 ∕ 492 for degrees of Fahrenheit
temperature, or 1 ∕ 273 for degrees Centigrade.
Thus omitting c for the present, we have—
v = √(2gh(t - t1)/492) = 8·2√(h(t - t1)/492)
Where t = temperature in the chimney,
t1 = temperature of the external air, and
h = height of chimney.
Example.—The chief means of ventilating a given room is by its open fire-place. The
temperature in the chimney is 100° F., that of the external air 40°, and the height
of the chimney 50 feet; what is the velocity with which air is leaving the room?
v = 8·2 √((100 - 40) × 50 ∕ 492)
= 20.
This gives the theoretical velocity, but the real velocity will differ from the
theoretical by an amount varying from 20 to 50 per cent.
It will be evident, from what has been said, that the movements of the air
in a confined space are dependent upon (1) the difference between the internal
and external temperatures; (2) the area and friction at the apertures through
which air enters and leaves the room; and (3) the height of the column of
ascending warm air. The higher the chimney (assuming it to contain warm
air), the greater the draught and the more efficient the ventilation of the room
communicating with it. Hence ventilation is more difficult in upper rooms of
large houses and in single-storeyed houses than in the lower storeys of large
houses.
Allowance for Friction.—Practically the friction varies greatly according
to the size, form, and material of outlet for air. A rough or sooty or angular
chimney greatly impedes the outgoing current of air.
It is usual to reduce the theoretical velocity by 20 to 50 per cent. Apart
from the friction which is governed by roughness and length of channels, that
due to bends in the channel may be calculated by the formula 1/(1-sin2 θ), θ being
the angle at any bend in this channel.
(It may be convenient to note that—
- sin2 90° = 1,
- sin2 60° = ¾
- sin2 45° = ½,
- sin2 30° = ¼.)
Thus every right angle in a bent shaft reduces the velocity in it by one-half.
The loss by friction in two similar tubes of equal sectional area varies (1)
directly with the square of the velocity of the air currents; and (2) directly with
the length of the outlet channel. In two similar tubes of unequal size the loss
by friction is (3) inversely as the diameter of the cross-section in each.
When two tubes are of different shapes, the loss by friction is inversely as
the square roots of the sectional areas.
Owing to the variable value of the co-efficient of friction (called c in the first
formula given), it is usually preferable to measure the actual rate of progress of
air through a given flue by means of an anemometer (wind measure). Then
the velocity of the current of air and the area of the cross section of the flue
being given, the volume of air discharged in a given time is represented by the
product of these two and the time which has elapsed.
Thus, q = a × v.
Where q = quantity of air discharged in a given time, a = area of cross
section of flue, v = velocity of current.
By means of this formula, the area of chimney required to discharge a
given volume of air at a given average velocity can be ascertained. Thus—
a = q ∕ v.
141
The application of the preceding principles and formulæ will be rendered
clearer by the following examples.
How much inlet and outlet area per head will be required to give 10 persons in a
room of 5,000 cubic feet capacity, 2,000 cubic feet of air per head per hour, supposing
that the outside temperature is 40°, while the internal temperature is 60°, and the height
of the heated column of air 20 feet?
First ascertain the velocity of entrance and exit of air.
v = 8·2√(h(t - t1)/492)
= 8·2√(20(60 - 40)/492) = 8·2 × ·902.
= 7·3964 = velocity in feet per second.
If we allow one-fourth for friction, then there remains a velocity of 5·5473
feet per second.
5·5473 feet per second = 19700·8 feet per hour.
Now, a = q ∕ v
= 2,000 ∕ 19700·8 = ·1015 square feet.
= 14·6 square inches.
Thus the size of the outlet required per head is 14·6 square inches. The
size of the room and the number occupying it do not enter into the question,
except for a short time at the beginning. (See page 135.)
The amount of inlet required will also be 14·6 square inches per head.
Theoretically it ought to be slightly less than that required for outlet, as the
outgoing air is more expanded than that entering the room; but practically no
allowance need be made for this fact.
The total amount of inlet and outlet required per head = 29·2 square inches.
If the mean temperature of a room is 61°, the external temperature 45°, while the
heated column of air is 50 feet, and the required delivery of air 2,000 cubic feet per
hour, find the size of inlet and outlet.
v = 8·2√(h(t - t1) ∕ 492)
= 8·2√(50(61 - 45) ∕ 492)
= 10·55 feet per second.
= 37,980 feet per hour.
If we make no allowance for friction, then
a = q ∕ v
= 2,000 ∕ 37,980 square feet.
= 2,000 × 144 ∕ 37,980 = 7·58 square inches.
This gives the required size of outlet. The size of inlet and outlet together
= 15·16 square inches.
If 3,000 cubic feet of air are supplied in one hour through an aperture of 12 square142
inches to a room containing 1,000 cubic feet of space, at what rate does the air enter the
room?
12 square inches = 1 ∕ 12 square foot.
a = q ∕ v
1 ∕ 12 = 3,000 ∕ v
Therefore
v = 36,000 feet per hour.
= 10 feet per second.
If a room is supplied with 3,000 cubic feet of air per hour, through a single
opening, what must be its area, if the rate of movement of the air is 5 feet per second?
5 feet per second = 18,000 feet per hour.
a = 3000 ∕ 18000 = 1 ∕ 6 square foot.
= 24 square inches.
As already stated, the difficulties connected with the estimation of amount
of friction greatly detract from the practical value of the formulæ just given.
Even the results given by anemometers are not always trustworthy, but by comparing
the results given by them with those obtained by the use of Montgolfier’s
formula an approximation to the truth can be obtained.
The ordinary anemometer consists of four tiny vanes fixed to a spindle, so
that revolutions are caused by the current of air the velocity of which is to be
measured. The revolutions are counted by a mechanical arrangement. The
value of the revolutions of the vanes has to be first determined by direct
experiment; a known bulk of air being forced through a channel of known size
at a uniform rate, and the instrument graduated accordingly. In Fletcher’s
anemometer a modification of the manometer or pressure-gauge has been used
for the same purpose.
Inlets and Outlets.—Having given the average velocity of the wind, the
size of a room, and the number of persons occupying it, the size of inlet opening
required can easily be calculated.
Find the size of inlet for air in a room occupied by one person, the air moving at
the average velocity of 5 feet per second, assuming that 3,000 cubic feet of air are to be
supplied per hour.
Let x = size of inlet.
Then x × 60 × 60 × 5 = 3,000.
Therefore x = 3000 ∕ 18000 = 1 ∕ 6 square foot.
= 24 square inches.
Given that the air moves at a velocity of 10 feet per second, and that the area of
the inlet aperture into a room is 12 square inches, find how much air enters the room in
an hour.
Let y = amount of air.
Then 10 × 60 × 60 × 12 ∕ 144 = y.
Therefore y = 3,000 cubic feet of air.
Calculations as to supply of air in a room founded on the average velocity
of air-currents are, however, much less trustworthy than when the velocity is
determined, as previously explained, by means of Montgolfier’s formula, or,
better still, by an anemometer.
The Commissioners on Improving the Sanitary Condition of Barracks and
Hospitals, in their report (1861) recommended for inlets, one square inch for
every 60 cubic feet in the contents of the room; or one square inch for every
120 cubic feet in the contents, if warm air is admitted round the fire-grate. For
outlet shafts on lower floors, one square inch to every 60 cubic feet, slightly
increasing for the higher storeys.
Amount of Air-space required.—We may take 3,000 cubic
feet of air as the average amount of air required hourly by each
individual, and inasmuch as the air of a room cannot be changed143
oftener than three times an hour without producing an unpleasant
draught, it follows that at least 1,000 cubic feet of space must be
allowed per person.
This may be compared with the amount actually supplied under various
circumstances.
| In the British Army for each soldier— |
|
In permanent barracks |
600 |
cubic ft. |
|
In wooden huts |
400 |
„ |
|
In hospital wards at home |
1,200 |
„ |
|
In hospital wards in the tropics |
1,500 |
„ |
|
|
In general hospitals |
1,000-1,500 |
„ |
|
In fever hospitals |
2,000-3,000 |
„ |
|
In workhouse hospitals |
850-1,200 |
„ |
|
In common lodging houses |
300 or 350 |
„ |
|
Do., if occupied night and day |
350 or 400 |
„ |
|
In workhouses |
300 |
„ |
| In schools— |
|
London School Board requires per scholar |
130 |
„ |
|
English Educational Code per scholar (minimum), in old schools |
80 |
„ |
|
Do., in new schools |
120 |
„ |
Floor-space has an important bearing on ventilation. In
calculating the available cubic space of a room, the height over 12
feet should be disregarded. Thus, if 500 cubic feet is allowed for
each individual, the floor-space should be 42 square feet. In
barracks, soldiers are allowed 50 square feet of floor-space.
In the Government regulations for workhouses it is stated that
there must not be more than two rows of beds, and that the height
of rooms above 12 feet must not be reckoned. This gives a
minimum floor-space of 25 square feet per occupant, or with
dormitories 17 feet wide, a bed-space of about 3 feet.
In hospitals, the question of floor-space is extremely important,
as it regulates the distance between the sick inmates and the
convenience of nursing. Assuming each bed to be 3 feet wide and
6½ feet long, the distance between any two beds should be at least
5 feet. This makes the wall-space for each bed 8 feet long, and
allows from 80 to 96 square feet of floor-space per bed. At St.
Thomas’s Hospital, London, the floor-space is 112 square feet, and
in fever hospitals it is from 150 to 300 square feet per bed. In
regard to the ventilation of hospitals, it has been well said that
nothing less than too much is enough.
Means of ascertaining Cubic Space.—Circumference of a circle =
Diameter (D) × 3.1416.
- Area of circle = D2 × .7854.
- Area of square = square of one of its sides.
- Area of rectangle = product of two adjacent sides.
- Area of triangle = base × ½ height, or height × ½ base.
- Area of ellipse = product of the two diameters × ·7854.
- Circumference of ellipse = half the sum of the two diameters × 3·1416.
- Area of any polygon found by dividing into triangles, and taking the sum of
their areas.
- Cubic capacity of a cube found by multiplying the three dimensions together.
- Cubic capacity of a cylinder = area of base × height.
- Cubic capacity of a cone or pyramid = area of base × 1 ∕ 3 height.
144
- Cubic capacity of a dome = area of base (circle) x 2 ∕ 3 height.
- Cubic capacity of a sphere = D3 x ·5236.
- Area of segment of a circle found by adding to ⅔ of product of chord and
height, the cube of the height divided by twice the chord.
(Ch x H x ⅔) + H3 ∕ (2 Ch)
Give the dimensions of a circular ward for 12 patients, each to have 1,750 cubic
feet of available air-space.
Capacity of ward = 1,750 X 12 = 21,000 cubic feet.
If we allow 120 square feet floor-space for each patient, then the total
floor-space will be 1,440 square feet. Consequently the height of the ward
= 21000 ∕ 1440 = 14·75 feet.
- Area of circle = D2 x ·7854.
- 1,440 ∕ ·7854 = D2.
- Therefore D = 43·2 feet.
- Circumference of circle = D x 3·1416.
- = 43·2 x 3·1416.
- = 135·7 feet.
The dimensions of the circular ward required are therefore a height of
14·75 feet, diameter of 43·2 feet, and circumference of 135·7 feet.
Find the cubic capacity of a circular hospital ward 28 feet in diameter, 10 feet high,
and with a dome-shaped roof 5 feet high.
- Area of floor-space = D2 x ·7854.
- = 614·8 square feet.
- Cubic capacity of the cylinder below the dome is 614·8 x 10 = 6,148 cubic feet.
- Cubic capacity of dome = 614·8 x ⅔ x 5 = 2049·3 cubic feet.
- Total cubic capacity of the ward = 8197·3 cubic feet.
In practical measurements of rooms, deductions must be made from the
cubic space for the furniture contained in it and for its inmates. About 10 cubic
feet ought to be allowed for each bed and bedding, and 2½ to 4 cubic feet for
each individual. Projecting surfaces must be allowed for by subtraction, and
recesses by addition.
A circular ward with a diameter of 36 feet has a dome-shaped roof, the height of
whose centre is 18 feet. The height to the dome is 12 feet. Find the floor-space and
total cubic contents. How many patients ought the ward to accommodate?
- Area of floor-space = (36)2 x ·7854.
- = 1017·8784 square feet.
- Cubic capacity of cylinder below dome = 1017·87 x 12 = 12214·5 cubic feet.
- Cubic capacity of dome = 1017·87 x ⅔ x 6 = 4071·48 cubic feet.
- Total cubic capacity of the ward = 16285·98 cubic feet.
Assuming that 1,500 cubic feet are required for each patient, then the ward
is large enough for 10 patients. It is well to test this conclusion by calculating
whether sufficient floor-space has been allowed for each patient. The floor-space
has been found to be about 1,018 square feet, which would give 100 feet for each
of 10 patients and more than the minimum standard previously stated.
What number of people should be allowed to
sleep in a dormitory 40 feet long, of which the
accompanying sketch is a section?
The cubic capacity of the quadrilateral
space below the roof = 16 x 9 x 40 = 5,760
cubic feet.
Area of floor = area of base of roof =
40 x 10 = 640 square feet.
- Cubic capacity of roof = 640 x ⅓(13 - 9).
- = 853·3 cubic feet.
- Total cubic capacity of dormitory = 6613·3 cubic feet.
145
If we take the low standard of common lodging houses and allow 350 cubic
feet of space for each inmate, then 18 persons may be allowed to sleep in the
dormitory.
How much space would a man occupy supposing him to weigh 175 lbs.? How
much is usually allowed for a man with his clothes, bed and bedding?
The space occupied by a man is stated by Parkes to be from 2½ to 4 cubic
feet (say 3 for the average). He gives the following rule: The weight of a man
in stones divided by 4 gives the cubic feet he occupies. Thus, a man weighing
175 lbs. would occupy 3⅛ cubic feet of space.
About 10 additional cubic feet must be allowed for clothing, bedding, and
bed for each person.
What size of inlet and outlet aperture should be allowed per head? How large
should each individual inlet be made? If an inlet aperture 100 square inches in area is
divided into four, with apertures of 25 square inches each, what is the loss by friction?
A size of 24 square inches per head for inlet and the same for outlet meets
common conditions.
It is desirable to make each individual inlet not larger than 48 to 60 square
inches in area, i.e. large enough for two or three men; and each outlet not
larger than one square foot, or enough for six men (Parkes). This ensures more
uniform diffusion of the air throughout a room. On the other hand, the loss by
friction is greatly increased by having a number of small openings instead of one
large opening. This loss is inversely to the square roots of the respective areas.
Thus the square root of 100 is 10; the sum of the square roots of the four
apertures of 25 square inches each is 20. The loss by friction is double in the
second case what it was in the undivided opening. It is evident, therefore, that
in order to get as much air through the four openings as through the original
large opening, each must be equal in size to half the original opening.
Why is ventilation more difficult in upper rooms of large houses and in single-storied
houses than in the lower storeys of large houses?
Cold external air being heavier than the internal warm air presses downwards
to the lowest point, and pushes up the warmer air. If there were a
vacuum in the room, air would rush into it with a velocity which, as seen before,
is represented by the formula—
v = √(gs).
Where g = 32, s = height of column of air, which we may take as roughly
5 miles.
From this formula we obtain v = 1,306 feet per second.
It is evident that in such a case the velocity of entry of air into a vacuum
on the ground floor would be greater than into a vacuum on any of the higher
storeys, owing to the greater velocity acquired through the increased action of
gravity.
And the same increased facility of entry of air into lower rooms must hold
good under ordinary circumstances, inasmuch as by Montgolfier’s formula (which
is founded on the fundamental formula v = √2(gs))
v = √2(gh(t-t1) ∕ 492)
h = distance between top of chimney and floor of room in question, and thus
the velocity with which air enters is governed by the difference between the
internal and external temperature, and the height from which the cold air
descends in order to take the place of the air which has escaped.
146
In most houses no special means of ventilation are provided,
windows, doors and fire-places being trusted for ensuring a sufficient
supply of fresh air. These do not suffice in well-built houses,
unless the inhabitants train themselves into enduring the currents of
air necessarily associated with open windows and doors. They are,
however, aided in the majority of houses by the porosity of walls,
by currents of air through crevices of wood-work, and so on. It is
desirable that adequate special provision for ventilation should be
made for every house when it is built, and that as much care and
forethought should be exercised in this respect as in the laying on
of a water-supply or sanitary appliances connected with drainage.
Whatever the system of ventilation adopted, it is wise to flush
rooms frequently with fresh air. This is best effected by throwing the
windows wide open whenever a room is left unoccupied. In this
way a much more thorough and complete purification is effected
than by any other means. This is especially important in the case
of bedrooms, in which organic impurities are most prone to
accumulate.
Not only should rooms be ventilated, but likewise the furniture
they contain. This again is most important for bedrooms. Beds
should not be “made” till sometime after using; and in the
interval, should be freely exposed to the air. The same applies to
night apparel.
It is well to allow rooms to lie fallow at intervals. Organic
matter accumulates about a room, and devitalises any air which
enters. If the room is vacated, and flushed with air for a continuous
period, it becomes sweeter and purer. The importance of
this is now well recognised in the case of hospital wards. Such
temporary disuse of rooms must not, however, be regarded as
sufficient without thorough cleansing of every surface in them, in
order effectively to remove all organic and other dust.
An Inlet and Outlet for air should both be provided. According
to some an inlet only is required, while others would only
provide an outlet; but a perfect system of ventilation requires both.
As heated air expands, the outlets should theoretically be larger
than the inlets; but as the average difference of temperature is only
10°-15° Fahr., the expansion is only slight, and may be practically
neglected.
147
The necessity for both inlets and outlets may be illustrated by a single
apparatus like that shown in Fig. 11. A taper is burning at the bottom of
the jar, in the stopper of which two tubes, A and B, are
placed. So long as both tubes are kept open the candle will
keep alight, but if A be blocked, the candle goes out.
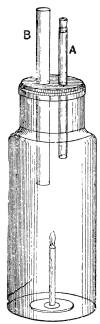
Fig. 11.
Illustrating Necessity of
Inlet and
Outlet.
Inlets should bring air from a pure source,
and should be arranged at intervals in large rooms.
Externally, inlets should be protected from the
wind; and the shorter the inlet tubes the better, as
thus a current is ensured, and they can be easily
cleaned. The position of inlets should not be too
near the outlets, otherwise the fresh air may escape
immediately. The best position for inlets is at the
floor, but this necessitates warming the entering air,
as otherwise it would be intolerable, except in
summer time. If the air cannot be warmed, it
should he admitted about seven feet above the floor,
and directed upwards. For size of inlets, see page
142.
Outlets, under ordinary circumstances, are best
placed near the ceiling. They should be enclosed
as far as possible within walls, so as to prevent the
outgoing air being cooled; and should have smooth
walls, reducing friction to a minimum. Where
artificial warmth increases the temperature of the
air, the discharge of outlets is much more certain and
constant. The chimney with an open fire forms one of the best
outlets. Gas, again, may be made to heat an outlet tube, which
carries off the products of combustion.
Two forms of ventilation are usually described—natural and
artificial. The former term is used to describe any plan not
requiring heating apparatus or the motive power of steam, or gas,
or electricity, while the latter implies the use of some such motive
power or source of heat. Obviously, however, there is no sharp
line of demarcation between the two. A lighted fire is strictly an
artificial plan of ventilation, but inasmuch as no apparatus intended
for ventilating purposes is required, it is hardly a means of artificial
ventilation.
Natural Ventilation.—The most important means of natural
ventilation are the window and the chimney; but openings in outer
walls and over the door may form valuable adjuncts.
The Window is perhaps the most important agent in purifying
a room—both the light and air it admits being essential for health.
The window is invaluable (1) for flushing the room with fresh air
at intervals. Where possible, opposite windows should be opened,
or window and door. Cross-ventilation by opposite windows open
at the top forms one of the best means of natural ventilation,
in large rooms, such as school-rooms. This can, as a rule, be borne
without discomfort, while the room is occupied, unless the wind is
very high.
(2) The Upper Segment of a window may be made to work148
inwards on a hinge, and turned so that the current of air may be
upwards. Where this plan is adopted, triangular pieces of glass
should be placed at the two sides to prevent cold air from falling
directly down at the sides of the opening.
(3) A Block of Wood, two or three inches wide, may be
inserted at the bottom of the window sash at A (Fig. 12), and then
the window pulled down on this. The consequence is that air is
admitted between the two sashes at B, its current being necessarily
directed upwards (Fig. 12). This plan answers admirably in
admitting pure air; but it possesses a disadvantage common to all
the plans in which external air much colder than the internal is
admitted into a room. The current of cold air passes upwards for
some distance, but may then fall down on the heads of those
occupying the room.
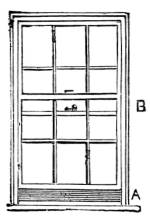
Fig. 12.
Window Ventilation.
(4) The top sash of the window may be opened,
and some zinc gauze fastened across the open part.
This is practically the same as the last arrangement,
except that the air is admitted through the apertures
of the zinc, and the amount admitted is
greatly diminished (page 136).
(5) In Louvre Ventilators, a number of
parallel pieces of glass, each directed upwards, are
substituted for a pane of glass. They may be fixed
or made movable, as in Moore’s ventilator. The
incoming current of air may be similarly directed
upwards, in an open window, by arranging Venetian
blinds with the laths inclined upwards.
(6) In windows that will not open, Cooper’s Ventilators are
often used. Each of these consists of a circular disc of glass,
having five oval apertures in it, which works on a pivot through its
centre, close in front of one of the panes of a window, which has
five similar holes pierced in it. Consequently, when the disc is
turned, so that its holes are opposite those of the window, fresh air
is admitted. The amount thus admitted is necessarily small.
The Chimney forms the best means of escape of foul air. No
room ought to be built without a fire-place, which should never
subsequently be boarded up. In bedrooms the chimney forms a
most important means of ventilation. If there is no fire, the
chimney occasionally furnishes an undesirable source of air; but as
a rule the current is upwards, owing to the aspirating action of
winds at the top of the chimney. The downfall of air from a
chimney chiefly occurs when there is an insufficient inlet for pure
air. This is the explanation of smoky chimneys in nine cases out
of ten; then the cure is easy by laying on a pipe from the outside
of the house to the hearth. When the smoky chimney is due to the
contiguity of higher buildings, the chimney must be raised, or a
cowl placed over it.
(1) The action of the chimney in carrying impure air away
from the room may be considerably increased by narrowing the
two ends, so as to produce a more rapid current at the entrance
and exit of air.
149
(2) The heat of the chimney may be utilised by having a
separate smaller flue alongside it, with openings from the rooms
on each floor. The air in this being heated aspirates the air from
each room in succession.
Openings may be made into the chimney-flue at a higher point
than the fire-place. These are very valuable for carrying off the
heated and impure air resulting from the combustion of gas, as well
as for carrying off the respiratory products, which, in their warmed
condition, tend to rise towards the ceiling.
(3) Dr. Neil Arnott first devised a valve for this purpose.
An opening being made through the upper part of the wall into the
chimney, an iron box was inserted, in which was placed a light
metal valve capable of swinging towards the chimney flue, but not
towards the room. The objections to this apparatus are that it is
apt to make irregular clicking noises, and to admit blacks from the
chimney when out of order.
(3) In Boyle’s Valve these objections are partially obviated.
It consists of an iron frame, across which lie iron rods; and from
these are suspended thin talc plates, only capable of moving in the
direction of the chimney (Fig. 13). Even this apparatus is rather
noisy when there is a strong wind.
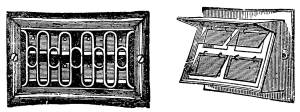
View from room.View from chimney.
Fig. 13.
Boyle’s Mica Flap Ventilator.
Neither of these plans answers so well as a second flue alongside
the chimney flue, communicating
with each
room near its ceiling; but
the latter can only be
arranged for when the
house is built, while the
valves may be inserted at
any time.
The Ceiling may be
utilised for removing foul
air; and thus serve to
diminish the draught which is often produced by the currents of air
towards the chimney, when this forms the only means of outlet.
In large rooms (1) a sunlight gas-burner forms an important
means of ventilation. It causes a strong up-current from every
part of the room. If there is a fire in the room, the burner is apt
to become an inlet for air, or the chimney to smoke, according to
the relative strength of the two currents.
(2) Benham’s and other forms of Ventilating Gas Burners
serve the same purpose. In each of them the products of combustion
are conveyed by special ducts above the ceiling to the
outer air.
(3) McKinnell’s Ventilator is useful in single-storied buildings,
like certain barracks. It consists of two tubes encircling one
another, the inner forming an outlet tube, because the casing of the
outer tube maintains the temperature of the air in it. It is made
higher than the outer tube, and is protected by a hood. The
outer tube forms the inlet for fresh air. The entering air is thrown150
up towards the ceiling and then to the walls by a flange placed at
the bottom of the inner tube. The air after traversing the room,
and becoming heated, passes upwards to the inner tube. When
doors and windows are open, both
tubes become outlets; if there is
a fire in the room, they may both
become inlets; but this may be
prevented by closing the outlet
tube.
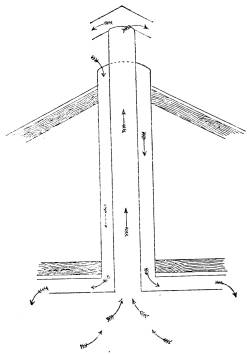
Fig. 14.
McKinnell’s Roof Ventilator.
(4) Various other means
have been devised for carrying
foul air from the ceiling through
channels between the ceiling and
the floor of the room above. All
share the disadvantage that the
channels become dirty and are
difficult or impossible of access
for cleaning.
(5) Various cowls connected
by metal tubes with the ceilings
of rooms have been placed on
roofs, and their aspirating effect
used in ventilating these rooms.
When a room is furnished with a
chimney such cowls are most
undesirable. In large rooms
without a fire-place they are helpful, but much more confidence can
be placed in cross-ventilation by hinged windows. It is doubtful if
any of the advertised fixed cowls produce materially greater
aspiration of air from rooms than a simple open tube of the same
size. It is desirable that the tube should be protected at its upper
end against the entry of rain, and that a grating should be provided
to prevent birds building their nests in the tube.
In the preceding plans of ventilation, the ceiling serves almost
entirely as an outlet for impure air. In the following plan, it is
used as an inlet for pure air.
(6) In Sylvester’s Method of Ventilation, the perflating
force of the wind is employed to produce an abundant entry of fresh
air. A cowl is placed, always turning towards the wind; the air
received is conducted to the basement, where it is warmed by a
stove or hot-water pipes, and then passed through tubes into the
upper rooms. From these it is carried by tubes above the roof,
these tubes being covered with cowls turning from the wind, so
that in this way the aspirating power of the wind is likewise used.
Ships are often ventilated in a somewhat similar manner. The
tube to which a windward cowl is attached above, ought to be bent
at right angles, so as to lessen the velocity of the entering air. By
covering other air-shafts with movable cowls, turning from the wind,
the aspirating action of the wind is brought into action to aid the
escape of foul air.
151
The Walls of a room, unless covered with an impervious
material, are constantly traversed by gentle currents of air, which
play an important part in the ventilation of rooms. Special
apertures may be made to furnish a freer supply, and these may be
in various forms.
(1) A Simple Grating, may be inserted; but this is apt to
become blocked with dirt, and does not allow a large amount of air
to enter. Louvred openings in the walls are objectionable, except
for very large rooms.
(2) Sheringham’s Valve is the most convenient means of
ventilating through the wall. An
opening in the external wall is made
by a ventilating brick or grating;
into the wall is fixed an iron box,
which has in front of it an iron valve
hinged along its lower edge, so that
it can open towards the room. On
the sides of the valve cheeks are
attached, which fit into the box when
the valve is shut. A heavy piece of iron pressing against the valve
from within the box, tends to keep it constantly open. By means
of a string and pulley, the valve can be opened or closed at will,
or fixed in any intermediate position.
Fig 15.
Sheringham’s Ventilator
In a very large room, it is better to have several medium-sized
valves, than a few larger ones, the air being thus more completely
diffused. If there are two valves, they should not be opposite one
another, as the air may then simply pass from one to the other,
without becoming diffused through the room. If there is only one
valve, it may occasionally serve as an outlet when the wind is to
leeward. By means of this form of valve, the air is projected
upwards in a diverging current towards the ceiling. The valve
should be placed above the level of one’s head, but not too near the
ceiling; as in the latter case, the current of air is driven hard
against the ceiling, and falls thence with considerable force towards
the floor. A combination of Sheringham’s inlet and Boyle’s mica
outlet into the chimney at the opposite side of the room ensures
efficient ventilation in a dining-room. Better than the outlet into
the chimney is an opening into a special flue alongside the chimney-flue,
if this be available.
(4) Ellison’s Inlet consists of a brick pierced with conical
holes, the apex of the cone being towards the external air. By this
means any great draught is avoided, and the air is distributed over
a considerable area. In order that this may prove an efficient
means of ventilation, a considerable number of bricks are required.
The Floor of a room is always the source of considerable
currents of air, even when well carpeted. Air mounts up through
the crevices of the wood-work, being aspirated into the room when
its temperature is higher than that of the rooms below. In the
case of rooms on the ground floor, air is often drawn from the
subjacent soil, or through dust-bins, etc.
152
Theoretically, in all measures of ventilation, the floor would
be the best point for the entry of cold air. This, however, is
intolerable when the incoming air is cold, and the floor must
therefore be abandoned as a means of ventilation, apart from
heating apparatus.
The floor may be used as a means of entry of fresh air in a
modified manner, by directing the air entering at the floor-level for
some distance up a tube at the side of the wall. This apparatus is
known as Tobin’s tube. It consists of a rectangular or cylindrical
tube from 4 to 6 feet high, which communicates at the lowest point
with the external air by means of a perforated brick or grating.
The air enters the room in an upward direction, and is consequently
sent towards the ceiling, where it becomes mixed with warmer air,
before diffusing itself throughout the room. But when the incoming
air is very cold, it may fall more rapidly, causing cold draughts on
the heads of those in the room.
As the air enters directly from outside the house, it often carries
with it particles of dirt, soot, etc. This may be remedied by
placing a pan containing a shallow layer of water at the lowest
part of the tube, or by placing cotton wool at the point of entry of the
tube into the room. The tray of water soon dries up and is rarely
replaced, while the cotton wool diminishes the amount of entering
air. It is very useful however in cold weather, or when fogs occur.
A gauze funnel is sometimes inserted in the tube, or a sheet of gauze
arranged diagonally across the tube from its highest to its lowest
point. The gauze does not keep out minuter particles of dust, and
requires occasional cleaning. All Tobin’s tubes, like other ventilating
openings, should be made to open, so that their interior can be
frequently cleaned.
Summary as to Domestic Ventilation.—Open windows, doors, and
fire-places may be in most instances trusted. If gas is used as an
illuminant, they should be combined with special arrangements
for carrying off the products of combustion from the room. For
delicate people, and especially in small rooms, outlet ventilation
into the chimney breast combined with a Sheringham’s valve on the
opposite wall is desirable.
Artificial Ventilation.—Artificial ventilation may include
two important and very different measures. In one of them
currents of air and an exchange of pure for impure air are effected
by means of various forms of heating apparatus. In the other
mechanical measures are used for the same purpose,—the air being
either driven out of the room or drawn out of it. In this chapter
we shall consider only the mechanical means of artificial
ventilation. There are two kinds, the first being known as
ventilation by aspiration, or the vacuum system; and the second
as ventilation by propulsion, or the plenum system.
In Ventilation by Aspiration the foul air is drawn out of
the room by machinery, its place being supplied by fresh air, which
may be warmed before entry or not. This plan and the next have
been employed chiefly in connection with large buildings, such as
hospitals, etc., and in mines.
153
The extraction of foul air may be effected by—(1) a steam-jet,
which is allowed to pass into a chimney, and sets in motion a body
of air more than 200 times its own bulk. Tubes from each room
of the building are connected with this chimney, and the strong
upward current extracts the air from them. This plan is useful
in factories, where there is a superfluous supply of steam.
(2) A fan or screw may also be used. The vanes of the fan,
when set in motion by electrical or some other motive power,
produce a powerful current of air, which can be regulated according
to requirements. As in the last plan, the aspirating influence of
the fan may be exerted over a system of rooms, by means of
connecting tubes.
In Ventilation by Propulsion a fan is used as in the last
plan, the air being propelled along conduits leading from it into the
room to be ventilated. The size of the conduits being known, the
amount of air to be discharged can be regulated by timing the
rapidity of the revolutions of the fan.
This plan is suitable for crowded places, where a large amount
of air is required in a short time. It is excellent for large schools,
churches, and theatres. Its superiority for large elementary schools
has been proved at Dundee by the experiments of Drs. Carnelley,
Haldane, and Anderson, the results of which are summarised in the
following table:—
|
NO. OF SCHOOLS. |
NO. OF ROOMS. |
CUBIC FT. ALLOWED PER PERSON. |
CARBONIC ACID IN 10,000 OF AIR. |
MICRO-ORGANISMS PER LITRE |
| BACTERIA. |
MOULDS. |
| Mechanical ventilation by warmed air |
6 |
32 |
160 |
12·3 |
17·5 |
1·0 |
| Natural ventilation andhot pipes |
17 |
43 |
176 |
16·3 |
96·5 |
1·1 |
| Natural ventilation and open fires |
33 |
84 |
145 |
19·2 |
153·2 |
4·8 |
The air to be admitted may be warmed by passing it over hot-water
or steam-pipes. In large establishments, as in hospitals,
theatres, etc., it has been arranged so that the incoming air is
passed through a screen of coarse cloth, which is kept wet by water
trickling down each cord. The air is thus kept moist and freed
from dust.
The great advantage of the plan of propulsion, is its certainty.
By it the temperature, moisture, and freedom from suspended
matters of the incoming air can be exactly regulated and controlled.
Its chief disadvantages are that (1) it is somewhat costly, and (2)
the apparatus requires skilled supervision. On the other hand it
maintains the air in crowded rooms in a condition which cannot be
secured by any other method. When combined, as is done in the
Houses of Parliament, with the use of a flue for the extraction of
foul air, this plan answers admirably.
154
The Relative Value of Artificial and Natural Ventilation
scarcely needs to be discussed. They are both valuable, but under
different circumstances. In dwelling-rooms natural ventilation by
doors, windows and chimney usually suffices, especially if the
products of combustion of gas are removed through a special flue.
Natural ventilation is always occurring, and only needs a little aid
in domestic life. For large rooms occupied by many persons
artificial ventilation is necessary to maintain pure air.
Whatever method of ventilation is adopted, the atmosphere
will remain to some extent polluted, if the room and its occupants
are dirty. In certain experiments made by Carnelley in schools,
it was found that dirty children increased the number of micro-organisms
per litre of air more rapidly than dirty rooms. Thus:—
| DEGREE OF CLEANLINESS OF |
CLEAN. |
MEDIUM. |
DIRTY. |
| Children |
63 |
99 |
159 |
| Rooms |
85 |
94 |
139 |
Number of micro-organisms per litre of air.
Hence cleanliness of rooms and of their occupants is quite as
important as a good system of ventilation.
155
CHAPTER XXIII.
VENTILATION BY THE INTRODUCTION OF
WARMED AIR.
Ventilation by the Burning of Coal. In winter and at any
time of the year when the out-door temperature is below 50° Fahr.,
the warming and ventilation of a room are necessarily combined.
If air is admitted unwarmed it will produce draughts, unless
directed upwards by Tobin’s tubes or otherwise. In dwelling-rooms
such contrivances may suffice; but in any larger building,
in order to ensure sufficient ventilation, it is necessary to warm
the incoming air.
The Open Fire-place forms the most common means of
ventilation by heat (see also page 159). The ascent of warm air up
the chimney, causes cold air to rush along the floor to the fire-place
from all parts of the room, especially the door. Part of the air
thus approaching the fire is carried up the chimney with the smoke,
while the remainder, after having been warmed, flows upwards
towards the ceiling near the chimney-breast. It passes along the
ceiling, and cooling in its progress towards the opposite wall,
descends, and is again drawn towards the fire-place. Thus there
is a continuous circulation of the air in a room.
In the experiments of the Barrack Commissioners (1861), it
was found that the amount of air passing up the chimney while a
fire was lit, ranged from 5,300 to 16,000 cubic feet per hour, the
mean of 25 experiments being 9,904 cubic feet. We may conclude,
then, that with an ordinary grate, a chimney provides outlet for
impure air sufficient for four or five persons. Its lack of economy
as a heat-producer will be considered later. Its efficiency as a
ventilator within the above limits is evident.
When a fire is burning in the grate, all other openings in the
room, except openings into the chimney, serve as inlets. If the
room is insufficiently supplied with openings, a double current may
be established in the chimney, with the result that occasional
down-puffs of smoke occur.
As a rule the chimney serves only as an outlet for impure air.
It may by appropriate means be made to serve as an inlet for pure
and warmed air, the heat which would otherwise escape up the
chimney being utilised for this purpose. Galton’s stove is one of
the best for this purpose. At the back of this stove is an air-chamber,
communicating with the external air, and in which the
fresh air is heated before it enters the room. On the back of the156
stove broad iron flanges are cast, in order to present as large a
heating surface as possible. They project backwards into the air
chamber; and their heating surface is aided by the iron smoke-flue,
which passes through the air-chamber. The warmed fresh air enters
the room by a louvred opening above the mantel-piece, or by an
opening in each side of the chimney breast. By this stove one-third
of the total heat of the fire is utilised, as against one-eighth in
an ordinary fire-place.
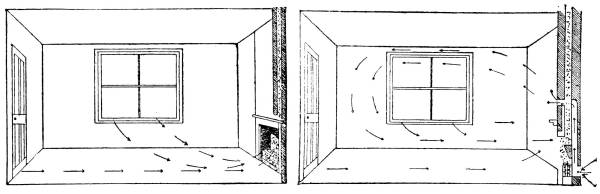
Fig. 16.
Vertical Section through Two Rooms, showing—A. Currents of cold air with an
ordinary fire; B. Direction of currents of warmed fresh air with a Galton’s
Ventilating Stove.
Shorland’s Manchester and other stoves are constructed on the
same principle as Galton’s.
The Ventilation of Mines is effected by lighting a fire at the bottom
of a shaft. The air for the combustion comes down another shaft (the intake
shaft), or down another half of the same shaft separated by a partition. The
consequence is that constant up and down currents of air are produced. The
air from the intake shaft is made to traverse the galleries of the mines, its
course being directed by partitions, before it is allowed to reach the fire and s
be carried up out of the mine.
In addition to, or instead of, an ordinary coal-fire, the power
for extracting impure air may be obtained from Hot Water or
Steam Pipes. There are various plans founded on this principle.
When hot-water pipes are used for baths, etc., they may also
be utilised for ventilation, in two ways:—1st. The hot-water pipe
may be made to coil round the tube by which fresh air is admitted
into a room, thus warming the air as it enters. 2nd. The hot-water
pipe in its course upwards may be enclosed in a shaft, which opens
into the external air above. The air in this shaft being heated, the
impure air may be collected and removed from the different rooms
by tubes connected with it. Thus, a hot-water apparatus, when
well arranged and complete, may furnish pure warm air, and carry
away impure air. The ventilation by this plan is found in practice
to be somewhat irregular.
The plan proposed by Drs. Drysdale and Hayward of Liverpool is similar
in principle:—Fresh air is warmed by a coil of hot-water pipes in the basement,
and is admitted into the staircase and landings, when it is supplied to the
different rooms by openings provided with valves. From the rooms, special
outlets converge to a foul-air chamber under the roof. This is connected with a
shaft leading from the kitchen-fire, the latter, therefore, acting as an extraction
furnace.
157
Lighted Gas may be employed to produce a current for
ventilating purposes, as well as fire or hot-water.
Sunlight and Benham’s Ventilating Gas-burners, have
already been mentioned in this connection (page 149). They are
extremely valuable means of ventilation, producing powerful
currents of air from all quarters of the room unless they are
specially enclosed.
In theatres and similar buildings the Chandeliers may be
made to extract vitiated air. Where a number of chandeliers exist,
they may be connected by tubes with a main shaft, and all made to
contribute to the same object. According to the experiments of
General Morin, the discharge of 1,000 cubic feet of air is produced
by the combustion of one cubic foot of gas.
Various forms of gas-stoves are now sold, which act as ventilators
as well as sources of heat. Among these is George’s
Calorigen Stove (Fig. 17). It can be obtained in various forms
suitable for burning coal-gas, or coal, or
oil. Within its outer case is contained
a special iron tube, which communicates
at its lower end with the outer air, and
opens at its upper end into the room.
The heat generated in the stove warms
the air in the spiral tube, which accordingly
ascends into the room. The ascent
of warm air causes a draught from below,
and the consequence is, that so long as
the combustion is going on, a current of
warm air continues to ascend into the
room. The products of combustion are
carried out of the room by the pipe F.
This stove is free from most of the objections
appertaining to gas-stoves; it can
be fixed into an ordinary fire-place, and
made to keep the temperature of a room
uniform.
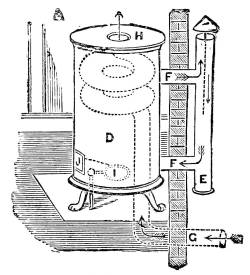
Fig 17.
George’s Calorigen Stove.
- A—The interior of the room.
- B—Exterior of building.
- C—Wall.
- D—The Calorigen.
- E—A cylinder.
- FF—Pipes communicating
with stove and cylinder to
supply air for combustion,
and to carry off the products
of combustion.
- G—Pipe for passage of
fresh cold air to Calorigen.
Can be carried above the
floor between the joists, as
may be more convenient.
- H—Outlet for air into the
apartment after being made
warm.
Bond’s Euthermic Stove is similarly
constructed to the above, but is
open below so that the air needed for
the gas combustion is drawn from the
interior of the room, and the continuous
change of air is thus favoured.
Objections to Ventilation by
Heating Apparatus.—When warmed air
is admitted into a room, it is very apt
to be dry and irritating. This can be
usually avoided by having water standing
in the room, so as to allow evaporation. A more difficult problem
is to ensure the complete absence of all products of combustion,
particularly of the products of incomplete combustion.
158
Physiological and Physical Considerations.—The warmth of
our bodies is naturally kept up by the oxidation changes constantly
going on in the system. In Chapter XL., p. 265, are discussed the
modes in which heat is lost by the system, and the influence of
clothing in controlling the amount of this loss. Artificial warming
of houses has a similar action to clothing. It diminishes the
demand on the system, and so economises the amount of food
required.
The degree to which this diminution of loss of heat by clothing
and artificial warming of houses may be carried varies with
circumstances. There can be no doubt that if food be abundant,
exposure to external cold, if not too extreme, is on the whole
beneficial, for vigorous people. But for old people and young
children, means of artificial warmth require to be more carefully
provided. Severe cold is for them often the harbinger of death.
The Degree of Temperature at which living-rooms should be
kept will vary with circumstances.
For healthy adults, any temperature between 50° and 60° Fahr.,
will be moderately comfortable; for delicate children and old people it
may be 65° with advantage.
For sick rooms and hospitals the temperature of 60° is usually
adopted, but this is by no means always necessary. A temperature
of the room as low as 50°, except for such diseases as whooping
cough and bronchitis, suffices if the patient is well covered with
warm personal and bed coverings.
Convalescents from any acute illness bear low temperatures badly.
The Different Kinds of Heat.—Heat may be communicated
by radiation, conduction, and convection. By radiation of heat
is meant the process by which heat passes from a fire or other
source of heat, through a vacuum, dry air or any other medium,
without heating any of the media through which it passes, but only
the bodies against which it finally impinges. The solid bodies
(including ourselves) which are warmed by radiant heat, by a
process of conduction then warm the surrounding air. This method
is the nearest imitation of the natural warmth of the sun.
Conduction of Heat is the passage of heat from one particle
to another, whether it be of a gas or solid. It is an extremely slow
process when air is concerned, and may be practically ignored.
159
Convection of Heat is the process by which a gas or liquid
actually carries the heat in itself from one part to another. The
heated particles are relatively lighter, and ascend to the higher parts
of a room, while colder and heavier particles descend, and are
subjected to the same process. Heat can be carried by convection
only by gases and liquids. It is quite possible, therefore, for a
person to be cold in a room filled with warm air, if the walls, etc.,
are cold; and on the other hand, to feel comparatively warm in a
room filled with cold air, if more heat is radiated from an open fire-place
or the warm walls to his body than he radiates to his
surroundings. The feeling of “draught” when sitting near a wall
is sometimes caused by radiation of heat from the body to the colder
wall. The ideal arrangement, were it practicable, would be to have
cool air to breathe, but to be surrounded by warm walls, floors, and
furniture. A room warmed by an open fire is more comfortable
than a room warmed by hot air from a furnace, assuming the
temperature of the air is the same in both instances, because the
walls of the room are several degrees lower in temperature in the
latter than in the former. For warming walls as well as the air
high pressure steam pipes are more efficient than hot-water pipes.
The great advantages of radiant heat are that—(1) it heats the
body without appreciably heating the air; while at the same time
(2) there is no possibility of impure gases being added to the air.
It has, however, considerable disadvantages. (1) It is costly,
though its expense may be greatly diminished by a well-constructed
fire-place. (2) It only acts on bodies near it to any useful extent.
Its effect lessens as the square of the distance; thus, its warming
effect at five feet distance, is twenty-five times less than at a
distance of one foot. It is evident, therefore, that for long rooms,
and for large assembly-rooms, a single source of radiant heat is
quite inadequate. The immense loss of heat in our ordinary fire-places
is slowly leading to their modification; and although it is
probable that radiant heat will always be the favourite source of
warmth in dwelling-houses, it will be used for larger buildings chiefly
as an adjunct to convection of heat.
The different sources of heat are employed, either singly or
combined, in the following methods of warming our dwellings and
other buildings:—
- 1. Warming by the open grate.
- 2. Warming by closed stoves.
- 3. Warming by hot-water pipes.
- 4. Warming by steam in pipes.
- 5. Warming by hot air.
- 6. Warming by electricity.
Warming by the open Grate.—In the open fire-place
radiation is the source of heat chiefly employed.
The position of the fire-place is important. It should not be
on the external wall of the house, as thus a large proportion of heat
is lost; but should be placed where the heat from the flue may be
utilised in keeping up the temperature of the house.
160
The construction of a fire-place is commonly faulty in several
respects. (1) The fire-place may be too far included in the wall, so
that the heat at once passes up the chimney. (2) It may be
composed chiefly of iron, which rapidly conducts away the heat,
and does not furnish a surface for radiation. (3) The bars and
bottom of the grate may be so arranged, that coal and cinders fall
out in an incompletely burnt condition.
It has been estimated that with an ordinary fire-place, seven-eighths
of the possible heat is lost, one-half being carried up the
chimney with the smoke, one-quarter carried off in the ascending
current of warm air, and one-eighth of the combustible matter
remaining unconsumed, forming the solid matter of the smoke.
The defects which have been indicated may be remedied by
bringing the fire-place rather further out into the room; by
substituting fire-brick for iron behind and at the sides of the fire,
and by having a layer of fire-brick at the bottom of the grate, or the
grate lowered, so that as in Teale’s stove, it lies on a bed of fire-brick
at or below the floor level.
The shape of the grate is important. The width of the back
of the grate should be about one-third that of the front, the sides
sloping out towards the front of the recess. The depth of the grate
from before backwards should be equal to the width of the back.
The sides and back of the fire-place must be made of fire-brick, thus
ensuring the heat being retained in the grate. And finally, the
chimney throat must be contracted so as to ensure more complete
combustion. The chief objections to an open fire-place are (1)
the great waste of fuel involved, even after the improvements
indicated have been carried out. (2) The unequal heating at
different distances from the fire. (3) The smoke and dust always
produced to some extent, from accidental smoking of the fire, or
from the escape of ashes. (4) The trouble involved in frequently
replenishing the fire. (5) The cold draughts produced by the
currents of air towards the chimney. These travel chiefly along the
floor, when, as is commonly the case, the space between the bottom
of the door and the floor forms the chief place for the entry of fresh
air.
Many patents have been brought out for the introduction of
the fuel at the lowest part of the fire. The uppermost part of
the fuel being first burnt, and the remainder attacked from above,
the smoke is consumed in passing through the red part of the fire.
Thus a comparatively smokeless fire is produced, and the amount of
heat evolved is greatly increased. So far none of these have been
altogether satisfactory. The production of a comparatively smokeless
fire is a great boon. Smoke means so much unburnt fuel, and not
only so but the sooty particles float about in the atmosphere,
rendering it impure, and changing comparatively harmless mists
into town fogs, which are loaded with soot and the products of
combustion, and do incalculable mischief to health and property.
The prevention of this smoke nuisance demands more consideration
than it has yet received. The Public Health Acts constitute the
emission of black smoke from the chimneys of manufacturing161
premises a nuisance; and manufacturers can if they use proper
boilers, especially those in which mechanical stokers are employed,
almost completely obviate this nuisance. The great principle is to
prevent the escape of smoke before it is completely burnt.
This may be accomplished by careful stoking, by keeping the
unburnt coal at the front of the fire, and by ridges exposing the
smoke to red-hot fire-clay before it escapes. In domestic fires, gas
is gradually replacing coal for cooking, with a corresponding
reduction of the smoke-nuisance.
The Utilization of the Heat Produced in the fire-place to
warm the air on its way into the room, as in Galton’s and other
similar stoves has been already described (page 156).
A larger amount of heat can be obtained out of a given
quantity of fuel by cutting off some of the cold air, which rushes
through the fire, and carries the half-burnt gases and much of the
heat up the chimney. This is effected by having a solid fire-brick
bottom to the grate, or by closing up the front of the open chamber
under the grate, by means of a close-fitting shield or door. These
“Economisers,” as Mr. Teale calls them, appear to answer better
than solid fire-brick bottoms, as they do not prevent the ashes
falling under the grate.
The Fuel burnt in an open fire-place may be either coal or
coal-gas. Occasionally coke is also employed. Coke and coal-gas
have the advantage over coal, that (1) no smoke is produced. Coal-gas
presents the additional advantages, that (2) it can be turned on
at any moment, without having to go through a tedious process of
lighting the fire; and that (3) the amount of heat can be exactly
graduated by regulating the supply of gas. A gas fire is however,
as a rule, more expensive than a coal fire.
Open Gas-stoves are made in various forms. In the common
one, small jets of gas are lit under the grate, which is filled with
pieces of asbestos. These become red hot, and radiant heat is
emitted. To obtain the greatest value from the heat generated by
the combustion of gas, a stove should be chosen in which the heat
generated is brought into contact with a large surface of the grate
before the products of combustion are allowed to escape to the flue.
Gas stoves which are advertised as not needing a flue, should
be avoided. A large amount of carbonic acid is discharged by them
into the room, and the sulphurous acid also produced by the
combustion of gas is not completely absorbed in the water of
condensation which collects in a tray under such stoves.
Closed Stoves form the most economical and efficient warmers
for rooms of moderate size, and coal, coke, coal-gas or paraffin may
be burnt in them.
The advantages rightly claimed for coal stoves of this type are
that (1) the amount of fuel consumed is small; (2) by adjusting the
damper, combustion may be rendered as slow as desired, so that
but little heat is lost by the flue or chimney; and (3) heat radiates
from all parts of the stove into the room, and not simply from a
small area of fire-front.
162
The chief objections to closed stoves are, that (1) they dry the air
excessively, rendering it somewhat unpleasant. (2) They produce
a peculiar close smell, apparently caused by the charring of minute
particles of organic matter in the air, coming in contact with the
stove. If the air of the room is not heated above 75° Fahr., no
smell is produced, and the relative humidity is not lessened to any
appreciable extent (Parkes). But when the heat produced by the
stove is excessive, these results do follow. The unpleasantness
may be modified though not entirely removed, by placing shallow
pans of water near the stove.
(3) Portions of the products of combustion may pass through
cracks or fissures in the stove, or even through the joints of the
stove. Independently of such accidental cracks, cast-iron stoves,
when red hot, appear to allow gases to pass through them with
comparative ease. Thus carbonic oxide and other gases may find
their way into the room, and it is probable that this rather than the
dryness of the air, is the cause of the unpleasant symptoms sometimes
complained of in rooms where closed stoves are in use. This
escape of carbonic oxide does not occur with earthenware stoves
properly encased with fire-clay.
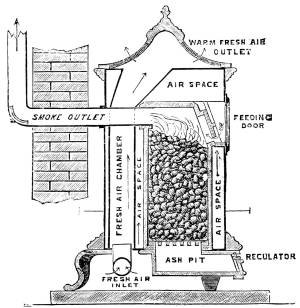
Fig. 18.
Slow Combustion Stove.
Many modifications of the older closed stoves are now in
common use. In the
stove shown in Fig. 18,
excessive heating of the
air is prevented by the
presence of two air
chambers, only the outer
one, which brings external
air to be warmed, having
its air emptied into the
room.
Warming by open
grates or closed stoves is
specially applicable to the
rooms of private houses;
warming by hot air or
steam, or hot water, is
chiefly used for large
buildings. It is quite
possible that these methods
will be applied at
some future time on a
large scale to the warming
of private houses. In some large towns of the United States this
has been already done, blocks of a hundred or more houses being
warmed from the same centre, by the same system.
But apart from such a central system, hot air and hot water
lend themselves to the heating of houses on what may be called the
Whole House System (page 156). We have mentioned in the last
chapter some methods of doing this, and shall now describe others.
Hot-water Pipes are probably the best means of carrying heat163
to various parts of a large house, and hot water is more thoroughly
under control and less dangerous than either hot air or steam.
There are two systems of heating by hot water.
In the first, which we may call the low pressure system,
there is a boiler from which water circulates through pipes to every
part of the building, and as it cools down returns again to the
boiler. At the highest points of the pipes, outlets are provided for
air. In this system the water is not heated above 200° Fahr., and
there is consequently no great pressure on the pipes.
In the high pressure system (Perkin’s patent), the pipes have
an internal diameter of about ½ an inch, and have thick walls made
of two pieces of welded iron. There is no boiler, but one portion of
the tube passes through the fire and the water is heated to
300-350° Fahr., thus subjecting the pipes to great pressure. In
dwelling-houses with the low pressure system, for every 1,000 cubic
feet of space to be warmed to 50°, 12 feet of 4-inch pipe should
be given; with Perkin’s pipes, probably about two-thirds of this
will suffice.
Steam Distributed by Pipes may be employed instead of hot
water. This method has been used in factories in which there is a
surplus supply of steam.
Warming by Hot Air is only applicable on a large scale, and
should only be used in association with a system of ventilation by
propulsion (page 153), in which the temperature, humidity, and
freedom from dust of the entering air are carefully regulated.
Warming by Electricity both for cooking food and for
warming rooms has a large future, but in most districts the supply
of electricity is not hitherto sufficiently cheap to be used for these
purposes. By its means the atmosphere will be prevented from
becoming impure, labour will be reduced, and life rendered more
pleasant.
Hot Water Supplies.—Nearly every modern house is supplied
with a bathroom, and this may be supplied with hot water either
from a geyser or from the kitchen boiler. In a geyser the water is
made to flow over a large heating surface furnished by burning
coal-gas, and with the best varieties a bath of 98° F. can be supplied
in from five to ten minutes. As the bathroom is usually small and
unprovided with an open fire-place, persons have occasionally been
suffocated by remaining in such a room while the gas continues
burning. This is due to the production and in-breathing of carbonic
oxide. No geyser ought to be allowed to be used which is unprovided
with a flue passing into the chimney flue or in its absence
through an external wall of the house. Short of fatal poisoning,
violent headaches often occur when a warm bath obtained by means
of a geyser is taken, unless such a flue is provided. In Ewart’s
lightning geyser, additional protection is furnished by the fact that
a dual valve is so arranged, that immediately the water is turned off
or the supply fails from any cause, the supply of gas is also cut
off.
Hot water supplies from kitchen boilers, unless carefully164
arranged, may be responsible for serious explosions during severe
frosts.
Four plans are in common use. (1) The worm-boiler system. This system is
unsafe unless the supply of water to the boiler is attended to; and as the hot
water supply to the kitchen is drawn from the boiler itself and not from the
worm, the hot water supply for the rest of the house may be deficient. Usually
the small feed cistern for the boiler in this system is too near the boiler to freeze.
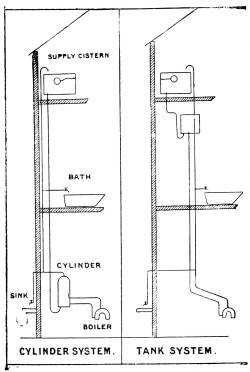
(2) The cylinder system is very effective. In this system a metallic cylinder,
capable of withstanding a pressure of 20 lbs. to the square inch, is placed in the
kitchen or bathroom between the cold and hot supplies, its contained water
being heated by circulation from the boiler, hot water ascending and cold
descending. On the top floor of a house is a cistern from which cold water is
supplied. Both the supply pipe and escape pipe for hot water may become
frozen during frost. Then the supply of water is stopped, and the boiler and
reservoir may boil dry. This would not occur without some indication in
unusually vigorous boiling. Boilers
sometimes explode, and cylinders
sometimes explode. This can be
effectually prevented by (3) A Double-cylinder
apparatus one within another.
In this the water in the outer
cylinder supplied from the main
cistern can only be heated to
212° F., and the water in the boiler
and inner cylinder supplied from a
lower feed cistern can only be heated
to 214° F., on account of the small
head of water. Two escape pipes give
free communication with the atmosphere.
(4) In the tank system, which
being cheap, is usually adopted in
poor houses, the tank is placed high
up in the system. The hot water
branch pipes are usually taken from
the flow-pipe between the boiler and
tank. Hence when the supply fails,
as during frost, the tank is drained
empty, the circulation of water ceases,
and the system is changed from a
circulation system to a high-pressure
one.
Safety valves cannot always be
relied on to prevent explosions. If
they lead to the lighting of fires in
frosty weather, when pipes are frozen, they may cause explosions. Explosions
from frost only occur when both pipes are blocked. Incrustation of the boiler
and pipes increases the danger of explosions; hence the necessity for their
periodical cleaning.
165
The Removal of Impurities.—In order that health may be
maintained in any inhabited house, it is essential that the impurities
produced by animal life should be removed. These impurities may
be divided into two classes—the first including the gaseous and
volatile products evolved from the lungs and skin; and the second,
the liquid excretion from the kidneys, and the solid from the bowels.
The former are got rid of by efficient ventilation and by cleanliness;
the latter, ought to be as quickly removed, but require more elaborate
arrangements to ensure this.
The average daily amount of solid excreta is about 4 ounces,
and of fluid excreta about 50 ounces for each adult male. Taking
all ages and both sexes into consideration, the amount per head is
about 2¾ ounces of fæces and 32 ounces of urine. When dried, the
daily fæces amount to 1·04 oz., the daily urine to 1·74 oz., so that
the manurial value as well as the possible polluting power of urine
is much greater than that of the fæces.
After a variable interval urine and fæces begin to decompose,
ammonia and fœtid gases being disengaged in large quantities.
Urea the chief constituent of urine is decomposed into carbonic acid
and carbonate of ammonia. Thus
CH₄N₂O + 2H₂O = (NH₄)₂CO₃
In addition to the excreta, house-slops have to be got rid of, and
“dust.” House-slops vary greatly in quantity, but probably
amount to as much as sixteen gallons per head daily. They consist
chiefly of the water used in cooking and washing and for baths. It
would be a mistake to suppose that only urine and fæces need
careful disposal. There are masses of decaying epithelium from
lavatories and baths, organic matters from soiled apparel, and
various organic matters from culinary operations, all of which may
cause serious nuisance unless promptly disposed of.
The Dust consists chiefly of the ashes from fires; but the
dust-bin also forms a favourite refuge for kitchen refuse, composed
of various animal and vegetable matters, as well as for broken pots
and tins. It is dealt with apart from the house-slops and excreta,
except in certain dry methods of disposal of sewage.
Two chief plans of getting rid of the sewage have been proposed,
though there are many varieties of these. They are—
- 1. The Water Method, and
- 2. The Dry Methods.
166
For towns the water carriage of sewage is indispensable, and
in this chapter we shall confine ourselves to the part of this system
which relates to the Drainage of the House.
The chief sanitary appliances of a house, which empty their
contents into the drain and thence into the street sewer, are—(1)
Rain-water Pipes; (2) Bath-room and Sink-pipes; (3) Water-closets;
(4) Soil-pipes; and (5) The House Drain. We will
consider these in detail.
Rain-water Pipes collect the water from the roof by means of
gutters, and carry it down to the house drain, except in the few
cases in which the rain-water is collected for use. The rain-water
or stack pipe was formerly joined directly at its base with the
underground drain. This was evidently bad, because the upper
end of the pipe was frequently near windows, and foul gases from
the drain might be conducted by it into the house. It is equally
objectionable to connect the rain-water pipe with the soil pipe and
for the same reasons as above.
The general rule with regard to all pipes carrying away water from
the house, with the sole exception of the soil-pipe, is that they must be disconnected
from the underground drain and discharge into the open air over
a gulley-trap. This rule applies to
- rain-water pipes, and to
- waste pipes from baths,
- lavatories,
- sinks.
It does not apply to the soil-pipes leading from
- water-closets and
- slop-sinks,
Overflow or waste-pipes from cisterns for drinking water or
from cisterns for flushing w.c.’s or to safe trays under the seat of
water-closets should all be made to discharge into the open air,
where the leakage can at once be discovered.
The form of gully-trap to be used at the junction with the drain
is described on page 167.
Other Waste-pipes as from bath, lavatories, and sinks, must
be similarly disconnected from the drain, and made to discharge
over gully-traps. When the pipe leading from the bath, lavatory
basin, or sink, is long, it is apt to become foul from the accumulation
on its inner surface of slimy matter, consisting of soap, dirt, and
other offensive matter. For this reason it is wise to have a syphon
bend in the waste-pipe near its junction with the sink or basin.
Such a trap is shown in Fig. 19.
The syphon bend alone in the waste-pipe without disconnection
from the drain at its lower end would not suffice to ensure complete
absence of nuisance, especially for sinks and lavatories which may
be disused for a considerable period. Under these circumstances
the water in the syphon trap may become evaporated, and then foul167
drain gases be wafted into the house. Furthermore, even if the
water in the syphon trap remained, foul gases may be absorbed from
the drain and given out at the end nearest the house (a, Fig 19).
Hence it is always best to disconnect all
waste-pipes from the drain, except the soil-pipe
which cannot be treated in this way.
The waste-pipe from the upstairs lavatory
or bath may be made to discharge over a
hopper-head and thence into the rain-water
pipe, which is disconnected below from the
drain. This plan should only be adopted
when the hopper-head is not close to a
bedroom window.
Fig. 19.
Syphon Trap under
Sink, with Screw-opening
for Cleansing.
Under the bath is usually placed a
leaden tray, called a safe, to catch any
accidental spillings of water. The overflow
pipe from this safe should discharge direct
into the open air. Formerly much evil was
caused by allowing waste-pipes from baths,
sinks, and lavatory-pipes, or overflow pipes
from drinking-water cisterns or from the
bath-safe, to be connected directly either
with the trap of the w.c., or with the soil-pipe
beyond it.
Fig. 20.
| A Stoneware Gully-trap. | B Section of the same, showing a
Water-seal 3 inches in Depth. |
Sinks are not uncommonly the source
of offensive smells, when made of wood or stone. A hard glazed
sink should be provided; as this is non-porous and can be kept
clean. The sink should be placed against an external wall, so
that the waste pipe can be carried through the wall to a gully-trap
outside the house. Formerly the sink-pipe was joined below
directly into the drain, the only obstacle to the entry of sewer or
drain gases into the kitchen being a bell-trap at the sink. This
is quite insufficient for the purpose. The waste-pipe from the
sink should have a syphon trap under it, with an inspection opening
at its lowest point (Fig. 19), and should discharge in the open air over
a gully-trap (Fig. 20), as in the case of rain-water and bath-waste
pipes. It is usually stated that the waste-pipes from sinks, etc.,168
should discharge at least 18 inches distant from the grating of the
gully. This is too far, because some of the foul water may become
dried up in the channel, and its solid particles be blown about.
They may be allowed to discharge directly over the grating of
the gully (Fig. 20) or even into the side of the gully below the
grating, but above and on the house side of the water-seal shown
at B. Fig. 20.
The gully-trap is connected with the socket of the first drain-pipe,
and the junction is made water-tight by means of a cement
joint. On this account, and because it gives a better water-seal
than the bell-trap or D trap, the gully-trap should be always
used. The best form of gully-trap, the P trap, is shown in
Fig. 20 B. This is better than the S trap (Fig 19), which
involves a bend in the drain at its junction with the trap.
Water-closets require to be skilfully constructed and well-situated,
if they are not to become a serious nuisance. In building
a house, the position of the closet should be carefully considered.
In all cases it should be in an out-standing part of the house,
against an external wall, and separated from other parts of the house
by a passage, preferably a passage which is cross-ventilated.
Instead of this, one commonly finds it in any convenient recess,
abutting on a bedroom, or where it cannot be properly ventilated.
Usually the closet is placed at the back of the house; and as the
main-sewer is generally situated in the front street, it follows that the
drain must in terrace houses pass under the house. Hence the
importance of having it completely water-tight. Water-closets in
bathrooms are very inadvisable.
The ventilation of the closet should be good—if possible, by two
opposite windows; and where practicable a cross-ventilated lobby
should intervene between the closet and the rest of the house. This
is now always provided in hospitals.
The water-supply to the closet should be abundant. Every
flush of water should be sufficient to carry the contents of the basin
through the soil-pipe and the drain into the sewer. The quantity
allowed by the Water Companies in London is two gallons, which
is barely sufficient for this purpose, unless the form of closet pan is
good, and the down-pipe to it of sufficient diameter. Each closet
should have a separate cistern, the best being the so-called “water-waste
preventer,” by means of which a certain quantity of water,
and no more, can be discharged each time the handle is pulled.
One of the best of these is shown in Fig 21. When the handle of
this is pulled, the whole of the water in the cistern is syphoned out
by the syphon and carried down to the water-closet, whether the
handle be held down or not.
The amount of fall from the cistern to the closet should not be
less than four feet, and the pipe should be free from bends in order
to ensure a thorough scouring of the trap and soil-pipe; and the
flushing-pipe should have an internal diameter of not less than 1½
inches. It is commonly supposed that a small flow of water,
trickling continuously down a closet, tends to keep it clean, and169
prevent smells; but the water thus used is simply wasted. Others
fasten up the handle of valve-closets so as to allow a large flow of
water. This does not answer the desired end, and renders the
offending person liable to a penalty for wasting water.
Many different forms of water-closet are in use. In all of them
the main requisites are that there should be (1) a good flush of
water, (2) a rapid removal of the excreta, and (3) no possibility of
reflux of gases. The chief varieties of closets are the pan, valve,
wash-out, and wash-down closets.
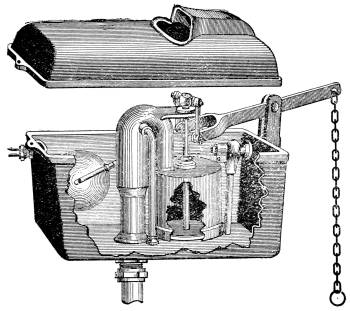
Fig. 21.
Syphon Flushing Cistern.
In Fig. 21 a portion of the bell of the syphon is shown cut out, so as to
display the movable plug at the bottom of the cistern. An objectionable feature
in most cisterns is their noisiness in use. In the above cistern, the pipe
admitting water is carried down to within an inch of the bottom of the cistern,
thus ensuring noiseless entry of water.]
Pan-closets (sometimes called double-pan closets) are essentially
bad, though largely employed in the past. The construction is
shown in Fig 22. Below the conical basin there is a metal pan
capable of holding a certain amount of water, the lower end of the
basin dipping into this water. By means of a pull-up apparatus the
contents of the pan can be tilted into a second larger pan or container,
and the bottom of the container is connected by means of a short
pipe with a leaden [bowl-shaped symbol] shaped trap, from the side of which the soil-pipe
passes out to be carried down to the drain. The arrangement
insures the production of nuisance. The container and [bowl-shaped symbol] trap170
always arrest a certain amount of foul matter; and each time the
handle of the closet is pulled up a puff of foul air comes into the
operator’s face. Occasionally the [bowl-shaped symbol trap becomes corroded by the
filth it contains, and foul gases from the drain
escape into the house.
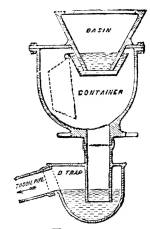
Fig. 22.
Insanitary Pan
Closet, showing
D Trap below.
Valve Closets differ from the last in having
no container, but only a small box containing a
movable water-tight valve, exactly fitting the
lower edge of the basin (Fig. 23). They are much
superior to the pan-closet, but require an overflow
pipe in order to avoid accidental flooding of the
closet. The overflow pipe should be made with
a syphon bend in it, and the flushing of the closet
should be so arranged that each time it is performed
water enters the overflow pipe. (See Fig. 23.)
The trap below the valve should be in the form
of a syphon (see under traps, page 179), as this is
not easily fouled. It is preferably made of lead,
securely jointed to the soil-pipe and to the valve box
of the closet. A lead tray or “safe” is required on the floor beneath a
valve closet, in view of accidental spillings or overflow; and this
should be provided with an overflow pipe discharging into the
open air.
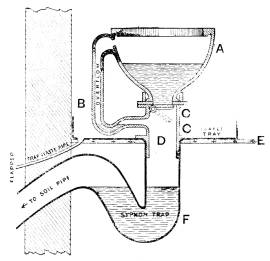
Fig. 23.
A—Pan. B—Overflow pipe with syphon
trap. C—Valve shut. C1—Valve open.
D—Valve box. E—Floorline. F—Water-seal
of trap.
Valveless or Hopper Closets, of which the Wash-out,
Wash-down and Syphonic Closets, are the chief forms, present
certain advantages over the valve closet. There is less apparatus
to get out of order and no metal to become foul. They do not
require an overflow pipe, as
water can escape freely
through the trap of the closet.
Valveless closets need not be
encased by wood-work, thus
ensuring freedom from spillings
of foul water, and they
are more easily used than
valve closets for the discharge
of bedroom slops, thus obviating
the necessity for a special
housemaid’s sink. Valveless
or hopper closets are cheaper
and simpler in use than valve
closets, and when in use are
equally sanitary. If a house
is left empty for a considerable
time, the water in the
trap may, however, become
evaporated, an event much
less likely to occur with a
valve-closet. The latter are
furthermore less noisy when flushed. With a valve to hold up
the water in the pan, as in the valve-closet, a much larger quantity171
of water can be retained than with a hopper closet. Hence the
importance of the latter having such a shape as shall prevent
fouling of the basin by fæces.
One of the older hopper
closets was the long hopper
shewn in Fig. 25. In this
form the pan is conical in
shape, its sides necessarily
becoming fouled by its use,
and the spiral flush, the point
of entry of which is shewn in
the figure is quite insufficient
for cleansing the pan.
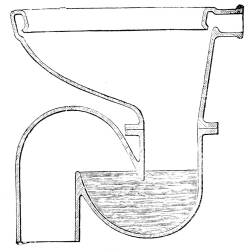
Fig. 24.
Rim Flushing Wash-down Basin.
Of Short hoppers the
best has a nearly vertical back
as shewn in Fig. 24, a rim-flush,
by means of which at
least two gallons of water are
discharged with the fæces,
and the pan is thoroughly
cleansed.
The wash-out closet is
shewn in Fig. 26. In it a
certain amount of water is kept in the upper part of the pan by a
ridge over which the fæces have to be driven before entering the trap.
The force of the flush is thus broken. In this closet a large area
is liable to be fouled, and it is now almost entirely disused.
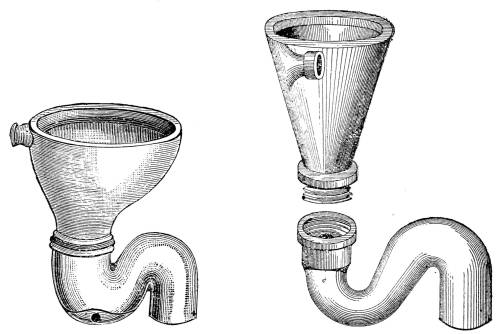
Fig. 25.
Short Hopper Closet. Long Hopper Closet with Spiral Flush.
Syphonic water-closets are wash-down closets, in which172
the flushing out is aided by syphonic action. They need to be
fitted with a flushing cistern, giving an after-flush as well as a
flush; otherwise the basin is left untrapped. One of the most
elaborate closets of this type is Jennings’
Closet of the Century. Fig. 27 shows that
the flushing cistern has two connections with
the closet, one in the usual manner with the
flushing rim of the pan, the other connected
to the long arm of the syphon (A Fig. 27).
B is a puff pipe allowing the escape of air
from this syphon when started. Thus while
one part of the flush scours the basin, the
other expels the air from A through the puff-pipe
B, fills both arms of the syphon with water, and thus starts
the syphonic action by which all the contents of the basin are
sucked out of it. In this form of w.c., syphonage is intended to
be produced, and the after-flush prevents the w.c. from being left
untrapped.
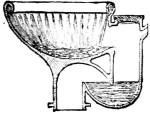
Fig. 26.
Wash-out Closet.
Syphonic water-closets appear to me to be unnecessarily
elaborate and complicated, and the only advantage over the wash-down
closet is the deeper layer of water in the basin. With a
well-shaped wash-down closet this is of little importance.
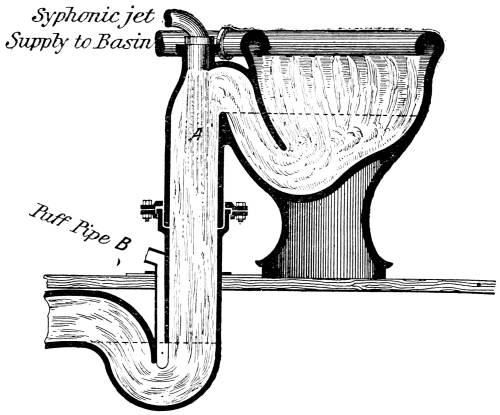
Fig. 27.
Syphonic Closet.
In other forms of wash-down w.c., unsyphoning may also
occur, for instance, by pouring the contents of a slop-pail into the
pan. This is particularly apt to occur, when two or three water173-closets
are on different floors of a house, one over another. This
unsyphoning is prevented in the case of the highest w.c. by the
soil-pipe ventilator, but not always for the lower w.c.’s. For these
it may be necessary to carry a pipe from the highest point of the
trap of the closet, where it joins the soil-pipe, through the wall into
the external air. Such a pipe is called an anti-syphonage pipe
(Fig. 28). It effectually prevents the water being sucked out of
the trap of a lower w.c. when the w.c. on a higher floor is being
flushed.
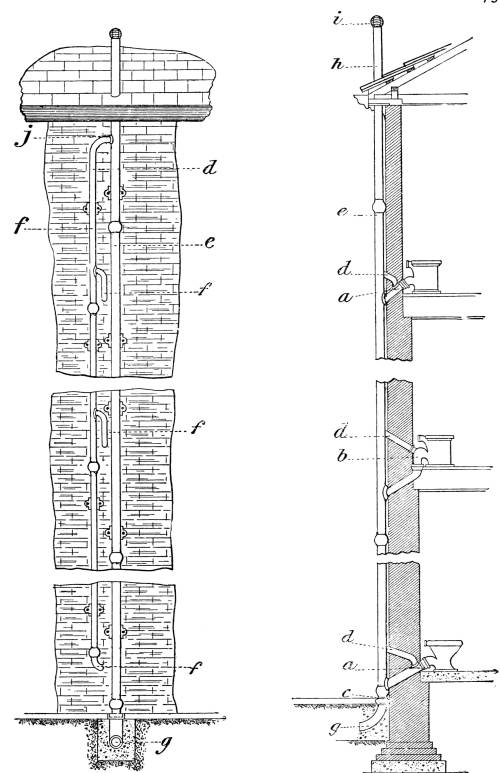
AFig. 28.B
A—Elevation. B—Section through wall of house, showing connection of
w.c.’s on three floors, with soil-pipe and anti-syphonage pipe. b—Junction of
closet trap with soil-pipe, a being a P and b an S trap. c—Junction of soil-pipe
with earthenware drain. d—Anti-syphonage pipe, seen best in elevation A. e—Soil-pipe.
f—Anti-syphonage pipe. g—Underground drain. h—Soil-pipe ventilator.
i—Cage-work protecting top of h. j—Point at which anti-syphonage
pipe is connected with soil-pipe ventilator, above the highest w.c.
174
In the forms of w.c. already described, clean water is used for
flushing, and we have seen that two gallons, the quantity usually
allowed, does not suffice for this purpose, unless the closet pan is
of the best possible shape, and the service pipe sufficiently wide to
project the water by means of a rim-flush forcibly over the pan and
into and beyond its trap. Other forms of w.c. have been employed
of which the most important are slop-water closets, and trough-closets.
In Slop-Water Closets the waste water from sinks and baths
is utilised for flushing, and thus a saving of water is effected. This
form of closet is used considerably in manufacturing districts, and
is less liable to freeze than an ordinary w.c. The sink discharges
on to a gully in the usual manner, but the outlet of this gully is
connected with a tilting vessel or tipper, holding 3½ gallons in
Duckett’s closet, which is the best known of this type. The tipper
is balanced on brass bearings, and tips over when full, discharging
its contents into the closet trap, which is thus flushed. The slop-closet
is a great improvement on the privy-middens or pail-closets,
which in some towns it has superseded, but is not so cleanly as an
ordinary w.c.
Trough Closets are also known as “latrines.” The best type
consists of a glazed earthenware trough under a series of w.c. seats.
The trough is slightly inclined towards the outlet, at which is a
weir, beyond which is a trap. An automatic flushing tank connected
with the upper end of the trough and five to six feet above it,
discharges water at intervals and drives the fæcal matter over the
weir and through the trap. This form of closet is only suitable for
factories. It is to be deprecated for schools, and even for factories,
unless there are exceptional reasons for its continuance, as fæcal
matter possibly of an infectious character may be retained a
considerable time in the trough.
The domestic Slop Closet or “housemaid’s sink” must not be
confused with slop-water closets mentioned above. The slop-closet
or sink is used for emptying the contents of bedroom pails. These
being necessarily foul and liable to early putrefaction must be
treated exactly like other sewage matters. An ordinary pedestal
w.c. with a lift-up seat answers excellently as a slop-closet; but in
large houses and public establishments a separate slop-sink is
desirable with a larger surface than most water-closets. The slop-closet
must be connected with the soil-pipe, just in the same way as
a w.c.
175
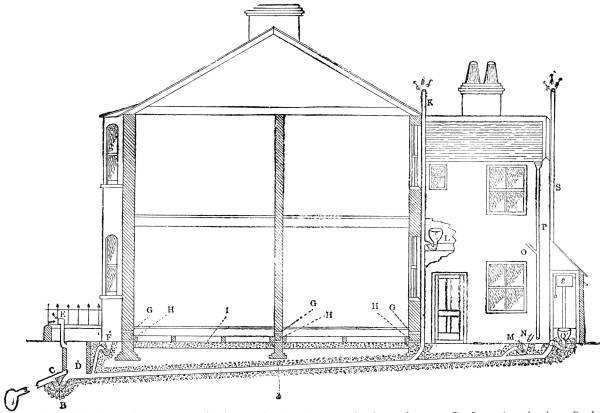
Fig. 29—Section through a House from Front to Back shewing
Drainage Arrangements.
A.—Sewer; B.—Intercepting trap; C.—Cleaning eye for pipes between chamber and sewer; D.—Inspection chamber; E.—Inlet
ventilator; F.—Gully-trap for forecourt; G.—Air-bricks for ventilation under floors; H.—Damp-proof course8; I.—Concrete 6” thick over site
of house; J.—Drain, fall 1 in 24, imbedded in concrete; K.—Soil-pipe carried up full size above eaves; L.—Upstairs w.c.; M.—Gully-trap
receiving water from N scullery-sink, O bath and P rain-water stack-pipe; S.—Ventilating pipe at upper end of drain; T.—Pipes leading to
same.
The soil pipe is the vertical pipe carrying the contents of
the water-closets in the drain. It must be distinguished from the
drain, which is chiefly, if not entirely, underground. The exact
position of the soil-pipe and its relation to the drain can be seen in
Fig. 28 and 29, which should be carefully studied. The soil-pipe should176
be made of drawn lead without seam, of uniform thickness throughout,
and of at least 7 lbs. or better 8 lbs. weight per superficial foot.
Any joints in the lead pipe should be of the kind known as “wiped,”
not a “slip” joint. The outside of a wiped joint is shewn in
Fig. 30B. Iron pipes if used must be 3 ∕ 16 inch thick, and have
sockets sufficiently wide and strong to permit of the joints being
caulked with molten i.e. “blue” lead, in the same way as water-mains
are laid.
The soil-pipe should be throughout its course under observation.
It should not be built into a wall, where it might be accidentally
pierced by nails, nor within the house, allowing foul gases to
escape from weak points in the joints. It should be carried
through the wall of the house immediately beyond the closet trap.
The soil-pipe should not be more than four inches in diameter.
It should be continued from its highest point at the junction with
the closet-trap above the roof by a pipe of the same diameter, with
its end wide open (Fig. 29 K). This ventilation of the soil-pipe is
essential (a) to prevent the entry of foul effluvia into the house,
especially when the water in the closet-trap is dried up; (b) to
prevent unsyphoning of the upper by the lower water-closets in a
house. (On this point see p. 172).
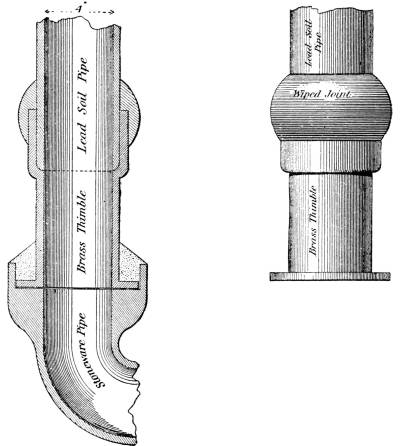
AFig. 30.B.
A.—Section showing a Good Method of connecting Soil-pipe to Drain
by Brass Thimble.
B.—Outer View, showing Brass Thimble wiped on to Soil Pipe.
177
The upper end of the ventilating pipe should be made to open
remote from any window. It may have a cowl attached to it, but
it is doubtful if this materially aids the aspiration of foul gases. It
is wise to cap the upper end of the ventilating shaft with a dome of
large meshed wire-netting to prevent birds building their nests in it.
The connection of the soil-pipe with the closet-pan is its
weakest point, and the most liable to leak. The main difficulty
consists in forming joints between earthenware and metal.
Socketed connections are not safe. The use of an india-rubber ring
inserted between the lead and earthenware flanges and bolted
together by means of a brass collar and hooked bolts makes a fairly
good connection. Various screwed connections are made. In
another form the earthenware collar is covered outside with lead,
so that a soldered joint can be made between the earthenware trap
and the soil-pipe. In the “metallo-keramic joint” the earthenware
joint is painted over with a metallic solution and fired. To the
metal film thus formed, lead or other metal can be firmly soldered.
The connection of the soil-pipe into the socket of the first pipe
of the earthenware drain requires also to be carefully made. This
pipe is curved, and at its upper end has a socket, into which the
soil-pipe enters. A length of brass or copper tubing known as a
“thimble” (about a foot long) should be soldered to the bottom of
the soil-pipe; the rim of this thimble rests in the socket of the
drain-pipe and the space between the two is filled with Portland
cement (Fig. 30A). With the ordinary connection between lead
soil-pipe and drain, the former is apt to become dented by blows,
and the latter is very liable to be partially blocked by the dropping
of cement inside the pipe when making the joint.
The House Drain under ordinary circumstances receives
waste water from sinks and baths, rain-water, and the discharge
from the closets.
Fig. 31.
Showing Depth of Fluid and
consequent Flushing Force of
Three Drains containing an
Equal Quantity of Sewage.
We may consider drains under the following heads: material,
form, joints, gradient, ventilation, trapping. The first essential is
that they should be water-tight, so that their contents do not
percolate into the surrounding soil. Socketed glazed stoneware pipes
and iron pipes best fulfil this condition. The best material for
making stoneware pipes is Devon or Dorset, or similar fine clay,
which makes a very strong pipe. Tested pipes free from cracks
and flaws must alone be used. The pipes should be laid in
straight lines, each pipe being
arranged with the spigot and not
the socket end directed towards the
flow of sewage. The fall should
not be less than 1 foot in from 40
to 60. If the fall is less than this
amount, artificial flushing from
the upper end of the drain is
necessary. Usually branch drains
are made 4 inches in diameter,
the main house-drain having a
diameter of 6 inches. A larger size than this is seldom necessary.178
Thus if A, B, and C be three drains with an equal fall and
conveying an equal amount of sewage, the rate of travel and
therefore the flushing force will be greater, because the depth
of the fluid is greater, in A than in B, and in B than in C. Small
drains are more completely self-cleansing than large drains. The
water-tightness depends on the character of the joints. In this
respect iron drains present the great advantage over earthenware
that there are fewer joints and that these can be rendered permanently
water-tight without difficulty by being run with blue lead and well
caulked. To render an earthenware drain water-tight, (a) it must be
laid on a solid bed of cement concrete at least 6 inches thick, so as
to prevent sinking, and under the house it should be covered with
an equal thickness of cement concrete (I, Fig. 29). (b) The joints
must be made with extreme care, the best Portland cement being
used for the purpose. Clay is inadmissible, as the fibrils of tree-roots
easily find their way through it. The inside of the joint must be
raked by the workmen, before the next pipe is laid, to make sure
that no fragments of hard cement are left projecting in its interior.
Such projections are not uncommon causes of subsequent blockage.
Various patent joints have been used, but they are no better than
the above when properly laid. Just before the drain leaves the curtilage
of the house and near its junction with the sewer, it is trapped,
and on the house-side of the trap (E, Fig. 29) an inlet ventilator is
provided. The general arrangement should be studied in Fig. 29.
Ventilation of the house drain from end to end is important,
a free escape of foul gases out-of-doors being induced. The exit is
provided by carrying the soil-pipe up full bore above the eaves, and
remote from windows. One opening alone would not induce a
current of air, and the other end of the drain being trapped from the
sewer (B, Fig. 29) it is necessary to provide an inlet for fresh air at
E. This may be placed a few feet above the ground or at the
ground-level. Usually a mica-flap valve is provided at its upper
end, which closes whenever a puff of foul air attempts to escape from
the drain. The necessity for this is doubtful. Ordinarily air enters
at the inlet and circulates through the drain, escaping at the upper
end of the soil-pipe ventilator.
It has been advocated that the soil-pipe ventilator should form
a means of ventilating the sewer as well as the house-drain, the
intercepting trap in the latter (B, Fig.
29) being removed. This is inexpedient,
an element of risk being introduced,
in view of the possibility of the drain
or some of the connections of internal
sanitary fittings being defective.
Fig. 32.
Weaver’s Intercepting Trap.
The drain as ordinarily arranged
is trapped from the sewer by an
intercepting trap. This is not
merely a trap, but a trap provided
with ventilation at its end nearest the
house. A form commonly employed
is shown in Fig. 32. B is the junction with the house-drain, at D179
is the water-seal, while at C fresh air enters the house-drain. E is
a cleaning eye, through which any chokage can be cleared towards
A, leading to the public sewer. It is important that the intercepting
trap should be accessible in the event of accidental stopping. This
is provided by an inspection chamber or man-hole. This if
close to the house is provided with an air-tight cover, the inlet
ventilator being conveyed above ground to a convenient point. The
man-hole itself is built with brick set in cement and lined with
cement. Note that in the man-hole itself half-channel pipes convey
the sewage instead of complete pipes.
Sometimes more than one drain-pipe converges to the same
man-hole, and then a more elaborate arrangement than that shown
in Fig. 29 is required, the branch-pipes converging into half-channel
pipes in the man-hole.
Varieties of Traps.—Traps are placed at various points of the
house-drainage system to prevent the admission of currents of foul
air into the house. They are all constructed so as to intercept a
water-seal between the drain and the house or yard at the upper end
of the trap. Traps are placed in four positions in connection with
the drainage of a house: (1) near the junction of the house-drain
with the sewer; (2) under the pan of each w.c.; (3) in the open
air at the ground level to receive waste water from bath, sink, and
lavatory basin; and (4) in the waste-pipe close under the bath,
sink, or lavatory, when the waste-pipe is long and apt to become
foul; (5) inside sinks at the upper end of their waste-pipes.
Intercepting traps between the drain and sewer have already been
described. They must always be ventilated (C, Fig. 32 and Fig.
29). A syphon shape with a water-seal of 3 inches is required, and
the trap should be self-cleansing, that is, whenever the w.c. is used,
the fæces ought to be carried beyond the intercepting trap into the
sewer. Other forms of intercepting trap were formerly used, one of
the worst of which is shown in Fig. 33. With such a trap as this,
an accumulation of filth is inevitable.
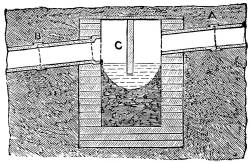
Fig. 33.
Deep Dipstone Trap, with Accumulation
of Filth in it.
A—Drain entering trap. B—Drain leaving
trap. C—Dipstone.
Water-Closet and Slop-Closet Traps are of the syphon or
anti-D type. The water-seal
in these must be at least 3
inches deep, and the trap must
be ventilated by an upright
extension of the soil-pipe,
otherwise the water in the trap
may be syphoned out when
the w.c. is used. Hellyer’s
“anti-D” trap is a lead
syphon trap, the calibre of
which is diminished at its
bent portion, while the portion
of the trap nearest the soil-pipe
or drain is square instead of
circular. The constriction
increases the force of the flush
of water and thus cleanses the whole trap, while the square shape180
impedes the free flow of water, and thus diminishes the risk of
syphonage. Various forms of trap are shown in Fig. 19 to 34. The
most objectionable of these is the old-fashioned D trap (Fig. 22),
the corners and angles of which become fouled, and consequently
the lead becomes corroded.
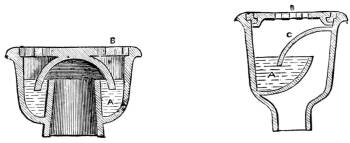
Fig. 34.
Bad Form of Trap: Bell Trap.
Bad Form of Trap: Antill Trap.
Gully-traps are placed in the yard, for the discharge over
them of waste-pipes (Fig. 20). A complete disconnection from the
drains is thus effected. Formerly bell-traps were used for this
purpose. In the Bell-trap not much water can get through, the
space A becomes blocked with dirt, the cover B is often taken off
and lost, and then the drain is untrapped; and even without this,
the water-seal is very slight, and the water quickly evaporates.
Traps under sinks, etc., have been already described (page 166).
Traps were formerly placed at the upper end of the waste-pipe
of the sinks when this was directly connected with the drain. Of
these the Bell-trap and Antill’s trap were most common. The
Bell-trap has been described above. In the Antill-trap the trap
is not removable, and the water-seal is deeper than with a bell-trap.
This trap is sometimes used instead of a gully-trap, but is not so
good.
Efficiency of Traps.—Eassie has said “honestly speaking,
traps are dangerous articles to deal with; they should be treated
merely as auxiliaries to a good drainage system.”
(1) The trap may have been imperfectly laid to begin with.
(2) It may be emptied by evaporation.
(3) Unless the precautions already mentioned (pages 172 to
173) are adopted, the flushing of one trap may empty another.
(4) The water of the trap may become impregnated with foul
gases, and these then escape on the house-side of the trap. When
a sewer becomes suddenly charged with a large amount of water, as
during heavy rain, sewer-gases may force their way through the
intercepting trap. With a ventilated drain and soil-pipe these
dangers are so small that they may be ignored.
Unsyphoning of traps has been already mentioned. It occurs
particularly when there are several water-closets one over another,
connected with the same soil-pipe. The method of preventing it is
shown in Fig. 28.
The Examination of Drains and Sanitary Appliances.—This
examination will involve the detection of (a) any deviations181
from the details of construction and ventilation of drain and soil-pipe,
form of w.c., disconnection of waste-pipes, already insisted
on; and (b) any defect or leakage in any part of these.
1. Testing of Water-closet.—The interior of the basin or pan
may be painted with a mixture of lamp-black, size and water. If
the usual flush applied immediately afterwards clears this off, the
form of pan and the flushing power are satisfactory. By removing
the wood-work around the w.c., leakage or spillings of slop-water
around the w.c. can be detected.
2. Testing of the soil-pipe may be effected by one of the volatile
tests named under the next heading. To give the test a fair trial,
the upper end of the ventilating pipe should be temporarily sealed
over.
3. Testing of the drain cannot be efficiently carried out unless
access can be obtained to the drain near the sewer. In a properly
constructed house-drain a man-hole is provided for this purpose.
Two chief methods of testing drains and soil-pipes are in use, by
smoke or volatile agents and by water.
The smoke-test consists in filling the drains with smoke, the
assumption being that this will find its way through any faulty joint
or trap, thus indicating the site of the defect. Various arrangements
are employed for pumping the smoke into the drain from the
combustion chamber of a pumping apparatus; or smoke is produced
by means of specially prepared rockets. All outlets or ventilating
pipes must be carefully stopped during the operation, and the place
where the smoke is smelt will then indicate any leaky point.
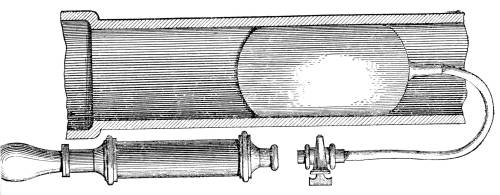
Fig. 35.
Showing Stopper for Water-testing of Drains.
Drain grenades are largely employed for the volatile testing of
drains, the essential constituent being phosphide of calcium. The
grenade, which is attached to a piece of string, is passed beyond the
trap of the w.c., and as the string unwinds the grenade opens and
discharges its contents into the soil-pipe. Or a tablespoonful of
strong oil of peppermint, mixed with hot water, is poured down the
highest water-closet in the house. If this is smelt by another
person in the lower closets, it indicates defective traps or soil-pipe.
182
All volatile and smoke tests have but a limited utility. They
are useful in detecting defective joints in traps and in the soil-pipe.
They may detect defects in an underground drain; but
if no smell or smoke is perceptible when a drain is tested by this
means, the drain may still be seriously defective. The only
absolutely trustworthy test for drains is the hydraulic or water
test. The lower end of the drain is stopped up by a suitable water-tight
stopper. Then the drain is filled with water by means of a
tap in the yard, the amount of water used being approximately
estimated by the rate of flow from this tap. The drain is filled up
to the level of the gully-traps in the yard. If it remains at this
level for half an hour, the drain is sound. More often it leaks so
rapidly that it will not fill, or the level of the water falls quickly
after filling, and it is necessary to strip and repair, or more generally
to relay the drain so as to make it water-tight.
Rats are an important indication of defective drains. The
presence of rats in a house should always lead to a thorough
investigation of its drains.
183
CHAPTER XXVI.
CESSPOOLS AND MAIN SEWERS.
The terms Sewer and Drain are used somewhat confusedly.
The term drain should be used to designate the pipes bringing the
sewage from the house into the street-sewer, or any pipes by which
the subsoil is drained; the term sewer being confined to the trunk
canals into which the house drains empty their contents.
Where the water-carriage system of sewerage is adopted, involving
the use of water-closets as described in the last chapter, the
sewage may be carried from the house either into cesspools or into
the main sewer.
Cesspools are only permissible in isolated country-houses
supplied with water-closets. They should always be situated a
considerable distance from the house, and should be emptied at
regular intervals, the sewage being placed in shallow trenches on
the land.
The construction of the cesspool requires careful attention.
Its walls should be of brickwork set in cement, lined inside with
cement, and surrounded by clay puddle. The bottom should have
a fall towards one side, where a pump can be fixed, to remove the
more liquid contents. The depth of the cesspool should never
exceed 7 feet. The drain emptying into the cesspool should be
trapped and ventilated, near its junction with the cesspool; and the
cesspool itself should be ventilated.
In connection with many old houses in towns, cesspools still
exist, sometimes under the basement or near the house, and so built
as to allow soakage in every direction. The surrounding soil
becomes contaminated for a considerable distance, the water in any
neighbouring well is tainted, or leaky water-pipes receive the
soakage. The cleansing of cesspools is always a disgusting
process, and even dangerous to the workmen employed. They
incur the risk of suffocation, and are very subject to ophthalmia.
To avoid these dangers a pump and hose connected with a partially
exhausted barrel is employed, but even with this provision some
nuisance arises. In the Bexley cart, which is used for this purpose,
a hose is used to connect the cesspool with an air-tight cylinder in
the cart, into which the contents of the cesspool are pumped.
A modification of the cesspool system, called the Pneumatic
System has been proposed by Captain Liernur. In it the cesspool
is not placed under the house or the courtyard of the house, but
under the street at the angle of junction of several streets. It is184
made of cast-iron and air-tight, and is connected with all the houses
of several streets by iron pipes. By means of a powerful air-pump
worked by steam, the cesspool is emptied into barrels in which it is
sent directly to farms; and the barrels being placed on ploughs
of peculiar construction, the manure is discharged from the bung-hole
of each barrel and covered over with earth in the progress of
the plough. The pipes tend in this and similar systems to get
clogged with fæcal matter, and large quantities of water are required
to keep them clean, so that the system merges into that of the use of
water-closets, but without the thoroughness of the latter.
Cesspools have been almost improved out of existence in some
continental towns, by the introduction of movable cesspools,—fosses
mobiles,—to which would correspond strictly the tubs and pails used
in some of our large towns. Such movable receptacles have been
still further “improved” by the adoption of separators, by which the
liquid parts are allowed to escape into the sewer, while the solid
parts remain comparatively inoffensive. But when this is done, the
cesspool may be as well abolished, as the foulness of the sewage is
not greatly increased by allowing solid as well as liquid excreta to
enter it.
Sewers are built of glazed stoneware or of impervious brick laid
on a bed of concrete to prevent sinking of any part, the parts being
most solidly put together with cement. Iron and steel pipes are also
used especially when extra strength is required, as when there is
some danger of the pipes sinking. For most small streets circular
stoneware sewers suffice. Oval brick sewers are more suitable for
main streets in which the amount of flow varies greatly. The cross-section
of these should be an egg-shaped oval with the small end
downwards, as this ensures the most rapid current. Sewers should
be laid in as straight a line as possible, and with a fall which
will ensure a flow of at least 2½ feet per second. The following
rule gives approximately the fall required for smaller sewers and
drains:
- A 4-inch pipe requires a fall of 1 in 40.
- A 6-inch pipe requires a fall of 1 in 60.
- A 9-inch pipe requires a fall of 1 in 80.
For further particulars as to the velocity of flow see page 187.
Where a town is very flat, and a proper fall of sewer impossible,
Shone’s ejectors are sometimes used to raise the sewage. The
sewers are laid in sections, each section falling to a certain point,
from which the sewage is raised by the ejector to a higher level and
so carried to the next section of sewers. Each section has a separate
system of ventilation. The provision of manholes for inspection of
an intercepting trap and of ventilation of the house drain near its
junction with the sewer has already been considered (page 179).
As sewers have commonly to carry away the rain-water in
addition to the waste matter from houses, their size must be
regulated accordingly. The rainfall being very various, the sewers
may occasionally become overcharged and flood the basements of
houses. During heavy rainfall large quantities of road grit are185
washed into the sewers, the intercepting gully tanks at the road
sides being insufficient to prevent this.
In addition to the above disadvantages associated with discharging
storm water into sewers, the sewage owing to its increased
bulk is more difficult of disposal, whatever method of disposal be
adopted. The size of the sewers and of storm-outfalls into the nearest
river or the sea must be regulated so that they are equal to these
sudden strains on them; or the Separate system, by which the rain
is carried in special conducts to the nearest river, must, in the
alternative be adopted. The objections to this plan are that it
necessitates a double system of sewerage, and does not allow of the
useful scouring effect of rain on sewers. Where it is feasible, and
particularly in small country towns, its adoption is advisable. In
such cases the old brick drains are used for carrying off rain-water,
while new pipe-sewers are employed for carrying the sewage. Such
pipe sewers are not liable to become fouled, and on account of the
decreased dilution of the sewage can be made smaller than brick-sewers.
Sewers being closed conduits containing sewage, it is highly
desirable that the gases resulting from decomposition should be
freely diluted. Such gases in unventilated sewers may find their way
through intercepting traps into the house drain. The danger from
this source has been considerably exaggerated. Ventilation of
the sewer is, however, desirable. This has been commonly
accomplished by gratings opening at intervals directly into the
middle of streets. In narrow streets, the stench from street-grids is
occasionally a source of complaint, and may cause malaise and
ill-health. Charcoal traps placed below sewer grids to intercept
offensive gases have been found to be of little use. The best plan
is to do away entirely with surface ventilators, and carry up iron
shafts or brick shafts lined with stoneware pipes above the level of
all neighbouring houses. The only difficulty in adopting this plan
is the difficulty in securing premises to erect such shafts up houses,
although no danger attaches to the practice.
Ventilating shafts should be erected at intervals and particularly
at the highest points of the sewage system of a town, the upper end
of these shafts being remote from the windows of any dwelling-house.
When sewers are laid with too steep a gradient, they act as
chimneys, the gases mounting to the higher part of the town,
and frustrating attempts at ventilation on lower levels. Various
attempts have been made to ventilate sewers by artificial means, as
by the aspirating effect of street lamps, etc., but these efforts have
not been successful, as the effect of the up-current only influences a
short length of sewer. The usual method of combined up-shafts and
street grids answers fairly well, but when any complaint of smell
from street-grids occurs they should be replaced by up-shafts. In
Bristol and a few other towns no provision is made for sewer-ventilation,
and no ill effects have apparently resulted. There can
be no question that the importance of sewer-ventilation has been
exaggerated. If the sewer has a sufficient gradient, and is properly
laid, and efficiently flushed, so that no offensive deposits occur, the186
provision of up-shafts at favourable points is all that is necessary.
Sewer effluvia have been credited with causing enteric fever,
diphtheria, and other diseases. These diseases rarely if ever owe
their origin to this cause. The microbes discovered in sewer-air
have always been those of the outside atmosphere, and not derived
from the sewage. Sewer effluvia might, however, predispose to
such diseases, if exposure to them were frequent or protracted, by
lowering the powers of resistance of the constitution.
Flushing of Sewers is required at intervals, in order to remove
any deposits of grit or other solid matter. Flushing is effected by
filling special flushing shafts placed at intervals in the sewers with
clean water and then suddenly releasing this.
Whenever there is stagnation, a foul odour is certain to be emitted.
The cardinal rule with regard to sewage is to keep it in rapid
onward motion, until it has passed the outlet of the sewer. The
introduction into a sewer of hot water or waste steam is an occasional
cause of nuisance.
The Outfall of a sewer requires to be large and perfectly free
in order that the progress of the sewage may not be impeded.
When the sewage is discharged into the sea above the low water
level, it becomes backed up in the main sewers when the tide is
high. The same condition of things has occurred when the outfall
is into a river below the water line, or into a tank out of which the
sewage has to be pumped. In all these cases the ventilation of the
sewers requires to be perfect, and great precautions taken to prevent
obstruction of the outflow.
In low-lying sewers where the outfall is impeded, mechanical
aids are required to prevent blocking of the sewers. This may be
obtained by pumping at the outfall, to enable sewage to escape at
all conditions of the tide, or to raise the sewage on to land for
irrigation. In the Shone system the sewage is raised to the
required height by means of compressed air. In this system the
sewage is received into “ejectors.” These are cylindrical reservoirs,
in which is a float on a counterpoised lever. When a certain
quantity of sewage has entered, a valve opens admitting the
compressed air, which forcibly raises the sewage into a higher
length of sewer or to the outfall.
187
CHAPTER XXVII.
PROBLEMS AS TO FLOW IN SEWERS.
In order to prevent deposit of solid matter, sewers should be constructed with a
sufficient gradient, and of a shape which presents the least surface for friction in
proportion to the amount of liquid to be conveyed.
All brick sewers should be egg-shaped, with the narrow end downwards.
The egg is formed by two circles touching one another, the diameter of the
upper circle being twice that of the lower.
This shape possesses the great advantage that when the depth of the stream
is diminished the amount of wetted surface of sewer (wetted perimeter) is
diminished in equal proportion, whereas in every other form of sewer it is
relatively increased. Thus the friction, which depends on the extent of the
wetted perimeter, is kept down to a minimum. Where, as in outfall sewers, the
volume of sewage is large, and does not vary greatly in amount, the circular
form may be preferable, as it is cheaper and stronger than the egg-shaped sewer.
Below 18 inches internal diameter, sewers should be circular in section, and
made of stoneware, not brick.
The velocity of flow depends upon (1) the hydraulic mean depth of the
stream, and (2) its inclination or fall.
The hydraulic mean depth means the depth of a rectangular channel whose
sectional area (and therefore the volume of whose current) equals that of the
curved sewer or pipe, concerning which the calculation is made, and whose
width equals the entire wetted perimeter of the sewer or pipe. It is thus equal
to sectional area ∕ wetted perimeter.
In the case of circular pipes, if we take the diameter to be 1, and assume
the pipe to be running full, the sectional area = πr2, where π = 3·1416 and r =
half diameter.
The wetted perimeter = 2πr, that is, the circumference of the circle formed
by the pipe.
Therefore hydraulic mean depth = h = πr2 ∕ 2πr = ¼;
Similarly when the pipe runs half full—
- πr2 ∕ 2
- h = ——— = ¼
- 2πr ∕ 2
The solution of problems where a smaller arc of a circle is occupied by
fluid requires trigonometrical methods, and is not usually needed in practice.
The quantity of fluid discharged in a given time is represented by the product
of the sectional area of the stream into its velocity. The greater the hydraulic
mean depth the greater is the velocity, if the inclination remains the same.
The velocity of flow is determined by Eytelwein’s formula, which states
that the mean velocity per second of a stream of water similar in form to those
now under consideration is nine-tenths of a mean proportional between the188
hydraulic mean depth and the fall in two English miles, if the channel were
prolonged so far.
- Thus if f = the fall (in feet) in two miles,
- h = hydraulic mean depth in feet,
- V = mean velocity per second,
- Then V = ·9√(hf),
- or if v = velocity per minute, then
- v = 55√(hf).
It is more convenient to let f = fall in one mile.
Then the formula becomes v = 55√(h × 2f).
How much sewage will a circular drain 3 feet in diameter running half full
convey, the fall being 1 in 400?
Here h = (πr2 ∕ 2)/(2πr ∕ 2) = r ∕ 2 = ¾.
- 1 in 400 = x in 5,280 feet (i.e. a mile).
- f = 13·2 in a mile.
- v = 55 × 4·4 = 242 feet per minute.
- = 55√h × 2f.
- S = πr2∕2 = 3·1416 × 9/(4 × 2) = 3·5343.
- v × S = 242 × 3·5343 = 855·8 cubic feet, discharged per minute.
In what way does the size and shape of a sewer affect the velocity of the sewage
flowing through it? If a 12-inch pipe sewer, laid at a gradient of 1 in 175, gives a
velocity of 3½ feet per second, what would be the velocity if the sewer had a gradient of
1 in 700 (the pipe running half full in each case); and would this latter velocity suffice
to keep the sewer clear of deposit?
An elliptical sewer gives greater velocity to flow of small quantities of sewage
than a circular one because it exposes a smaller surface for friction.
By formula = v = 55√h × 2f.
- h = ¼ ∴ √h = ½.
- f = 1 in 175 = 30 feet in one mile.
- v = (55 ∕ 2)√60 = 212·85 ft. per min., i.e. slightly over 3½ ft. per sec.
In the second case f = 1 in 700 = 7·56 feet in one mile.
v = (55 ∕ 2)√15·12 = 106·97 feet per minute.
Thus in the first case there is a velocity of 3·55 feet per second, and in the
second case of 1·78 feet per second. The latter velocity is quite insufficient to
keep the sewer free from deposit, 3 feet per second being the minimum velocity
required for that purpose.
Given a sewer 3 feet in diameter, with a fall of 1 in 1,760, what would be the
relative discharge if the fall were 1 in 5,280?
In the first case, 1 in 1,760 = 3 in mile.
1 in 5,280 = 1 in mile.
h = r ∕ 2 = ¾.
v = 55√(h × 2f)
= 55√(¾ × 6) = 165/√ = 118.
In second case v = 55√(¾ × 2) = 55√(3 ∕ 2) = 67·9.
Thus the velocity of the two streams would be as 118: 67·9.189
Supposing a sewer to have a gradient of 1 in 300, how much would the velocity and
discharge be increased by altering the gradient to 1 in 100?
- 1 in 300 = 17·6 in mile.
- 1 in 100 = 52·8 in mile.
As h is not given, we must assume it = ¼;, as it does in circular sewers
running full or half full.
- v = 55 √(h x 2f)
- = 55 √(35·2 ∕ 4) = 163 feet per minute.
- v1 = 55 √(104 ∕ 4) = 281 feet per minute.
The increase in discharge may be similarly calculated.
Describe the relation existing in a sewer between gradient, volume, velocity, and size.
By the formula v = 55 √(h. f.)
- Where v = velocity in feet per minute.
- h = hydraulic mean depth = (area of cross section of stream)/(wetted perimeter).
- f = fall in feet in two miles.
In circular sewers h = diameter ∕ 4.
Thus the velocity varies as the square root of h or f.
The volume discharged varies with the value of the factor v × s where s =
sectional area of stream.
If h remains constant, with a varying volume of s, then the volume discharged
may remain constant. Thus h and v in a circular sewer are the same,
whether the sewer runs full or half full. In a V-shaped channel the velocity
remains the same whatever the depth of the stream, as its bed and area preserve
the same proportions. An egg-shaped sewer approximates the V shape in form.
Similar volumes of sewage have velocities which vary not only with the
amount of fall, but the size of the sewer. The friction, as represented by the
wetted perimeter, would be much less with sewage half filling a circular sewer,
than with the same amount of sewage forming a broad shallow stream on the
invert of a large sewer.
190
The water-carriage system of sewage is, as the late Dr. Parkes
put it, “the cleanest, the readiest, the quickest, and in many cases
the most inexpensive method.” But when the sewage is conveyed
to the outfall of the sewer, its ultimate disposal is still one of the
most difficult problems of the present day. Various plans have been
adopted, of which the following are the chief:—
| 1. Discharge into running water. |
| 2. Discharge into the sea. |
| 3. Separation of solid and liquid parts |
 |
By settlement. |
| By precipitation. |
4. Filtration through various
artificial media or through land. |
| 5. Irrigation. |
| 6. Bacterial methods. |
Discharge at once into running water was formerly the
favourite plan, as it was certainly the most convenient. The sewage
was turned into the nearest water-course, regardless of the facts that
this might have to supply the drinking water of people at a lower
point, that the mouth of the river tended to become obstructed by
sewage mud, that valuable stocks of fish were destroyed, and
that the river which had practically become a sewer was a source
of annoyance and danger to all on it or near it. The enforcement
of the Rivers Pollution Prevention Act of 1876 has not been
followed by as great improvement as is desirable.
The sewage entering rivers undergoes considerable purification
by subsidence, by oxidation, by the influence of water plants, and
still more by the active work of microbes, causing nitrification of
nitrogenous matter. The vitality of the typhoid bacillus and of the
cholera vibrio when discharged by sewage into a large river is
probably not very protracted; but water from such a river would
form a very dangerous source of domestic supply.
Discharge into the Sea is resorted to in seaboard towns. The
outfall must be carried well below the lowest low-water mark, and
to such a point that the incoming tide or wind will not bring the
sewage back upon the shore, or on the shore of neighbouring
places.
Discharge into an Estuary is only justifiable when the flow
of the river is rapid, when the volume of water passing out to sea is
very greatly in excess of the volume of sewage, and when there is no191
possibility of contaminating oyster-layings or beds of mussels or
other molluscs.
Objection has been taken to the above method on the ground of
waste of manure; but modern sewage is so dilute that its profitable
utilization on land still remains a dream.
For a single house or small village, the sewage may be stored in
a tank, with an overflow-pipe, out of which the liquid parts
escape, and are systematically used to irrigate land, while the
solid parts are removed at intervals.
A similar subsidence system has been employed on a larger
scale, the liquid parts being irrigated over land, while the solid parts
are mixed with street sweepings, and sold as manure.
If the liquid parts in any such system as this are turned into a
stream, they are as dangerous as the entire sewage, and the legal
prohibition to discharging sewage into streams applies equally
to them.
The precipitation of the solid parts of the sewage is rendered
much more perfect by the use of chemical agents, and at the same
time the dissolved matters are to some extent removed.
Milk of lime has been employed, 6 to 12 grains of quicklime
being used for each gallon of sewage. Secondary decomposition is
apt to occur in the effluent, causing an offensive smell. Salts of
alumina, iron salts, and various combinations of these have also
been employed, but with imperfect results.
The London sewage for some years has been treated by adding
2·5 grains of sulphate of iron and 3·7 grains of lime to every gallon
of sewage; a reduction of 15 to 20 per cent. in the amount of
dissolved organic matter being secured. Polarite or magnetic
spongy carbon is used as a filter in certain places, the solid and
some of the dissolved sewage being first precipitated by magnetic
ferrous carbon (ferrozone). The Amines process consists in
applying a mixture of herring brine and lime to the sewage. The
sewage is stated to be sterilized by this means. Electrolysis has
also been applied to the purification of sewage, as in the Hermite
process. In this process sea-water is electrolysed, oxygen-yielding
compounds and chlorine being produced.
With regard to all the chemical processes hitherto introduced,
the following general statement appears to hold good: they are
expensive and not thoroughly efficient.
Sewage sludge is deposited in the tanks in chemical processes
and needs separate disposal. At Birmingham the amount of sludge
produced daily from the sewage of a thousand persons is nearly a
ton. This sludge has been run into rough filter beds and left to dry
or carted away for manure, but in its crude state its manurial value
is very slight. At Ealing it is mixed with house-refuse and burnt in
a destructor. The more modern method is to pass it through a
filter-press, thus compressing it into solid cakes which can be sold
for manure.
Filtration of the sewage matter has been accomplished in
various ways.
192
Intermittent downward filtration through a considerable depth of
soil was stated by the Rivers Pollution Commission to be attended
with good results. A porous soil is chosen, and the purified water
is received in drains under it. A large part of the organic matter is
removed by bacterial agency. Frankland’s experiments shewed
that upward filtration through the same media did not purify.
Filtration through artificial media has not been successful with
crude sewage. Precipitation by ferrozone followed by filtration
through polarite is said to be satisfactory.
Broad Irrigation purifies the sewage efficiently under
favourable conditions, the possible exceptions being during rainstorms
and during frosty weather. The effluent into the river
cannot, however, be regarded as certainly innocuous, though it is
better than the effluent from most other processes. Sewage farms
are not a commercial success. In such a farm liquid sewage is
allowed to flow at intervals over the land, different fields being
irrigated in rotation. Immense crops of grass are obtained, but the
grass is coarse and rank.
The soil to be irrigated should have a gentle slope, and the
effluent be conveyed by subsoil drains about 5 or 6 feet deep into
the nearest water-course. The sewage should be delivered in as
fresh condition as possible, and should be freed from its coarser
portions by settlement or precipitation. The amount of land
required is about 1 acre for the sewage of 100 persons. The
irrigation must be on the intermittent plan, in order that the soil
may undergo aeration; as it is only in this way that the best
purifying results can be obtained. The sewage farm should be well
drained by deep-laid agricultural drains. The chief purification of
the sewage occurs in the superficial layers of the soil. Nitrification
ceases at a depth of about 18 inches. The great point, therefore, is
to keep the superficial soil in good condition. A similar nitrification
occurs in earth-closets (page 195). No nuisance need arise in
connection with a sewage-farm, and the supposition that milk and
other products from such a farm are less wholesome than the same
products from other farms has proved to be unbased.
Bacterial Methods of Treating Sewage.—Chemical precipitation
of sewage is likely to be completely superseded by biological
or bacterial methods of sewage disposal. When sewage is treated by
filtering through land or by broad irrigation the process is bacterial,
bacteria or microbes in the soil converting injurious organic matter
into innocuous mineral products. The typical process is one of
nitrification. The novelty of recent methods is in utilising bacteria
for the whole process of purification, and not only for its final stages.
The object is, in fact, not as in chemical processes to arrest, but by
confining the sewage in tanks to aid and hasten decomposition or
putrefaction. Two kinds of microbes serve in this process; those
living in air, known as aerobic, and those living in other gases than
air, called anaerobic.
Three biological methods of preliminary treatment of sewage
are employed. (1) Mr. Scott Moncrieff passes the sewage slowly193
upwards through a filter 14 inches thick, consisting of successive
layers of flint, coke, and gravel. This is called a “cultivation tank.”
The solid sewage becomes liquefied in passing through this medium,
the microbes in the filter dissolving the sewage. (2) In the “septic
tank method,” introduced by Mr. Cameron at Exeter, a tank is
employed which is covered in to exclude light, and to a large extent
air. The tank is large enough to hold 24 hours’ flow of sewage.
The microbes in the sewage under these conditions multiply rapidly,
attack, and liquefy the sewage. As in the first process little or no
sludge is left. The ultimate products of the decomposition are water,
ammonia, and carbonic acid, and other gases. The effluent from the
tank is comparatively clear and inoffensive. (3) Aerobic biological
filters are employed, as in Mr. Dibdin’s installation at Sutton, where
the filtering material is coke. The sewage slowly passing through
the filtering medium becomes liquefied, the solid matter being
peptonised. This action is in part at least due to anaerobic
microbes. The filtering beds are used intermittently to allow of
aeration, and the liquefaction of solid organic particles entangled in
the filter probably chiefly occurs at this stage. It is desirable to
have small subsidence tanks, for the removal of large suspended
matters and of road debris, etc., before the sewage is spread over
the filtering beds. The material used in the filter varies. Most
commonly coke-breeze has been employed, but coal slack and other
material have also been utilised.
After the preliminary treatment above described, the sewage
requires to be passed over finer filtering beds, in which aerobic
microbes complete the purification by changing the dissolved organic
matter into inert inorganic compounds, by the process known as
nitrification. The two processes run into one another, to some
extent going on together.
Hitherto the Local Government Board have required filtration
of sewage through land before any sewage effluent is allowed to pass
into a stream. In view of the successful results now obtainable by
bacterial processes this requirement will be occasionally waived. It
is unsafe to assume, however, that the clear effluent obtained is
free from all disease-producing microbes; and drinking water should
not be obtained from even a very large river below the point of
discharge of such an effluent, without the most efficient sand
filtration.
194
The refuse to be removed from a house consists of fouled water,
which is at least equal in quantity to the water-supply of the house;
the excreta of the inhabitants; and “dust,” which contains, besides
ashes, considerable kitchen refuse; consisting of both vegetable and
animal matters.
In dry methods of removing refuse, the “dust” is often added
to the excreta, and the two removed together; or the “dust” may
be separately removed. In either case the foul water, and to a
large extent the urine, remain to be dealt with, and require special
drains for their removal. Thus in large towns, whether dry or wet
methods of removing sewage are adopted, drains for the removal of
foul-water and rain-water will be required, and it is found that they
are practically as foul as if they contained the solid excreta.
The dry methods of removing sewage involve a certain amount
of retention about the house; hence the general name of conservancy
methods. Of these the most important are—
1. The pail system.
2. The dry-earth system.
3. The midden or privy system.
The Pail System implies in reality the use of a movable
cesspool. The pail may be used alone, or may contain ashes and
house refuse, or some deodorant. Where the pail is used without
any admixture of foreign matter, it should be emptied daily, and
care should be taken that the pails for different houses are not
exchanged.
In the Goux System the tubs are lined with a composition
containing clay and furnished at the lowest part with some
absorbent material such as chaff, straw, or hay, which serves to
absorb the urine and retard putrefaction. This is, when well
managed, somewhat less offensive than the ordinary pail system.
The pails may be supplied with a deodorant, such as sulphate
of iron, as at Birmingham, Leeds, etc.; they may be packed with
absorbent material, as in the Goux system (Halifax); the ashes
and house-refuse may be deposited in the same pail (Edinburgh,
Nottingham); or coal ashes may be scattered over the excreta
(Manchester, Salford); but all these systems are rapidly being
superseded.
Although the pail or tub system is an improvement on the
midden system, it is necessarily a cause of considerable nuisance,195
and its replacement by water-closets should be recommended in
towns. In detached country houses it may be retained without
nuisance, if the pail or tub is emptied daily, and its contents at
once placed in the garden beneath a shallow layer of earth. The
pails in large towns are usually collected in specially-constructed
closed wagons. In some towns the pail contents have been burnt
in a “Destructor” (page 200) after having been mixed with ashes.
In other towns attempts have been made to utilise the excreta,
either by selling in their crude condition or after drying and
deodorising them by heat. None of these methods repays the cost
of collection. Mixing ashes with the excreta diminishes any
possible value they may possess as a manure.
The Dry-earth System is an important modification of the
pail system. In it dry earth or some other material is added to the
excreta, thus converting them immediately into an inodorous mass.
Probably the best contrivances for thus deodorising the excreta, as
soon as they fall into the receptacle, are Moule’s or Moser’s
Earth-Closets.
It is found that 1½ lbs. of dry earth completely deodorise the
closet each time it is used. Loamy earth is the most valuable
material; a mixture of peat and earth or ashes is very good; sand,
gravel, and chalk are practically useless. It is necessary that the
earth should be very dry, and that it should be finely sifted. If
the earth is damp, decomposition of the excreta speedily occurs.
The act of sitting and rising works a hopper which scatters a
supply of earth.
Charcoal and sawdust have also been used in connection with
Moule’s or Moser’s closet, and with good results. Charcoal has been
obtained cheaply for the purpose from street sweepings, and from
seaweed, as in Stanford’s closet, in which ½ lb. of charcoal from
seaweed is used each time. Mr. Stanford found that while dry clay
absorbs only 4 to 5 per cent. of water, dry charcoal prepared from
seaweed absorbs 14·7 per cent. The best material, however, is
dry earth, but it must be thoroughly dry. The microbes in the earth
disintegrate the excreta, converting them into mineral compounds,
such as nitrates. Even the paper used disappears. Hence the
same earth may be used over again after being stored dry for six
weeks. Whether the excreta of an infectious patient are freed from
infection by this process is doubtful; if not, the infection might be
scattered by means of dust.
The dry earth system is more expensive in use than the pail
system, and although applicable to villages and isolated houses,
is quite unsuited to large towns, owing to the practical difficulties
connected with the procuring and storing of dry earth. The dry
earth closet requires frequent attention, in addition to not being so
convenient as the pail closet; and there is much less manurial
value in the contents of earth closets than in those of pail closets.
The advantages of the earth-closet as compared with the water-closet
have been thus summarised by the late Sir Geo. Buchanan.196
“It is cheaper in the original cost, it is not injured by frost, it is
not damaged by improper substances driven down it, and it very
greatly diminishes the quantity of water required by each household.”
These advantages only accrue when the system is perfectly
worked, and do not counterbalance the immense advantage and
greater safety of the water-carriage system in towns.
The Privy or Midden System, involving the use of a fixed
receptacle, is still prevalent in many towns as well as in innumerable
villages. In its worst form, the receptacle consists of a pit with
sides of porous materials, allowing percolation of filth in every
direction; and in this pit the excreta of whole households are
allowed to collect for months. It has been improved by providing
a cover to keep out the rain, and thus retard decomposition; still
more by providing a drain for the excess of liquid; and by making
the sides and bottom of the pit impervious to moisture. The
addition of dry ashes to the excreta tends still further to prevent
any smell; and the greatest improvement of all consists in raising
the receptacle above the ground level, and providing for easy
cleaning from the back. The raising of the receptacle involves a
diminution in its size, and so prevents the retention of putrefying
matters near a house for a long time.
The model Bye-laws of the Local Government Board recommend
a capacity for the privy not exceeding 8 cubic-feet, the
provision of means for the frequent application of ashes, dust, or
dry refuse; they forbid any connection between the privy and the
drain; insist on its being at least 6 feet from a dwelling-house (too
low a limit); and require a flagged or asphalted floor at least 3
inches above the level of the surrounding ground.
The Nottingham tub-closet forms a link between the pail and
midden system. It is really a small movable middenstead, used for
receiving excreta, vegetables and ashes.
Even when carefully supervised, middens are almost certain
to be productive of evil. They possess two great disadvantages
as compared with pails or dry closets. (1) The time between
collections of excreta by the scavengers is much longer; and (2)
the receptacle for the refuse is part of the structure of the building,
and cannot easily be renewed when it has become saturated with
excreta.
The use of pails or dry-earth closets is a great improvement on
the old middens, but even these compare very unfavourably with
water-closets in two respects. (1) The excreta require to be
retained about the house for a longer or shorter period, whereas
with an efficient water-carriage system, they are at once projected
into the sewer. (2) In removing the excreta, the weight of the
receptacle has to be added to that of the excreta, while in the
water-carriage system, the water serves as the means of transport.
In villages and isolated houses, where no drains are provided
for waste water, and the dry system of closets is adopted, the
disposal of waste water requires special provision. Very
commonly the slops are thrown out of the door, and soak into the
ground about the house. They should be carried by means of a
waste-pipe into a water-tight cesspool, remote from the house,197
whence they can be pumped into a field, or carried away by special
conduits.
Relative Merits of Dry and Wet Methods. No absolute
answer can be given in exclusive favour of either plan. Each is
the best under different circumstances; the dry method being
chiefly suitable for small villages, and for temporary collections of
people, as in camps; and the wet method for towns. The question
of value of manure does not enter into the problem, as it seldom
repays for carriage.
The objections to the water-carriage system are really
due to its not being carried out in an efficient manner. When
sewers are properly laid; when they, as well as house-drains, are
freely ventilated; when house-drains are efficiently trapped and
ventilated near their junction with the sewer; when the drains are
efficiently flushed, and the outflow from the sewer is unimpeded,
the objections disappear.
These objections are that—(1) the sewers, as underground
channels, transfer effluvia and the germs of disease from one place
to another; (2) pipes become disjointed owing to being badly laid,
and the ground is contaminated; (3) the water supply is in danger
of receiving impurities from the sewers. These objections do not
hold good in practice. The contamination of water-mains or of
wells from sewers implies gross carelessness in the method of laying
of sewers or pipes.
The only objections which are of any force, are (4) that water-closets
require a large amount of water, and the sewage obtained is
greatly diluted, and consequently diminished in value; while (5)
the disposal of such an amount of water, in the case of a large inland
town, is a problem of the utmost difficulty. Modern engineering
enterprise by bringing water from a greater distance, and by aiding
the discharge of sewage when necessary by pumping, has overcome
these difficulties.
There are many objections to the dry methods of removing
excreta. (1) Whatever dry method be adopted, the excreta are
retained for some time in or about the house, instead of being
immediately removed.
(2) Although the initial outlay in closets and sewers is less than
with the water-carriage system, there is the constantly recurring
expense of removing the excreta, as well as of cleansing the
pails, etc.
(3) In the dry-earth closets, the provision of dry earth or other
material involves some expense.
(4) Whatever dry method be adopted, sewers are always
required to carry off the foul water, as well as liquid trade products,
and a certain proportion at least of the urine. It is impossible to
supply sufficient dry earth to absorb all the urine and slops of the
population.
Thus, as the Indian Army Sanitary Commission said, speaking
of barracks, “to have two systems of cleansing stations—a foul-water
system, and a dry-earth system—would simply be paying
double where one payment would answer; or, if all the excreta,198
solid and liquid, are to be carried away, this must be done at a cost
ten times greater than that which would be necessary, if all the
excreta were removed by drains.”
With some of the dry methods, as where middens or cesspits
are drained into the sewers, the sewer-water is more offensive than
in towns supplied with water-closets. When a midden or cesspool
is drained, the principle of conservation, which distinguishes the
dry system from the wet, is practically abandoned; and not only so,
but the solid matters still remain to be disposed of, by a tedious
process.
(5) The dry systems, involving the retention of excreta about
the house, poison the atmosphere. In all towns where the refuse
matters are not removed immediately, there is a high mortality,
especially among children.
On the other hand, the introduction of the water-carriage system
into large towns, with the abolition of midden-heaps and cesspools,
has been followed in nearly every case by a diminution in the
death-rate, and especially a considerable diminution in that from such
diseases as enteric fever. It has furthermore increased the comfort
of life, and removed those serious nuisances which are inevitably
associated with privies and pail closets, and to a less extent, when
care is not exercised, with earth closets.
HOUSE REFUSE.
In an ordinary household the disposal of ashes from fires, of
broken pots and cans, of waste-paper, and of vegetable and animal
debris form a serious difficulty. The difficulty is one that can be
minimised by the careful housekeeper. Old newspapers, etc., may
be sold, though their value is very small; other waste-paper should
be burnt. All vegetable and animal debris should be burnt. This
may be effected without nuisance if coal-fires are in use, by placing
potato-peelings, cabbage leaves and similar substances under the
fire until thoroughly dried, and then burning them. The careful
housewife will not waste bones, but utilise them for soup. After
being boiled they are much less liable to putrefy in the dust-bin;
but should even now be burnt in the fire. If this plan be pursued,
the contents of the dust-bin will be simply ashes, broken pots and
cans, and a few cinders—here again a sifter is desirable—and no
nuisance can arise. It is only organic refuse that smells. If only gas
or paraffin stoves are in use, as during the summer months, any
possible nuisance in connection with the dust-bin is minimised by
allowing all refuse to dry before it is placed in the dust-bin, or by
wrapping all putrefiable substance inside several layers of
newspaper.
In emptying the dust-bin or ash pit, care must be taken that the
bottom is thoroughly scraped out. It is well to keep some quicklime
(thoroughly dry) for sprinkling on the bottom and sides of the
receptacle each time after it is emptied. This greatly helps in
keeping it dry and diminishing nuisance during summer.
199
In many households a separate receptacle is kept for what is
known as “hog-wash,” containing waste-food, often in a foul and
putrefying condition. In well-ordered households, except in hotels
and similar establishments, there is no necessity for a “hog-wash”
tub, and its presence argues wastefulness and carelessness. Food
which cannot be eaten because it has “gone bad” should be
burnt.
Two forms of receptacle are used for house-refuse, an ash-pit or
a dust-bin. An ash-pit is a fixed receptacle for the reception of
house-refuse. In many towns the same receptacle is used for
excreta. Then we have a privy or privy midden, according to size
(see page 196). Ash-pits for household refuse alone should be small,
so as not to hold more than a week’s refuse. No part should be
below the ground level. The floor and walls should be lined with
impervious smooth cement, and the ash-pit should have a hinged
cover to keep out rain, and a door on one side to facilitate emptying.
The ash-pit should be at least six feet distant from any wall of the
house. Even the best constructed ash-pit is as much worse than a
dust-bin, as a privy is worse than a pail closet. A fixed is always
less easily cleansed than a movable receptacle.
A dust-bin is usually made of galvanized iron with a tight-fitting
lid. This receptacle can be kept clean, and can be carried
without any transference to another tub direct to the cart.
The removal of house refuse constitutes an important part of
municipal work. In most towns it is carried out weekly, sometimes
less frequently, while in some towns removal twice or three times a
week is secured. A daily removal is carried out in a few towns, and
this is by far the best plan, as decomposition and the dangers
associated with it have then no chance of becoming serious. The
house refuse should always be conveyed through the streets in
covered carts.
The disposal of house refuse constitutes a problem of
increasing difficulty. Unfortunately in the suburbs of many towns
it is deposited on low-lying land in disused quarries and brickfields.
When land has been thus levelled, it often next appears as “an
eligible building site.” A very common practice has been to
excavate gravel and sand upon the site of proposed dwellings, and
allow the excavation to be filled with dust-bin refuse. Before
building on such a soil it is necessary to excavate down to the
virgin earth, and to render it impervious by a layer of cement
concrete.
A second method is to sift and sort the refuse, separating by
means of sieves the finer ash and dust from the coarser parts. This
is usually carried out in a large dust-yard adjoining a river
or railway-siding. The “breeze,” consisting of cinders and
coals, along with the fine ash, are sold to brickmakers; the
“hard core,” consisting of clinkers, broken crockery, etc., is used
for road making; and the “soft core,” consisting of animal and
vegetable refuse, to which is often added stable manure, is sold for
manure. Iron, tin, paper, rags, bottles, and corks are separately200
collected and sold. This disgusting process, often carried on by
women, is now gradually being disused.
A third method is to cremate the house refuse. This has
been done to a large extent by burning the house refuse for
making bricks (page 124). This method of slow and imperfect
combustion necessarily involves a nuisance. A more elaborate
means of securing the same end is by the modern Destructor,
which has been gradually brought towards perfection. A
destructor is a large furnace, in which, after the fire has been
first lit, the combustible matter in the house refuse suffices
to keep it alight. Various mechanical devices are in use for
emptying the trucks of house refuse on to the fires without handling
it, for clearing out of the fire the inorganic refuse, and for ensuring
sufficiency of draught. The amount of draught has in the older
destructors been dependent upon the height of the chimney. In
some more recent destructors the same end has been more efficiently
secured by injecting a steam blast into the furnaces. A temperature
of about 2,000° F. is reached in certain parts of the destructor,
the rapid draught ensuring enormous heat. In view of the
possibility of a portion of the smoke not being completely burnt, a
second “fume cremator” is often provided, through which the
products of combustion in the furnace are passed. The fuel in the
“fume cremator” is coke. Besides incomplete combustion of
combustible material, which is rare when the fume cremator is
provided, the escape of fine dust up the chimney requires to be
guarded against. This is partially prevented by ledges near the
bottom of the chimney. In a destructor the house refuse is reduced
to about one-third of its original bulk, the residue being innocuous
clinker, metallic refuse, and dust. This material can be utilised for
making roads, and in the manufacture of mortar. The waste heat
of the destructor has been partially utilised for various purposes.
This method of disposal of house refuse is usually the best available
for large towns, and offers the additional advantage that no nuisance
is caused by the deposit of offensive material in neighbouring
districts.
201
Lord Bacon said: He who builds a fair house upon an ill seat
committeth himself to prison.” The first considerations, therefore,
in choosing a house are those of aspect, surrounding objects, and
soil. On the first of these considerations, that of aspect,
Thomas Fuller’s quaint remarks give the essential points. He
says:
“Light (God’s eldest daughter) is a principal beauty in a building; yet it
shines not alike from all parts of heaven. An east window welcomes the beams
of the sun before they are of strength to do any harm, and is offensive to none
but a sluggard. A south window in summer is a chimney with a fire in it, and
needs the screen of a curtain. In a west window in summer towards night the
sun grows low and even familiar, with more light than delight. A north window
is best for butteries and cellars, where the beer will not be sour from the sun
shining on it.”
A workroom or study requiring steady light, should point north
or some point between north-east and north-west. A breakfast
room should face north-east to south; while one aspect of a drawing-room
should be south-east to north-east. Store-rooms, dairies,
larders, should have a northerly aspect. It is preferable, as a rule,
for the house not to face in the direction of the four points of the
compass, but diagonally to these.
Surrounding Objects of an objectionable character, as
factories, noisy or offensive trades are to be avoided. The possibility
of neighbouring cesspools contaminating the water supply must
be considered. Trees close to a house are objectionable, rendering
it damp, and preventing the free access of sun and air. More
remote from the house they form a useful shelter, especially when
to the north or east.
The banks of water courses are to be avoided for similar
reasons. If there is a choice, the slope of a hill should be selected;
and it is essential that no part of the dwelling should rest against
sloping ground at a higher level. Rank vegetation indicates a
damp clayey soil.
The main point is to secure that the house shall receive
ample light and ventilation. In calculating the amount and
intensity of sunshine which a house built on a given site will
secure the variations according to season must be remembered.
The direction (orientation) of the sun is the same all the year
through; but the altitude of the sun varies with the latitude.
Thus in a house facing directly south in the latitude of the south
of England the sun’s altitude at noon on the 21st of December is202
15° 4´, on the 21st of June 62° 4´. A ray of light entering the
highest point of a window facing south at each of these seasons
will illuminate a much larger part of the room in summer than
in winter. Not only so, but inasmuch as it enters the room more
nearly vertically it is more powerful than when entering at an angle
more nearly approximating a horizontal direction, in accordance
with the general law that the intensity of illumination falling on a
horizontal surface (as the floor of a room) is inversely as the
square of the width of the area embraced within the same angle
of incidence of light.
In houses in a street the angular aperture through which
light enters is greatest in the upper stories. It may be increased
(a) by increasing the height of rooms; (b) by carrying the window
heads nearly to the level of the ceiling; and (c) by avoiding the
proximity of other buildings which would impede the access of
light. Fig. 36 shows the importance of the last consideration.
This represents a three-storied house in a street, of which the
opposite house L is of the same height. It will be observed that
each room is divided into two regions of different degrees of
illumination by a plane Lm, formed by a line connecting the ridge
of the roofs of the houses on the opposite side of the street with
the interior surface of the rooms and touching the uppermost point
of the window in transit. Below this line there is “sky-light”
sufficient in quantity; above this line light is insufficient in amount
and is diffused and reflected. The area receiving sufficient light
increases from the ground floor upwards. We have already seen
that its intensity similarly increases in the higher stories, the rays
of light being more nearly vertical in these.
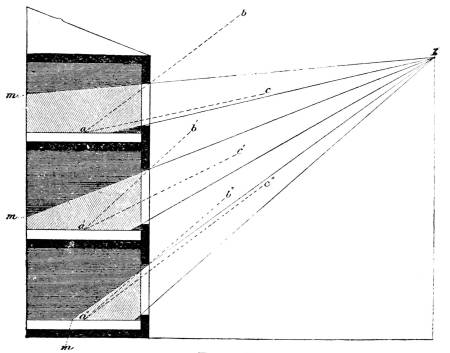
Fig. 36.
Showing Variations of Illumination in Different Stories.
The amount of sky-light visible can be expressed in terms of the angle of
aperture, i.e. the arc of sky visible at any given point a in the room. Thus in203
Fig. 36 the triangle of aperture bac is greater than b´a´c´, and this greater than
b´´a´´c´´. The sides of the angle of aperture, it will be seen, are formed by
drawing one line from the point a to c, which, if prolonged, would touch L, and
another line to b, which passes through the highest point of the window.
The amount of light received in a dwelling-house is largely
determined by the width of the street and the distance between
the backs of the houses
in adjacent streets. The
model Bye-laws of the
Local Government
Board insist that no new
street shall be less than
36 feet wide if it exceeds
100 yards in length or is
intended to be a carriage
road, not less than 24
feet in any case. Furthermore,
a new house
must have in the rear an
open space exclusively
belonging to the house,
at least 150 square feet
in area, and free from
any erection above the
ground level. This must
extend along the entire
width of the house, and
must never measure less
than 10 feet from every
part of the back wall of
the house; the distance
must be at least 15 feet,
if the house is 15 feet
high; 20 feet if 25 feet
high, and 25 feet if 35
feet high or more.
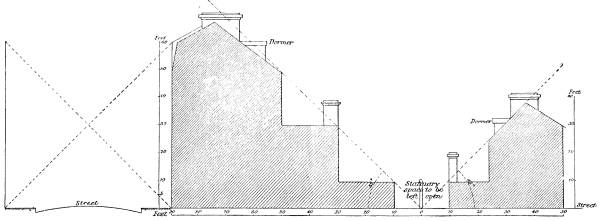
Fig. 37.
Diagram Illustrating the Necessary Requirements as to Open Space in Front of and at the
Rear of Dwelling-houses.
Streets should never
be less in width than
the height of the houses
in them; and a line
drawn from the ridge of
the roof to the foot of
the wall of the opposite
houses (Fig. 37) or in
the rear to the foot of
the wall or fence dividing
the back yards of contiguous
houses, should not make an angle of more than 45º with
the ground (Fig. 37). This is the angle required for new
buildings in the residential parts of Liverpool, and was proposed for
London, but unfortunately not made obligatory. The size of204
windows is discussed on page 216. The light received in a given
house is often diminished at corners of streets by contiguous houses.
The Soil has an important influence on the healthiness of a site.
The relative merits of the different kinds of soil are discussed on
page 219. Undrained soils of whatever kind are bad, and made-soils
are always to be regarded with profound distrust.
The planning of a house should be carefully considered. The
principle is that the sun should enter every living room at some
time of the day. The relative positions of fire-place, window, and
door in each room are important. With the sole window of a room
in the same wall as the fire-place the area ventilated is the least, with
it situated on the opposite wall the area ventilated is the greatest.
The door should be as remote from the window as possible, in order
to secure occasional perflation of air; the two being preferably on
opposite walls. Staircase windows are indispensable to secure
through ventilation of a dwelling. Houses constructed “back to
back” cannot be properly ventilated as no through current of air is
possible. Hence the necessity for open yards at the area, as well as
air-space in front of the house. (Fig. 37).
In the construction of a house, apart from access of light and
air, the main problems are to secure dryness and equability of
temperature. We shall consider in this connection the materials
used in the construction of walls, floors, and roofs.
205
CHAPTER XXXI.
THE MATERIALS USED IN THE CONSTRUCTION
OF A HOUSE.
In this country walls of houses are usually built of brick, stone,
timber, or concrete, of which the first two are the most important.
Timber is, owing to its inflammability, only allowed to be used in
towns under special restrictions. Bricks and stones are bonded
together and imbedded in mortar or cement.
There are several kinds of bonds in brickwork, of which the
strongest is the English. This consists of alternate courses of
“headers” and “stretchers,” the former being bricks carried
through the wall from face to back, the short end showing on the
face, and the latter bricks laid lengthwise along the face of the
wall. Hence the wall is held together in every direction. A
Flemish bond consists of alternate headers and stretchers in the
same course. It is used where a specially smooth wall is desired,
but is not so strong as the English bond.
Bricks are generally of a uniform size, of 9 inches in length by
4½ in width and 2¾ inches in thickness. Those bricks which are
heaviest and hardest are generally the most durable; bricks of good
quality when knocked together give a clear ringing sound.
The relative conductivity for heat of brick as compared with
other materials, is shown in the following table, from Galton, which
gives the units of heat transmitted per square foot per hour by
a plate 1 inch thick, the two surfaces differing in temperature 1°
Fahr.:—
| Stone—ordinary free stone |
13·68 |
| Glass |
6·6 |
| Brickwork |
4·83 |
| Plaster |
3·86 |
| Fir planks |
1·37 |
| Brick dust |
1·33 |
It is evident that in this respect, brick walls compare very
favourably with stone walls, and are much more economical of heat.
Increased conductivity of a material may be counteracted by
increased thickness.
Brick is very porous, as shewn by its power to absorb moisture.
A good brick can absorb from 10 to 20 per cent. of its weight of
water; while good granite only takes up ½ per cent., sandstone206
usually from 8 to 10 per cent., marble only a trace, and Portland
limestone 13½ per cent.
Being porous, brick allows the passage of a considerable amount
of air, unless its pores are occupied by moisture. The following
table, from Galton, shews the number of cubic feet of air which
every hour pass through a square yard of wall-surface of equal
thicknesses, built of the following materials, there being a temperature
of 72° Fahr. on one side the wall, and of 40° on the
other:—
| Wall built of |
brick |
7·9 |
cubic feet. |
| „ |
quarried limestone |
6·5 |
„ |
| „ |
sandstone |
4·7 |
„ |
| „ |
limestone |
10·1 |
„ |
| „ |
mud |
14·4 |
„ |
Mortar should consist of clean sharp sand and slaked lime,
usually in the proportion of three of the former to one of the latter.
Grouting, or liquid mortar, is merely ordinary mortar to which a
larger quantity of water has been added. It is used for filling up
the crevices between the brickwork about every fourth course, and
is required to a greater extent in stone work, owing to the difficulty
in filling up spaces left by inequalities in the stone.
The sand used in mortar should be free from small stones. It
should not contain any earthy or clayey matters, as these greatly
diminish the adhesive quality of the mortar, which depends on the
combination of the sand and lime. All the sand used in a building
should be washed, unless it is perfectly clean, in order to remove
impurities. Many builders use an inferior mortar, in which
other materials, such as “road scrapings,” are substituted for sand.
Sand taken from the sea-shore is unfit for making mortar, as the
salt contained in it is apt to deliquesce and weaken the mortar.
Lime is obtained by burning chalk or limestone in a kiln.
Thus CaCO₃ = CaO + CO₂. There are three kinds of lime: (1)
Fat or quicklime, used for internal plastering, (2) stone lime, used
for ordinary building work, and (3) hydraulic lime, used for building
in damp situations. The last named contains a quantity of silicates,
and sets under water.
Common mortar crumbles away, if laid under water before it
has had time to harden.
Portland cement is an artificial cement, of a dark grey colour.
It is made by grinding chalk, mixing it with blue clay or river-mud
in certain proportions, and then burning it in a kiln and afterwards
grinding it to a fine powder. It is used, mixed with sand, for
external plastering (“compoing”) of walls, for making concrete,
or instead of lime for making mortar if extra strength is required.
Compo consists of Portland cement and sand, and is used for
covering walls when an impervious smooth surface is required, and
for keeping out rain. It is laid on in two coats. The first or rough
coat ¾ inch thick, is composed of one part cement to 5 parts compo207
sand, i.e. coarse sand mixed with fine beach. The outer or fine coat
is composed of two parts fine or washed sand to one part cement.
To “render” or “compo” a wall is to cover it with this material.
The internal plastering of a chimney flue is called “pargetting.”
Concrete is of two kinds, lime or cement concrete. It is
composed of three parts broken ballast or large beach, two parts of
sand, and one part of lime or cement. Lime concrete has no
resisting strength, and is only used for surrounding drain-pipes, or
where no great strength is required.
Stone varies very greatly in character. It is uncommon for
the whole thickness of the walls of a house to be built of stone;
usually there is merely a facing of stone and a backing of
brickwork. If good stone is not available, the less it is used the
better.
The stone chosen should be durable, and able to resist the
action of the sulphuric, sulphurous, and carbonic acids absorbed
from the atmosphere, and brought in contact with it by means of
rain. The stone of which a considerable part of the Houses of
Parliament consists is dolomite, a double carbonate of lime and
magnesia. The acid fumes in the air produce on its surface
sulphate of magnesium, which is washed away in successive layers.
If the stone presents any stratification, it should be laid in the
wall in the same position as that in which it was originally
deposited in the quarry. Thus, any planes of stratification will be
horizontal, and the scaling off by the action of frost and rain is
minimised. Comparatively homogeneous stones, such as granite
and millstone-grit, can be laid in any position. In testing the
character of any stone, the least porous, densest, and most resistent
to crushing, will as a rule be the most durable.
The chief difficulty in the use of stone for the walls of houses, is
that of keeping out the wet. To obviate this, stone-houses are often
built of great thickness, and are consequently cooler in summer and
warmer in winter.
In and near large towns brick is chiefly used for walls of
houses, and stone employed only for window-sills, columns, steps,
etc. It is even more important in these cases to carefully select
the stone, as the parts where it is placed are those most exposed to
the weather. If a soft, friable freestone is used, after a sharp frost
large scales are seen falling off in flakes, owing to the freezing and
subsequent thawing of the moisture in the stone.
Portland stone is the best-wearing stone to be had in the
neighbourhood of London. Bath stone is also considerably used,
but it varies greatly in quality, and should be very carefully
selected. For landing-steps and paving, Yorkshire stone is extensively
used, but artificial cement pavings are replacing it to some
extent. Most kinds of stone can only be economically used near
the quarries from which they are derived.
The Slate used for roofs is an altered form of clay, possessing a
laminated structure. The ease with which it splits along the
planes, renders it peculiarly suitable for this purpose. The Welsh
slates are considered the best.
208
Terra-cotta is made from certain kinds of clay, mixed with
glass, pottery or sand; then ground up, strained, and kneaded; and
lastly thrown into moulds and baked in a kiln.
Iron and Wood have occasionally been employed alone in
building houses. The former, owing to its good conducting powers
for heat, is cold in winter and hot in summer; while the latter
becomes rotten from exposure to wet, and is also very combustible.
Corrugated iron buildings lined with wood are also employed, but
are not very satisfactory.
For roofs, slates or tiles are the materials most frequently
employed; but occasionally lead and corrugated iron are used, also
thatch in country places, and tarred felt for temporary buildings.
Lead is the most suitable metallic covering for roofs, as it is
durable and easily worked. It is, however, heavy and demands
considerable strength in the timbers by which it is supported.
Galvanized iron has also been largely used. It is cheaper and
lighter than lead. Both lead and zinc require very careful laying if
they are to be weather-tight.
Thatch protects the interior of a house well from extremes of
heat and cold.
209
In preparing to build a house, or in entering into a house already
built, the following requisites should each receive careful attention:—
1. The site of the house should be healthy, and its relation to
surrounding objects in accordance with the laws of health. (See
page 201).
2. The house should be warm in winter, and cool in summer.
3. It should be always dry.
4. There should be an abundant and uninterrupted supply
of air.
5. The water supply should be abundant, conveniently arranged,
and pure.
6. The excreta and waste-water should be immediately removed
from the house and its annexa.
The three last requisites have already received consideration.
Of those still to be considered, dryness is the most important.
A damp house is certain to be an unhealthy one. It is this for two
reasons:—1st, it is a cold house, as damp walls, like damp clothes,
conduct the heat of the body away much more rapidly than dry
walls; 2nd, if the pores of the bricks are occupied by water, air
cannot pass through, and thus the ventilation and purification of
the house are greatly impeded. Damp may arise from the ground
on which a house stands, or from the rain beating against the walls,
or from a defective roof. Unless special means are taken to prevent
it, moisture rises by capillary attraction through brick after brick.
The Foundation requires to be solid and substantial, otherwise
sinking occurs, with cracking of the walls, resulting in an unsafe
condition, and an exposure to rain and wind.
In making the foundation for a house, the ground should be
excavated, so as to secure a solid bed of earth or rock not liable to
be affected by the weather. A continuous bed of the best cement
concrete should then be laid, not only under the walls, but covering
the entire site of the house, and extending on every side at least 6
inches beyond the footings of the wall; and for footings it should
never be less than 18 inches thick. The concrete serves two
purposes: it, to a large extent, cuts off the entrance of the ground-air
through the basement floor into the house; and prevents the
entrance of damp into the house from below. To further ensure
dryness where the floor is below the level of the adjacent ground, a210
dry area is frequently provided, that is, a closed chamber lined with
stone or cement below the ground level of the house, and surrounding
the underground part of its four walls, or a hollow wall is built
below the ground-level, as shown in Fig. 38. Neither a dry
area nor a hollow wall constitutes the best arrangement, as the
cavity is usually inaccessible, and rather aids than hinders the
entry of the ground air into the house. The best plan is to
provide a solid wall, impervious to both moisture and air. A
vertical layer of roofing slates is sometimes used for this purpose;
or, still better, a narrow cavity about ½ to ¾-inch wide is provided
in the body of the wall, and this is run full with molten asphalte.
The Walls of the house must be provided with a “damp-proof
course” carried through their whole thickness, slightly above the
highest point at which the
ground is touched. It may
be formed by (1) sheet lead,
which possesses the disadvantage
of being costly;
(2) two layers of ordinary
roofing slate, set in cement,
with broken joints,” i.e.
the joints of the upper layer
over the centre of the slates
below them; (3) a layer of
good asphalte, about ¾-inch
thick; (4) perforated glazed
stoneware slabs; or (5) two
or three courses of hard blue
Staffordshire bricks, laid
without mortar. The use of
asphalte is an excellent plan,
and is now commonly
adopted in good buildings.
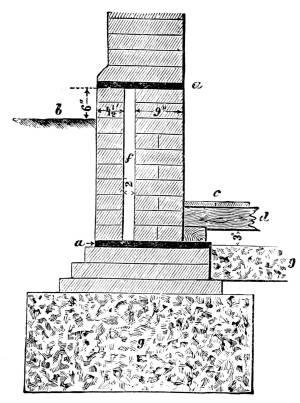
Fig. 38.
Double Damp-Proof Course and Hollow Wall.
a, a—Damp-proof courses. b—Level of
neighbouring ground. c—Floor-boards. d—Floor
joist. f—Vertical space in wall.
g—Concrete under foundation and over site
of house.
This suffices when there
is no basement. If there is
a basement an open area
around the house is desirable.
Where for a given
wall this cannot be secured,
the vertical damp-proof
course already described
must be made to extend from
the foundations well above
the ground level. The open
area, however, should be
insisted on whenever practicable. The damp-proof course protects
the walls from damp proceeding from the soil around or beneath the
house.
It is necessary also that the walls above the level of the ground
should, as far as possible, be kept free from damp. Damp walls,
not due to ascent of moisture, may be caused by
211
(1) Rain falling on window-sills which do not project beyond
the walls, and consequently do not throw the water clear of them.
This is remedied by constructing the window-sills so as to project
beyond the walls, and “throating” them to prevent rain from
running along the bottom of the sill. The throat is shown at
a, Fig. 39.
(2) Rain falling on cornices and other projecting portions of the
wall itself. The evil from this source may be diminished by
sloping the top of the projection, downwards from the face of the
wall.
(3) Parapet walls, gables, etc., not being properly covered with
coping. All such walls should be topped with a projecting slab of
stone, or with a damp-proof course under the top course of bricks,
which should be laid on edge.
(4) Overflow from defective roof-gutters or rain-water pipes. In
this case, either clearing out, repairing,
or renewing is required.
(5) Rain beating against the
walls. This as a rule produces no
great harm, if the walls are well
constructed. Most of the water runs
off as it falls on the surface. It
is advisable, however, to protect a
much exposed wall by a coating
of Portland cement, or in extreme
cases with slate. Various impervious
paints have also been
employed.
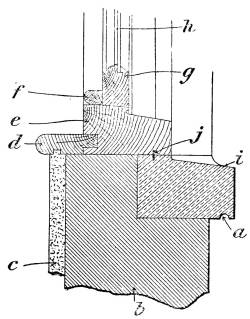
Fig. 39.
Section Through Window.
Showing stone sill “weathered”
at i, and “throated” at a. b—Wall.
c—Inside plaster of room. d—Window-board.
e—Oak sill. f—Beading
and g—bottom rail of window-sash.
h—Window. j—Iron tongue let into
slot in i and e to prevent rain driving
in.
If it is not proposed to coat
exposed surfaces of brickwork, the
wall may be formed of two parallel
walls, two inches apart, and tied
together by a sufficient number
of bonding-ties of iron or glazed
stoneware, or some other non-absorbent
material. This arrangement
is shown in Fig. 40.
An excellent plan is to fill in
the narrow space between two such
walls, as the building proceeds,
with asphalte or slab slate, thus
forming a vertical damp course, in the
same way as below the ground level. The evils arising from
damp can be avoided in every new house by proper methods of
construction. In an old house, however, they are much more
difficult to remove. The dampness is indicated on entering, by
a peculiar mouldy smell, and by the discolouration and destruction
of wall-papers, and dry rotting of floor timbers. In such a case
a damp course may, with care and patience, be inserted in the
wall, and the soil under the basement may be covered with concrete,212
and a dry-area excavated around the basement. Free ventilation
under the floor-boards of the lower floors also helps in keeping
the house dry.
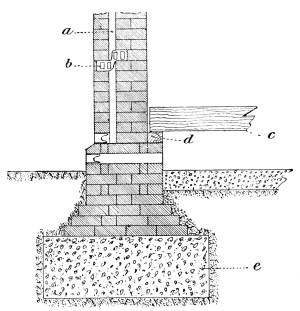
Fig. 40.
Showing Hollow Wall and Bonding-ties
of Glazed Stoneware.
a—Cavity. b—Tie. c—Floor-joist. d—Wall-plate.
e—Concrete foundation of wall.
The thickness of the walls of a house requires to be sufficient to
ensure stability, to keep
out the damp, and to
prevent a too rapid loss of
heat from the walls. The
relative merits of the
different materials employed
for these purposes
have been already considered.
A thin-walled
house is hot in summer,
and cold in winter. The
upper stories of houses
are often built with too
thin walls, the result being
chilly bedrooms. A single-brick
wall (9 inches thick)
will rarely keep out the
weather effectually, and
frequently a brick-and-a-half
wall (14 inches thick)
is insufficient for this
purpose. The bricks
should be so interlaced as
to “bond” or tie the wall
together in all directions.
The strength of walls may be increased by the introduction of
hoop-iron between the courses of brickwork.
In the construction of fire-places and chimneys, it is important
to avoid the proximity of timber and wood-work to the inside of
flues, as this is a common cause of fires.
Inside Coverings of Walls.
Plaster is made of lime mortar, or cement mortar; the former
is generally preferred for domestic dwellings because it remains
porous and moisture does not condense on it.
In houses built by speculative builders, the plaster commonly
used consists of a mixture of lime with road scrapings. The result
is a composition which unless supported by the wall-papering, is
soon damaged.
Ordinary plaster consists usually of three layers. The first is
laid on with a mixture of about equal parts of lime and sand with
long ox-hairs if required for ceilings. The second coat consists of
slaked lime, mixed to the consistency of cream. The last or setting
coat consists of a thin layer of slaked lime called plasterers’ putty.
Some plaster of Paris (gypsum) may be added, to ensure rapid setting,213
but it should only be used in small quantities. For the internal
plastering of rooms serapite (a form of cement) is now commonly
employed. This is not so absorbent as mortar, but is sufficiently so
to prevent condensation of moisture on the walls. Its chief
advantage over plaster is that it hardens quicker and is smoother,
and can be used in a single thin layer. This, however, diminishes
the impermeability of ceilings for sound.
Keene’s cement and Parian cement are mixtures of calcined
gypsum and other substances; Keene’s cement being the hardest,
and capable of receiving a high polish.
Selenitic cement contains a small proportion of plaster of Paris
ground along with lime. Lime may also be selenised by the addition
of any other sulphate, or of sulphuric acid. The presence of the
sulphate causes the lime to set rapidly. Selenitic cement is useful
in plastering, as a backing of cements, such as Parian.
The treatment of the internal wall-surface of a room differs
according to circumstances. Lime-washing is suitable only for
stables and other outbuildings. It is made by the addition of water
to quicklime, no size being added. It is an excellent germicide and
insecticide. Whitewashing is quite different from limewashing.
“Whiting,” i.e. finely-ground chalk, to which a certain proportion
of size and alum had been added is mixed with water. The size and
alum are added to prevent the whitewash from being rubbed off.
Distempering is identical with whitewashing, except that pigments
are added. It is distinguished from painting in oils, by the fact
that the pigments are mixed with size, instead of with linseed-oil and
turpentine. Painting in distemper is practically limited to plaster,
which should first receive a coat of whitewash to diminish its
porosity. Oil-paints are impervious, distemper is as absorbent as
plaster or whitewash. Various washable distempers, as duresco,
are made, which are more durable and non-absorbent. Water-glass
consists of silicate of potash, which in the gelatinous form is
soluble like size in hot-water, but when allowed to dry forms an
impervious film. It can be used for protecting porous stone from
the effects of weather; and renders internal surfaces of walls non-absorbent
and washable.
Oil-painting renders wall-surfaces impervious, and enables
them to be easily washed. The importance of this in the event of
any infectious disease occurring, is obvious. The question arises
whether distempered or papered walls, which are porous, or painted
walls, which are non-porous, are preferable from the standpoint of
health. The difference between the two is seen during damp
weather, when moisture condenses and runs down the latter and is
invisible in the former. In practice in domestic dwellings the former
are preferred; but although some advantage is thus secured in
ventilation through the wall-substance, there is the serious
disadvantage that particles of dirt accumulate and may seriously
interfere with the purity of the air of a room. Hence the
importance of rubbing down the internal surface of a room, whether
distempered or papered, at intervals with bread crumb or dough
(see page 332). This effectually removes all accumulations of dirt.214
A painted wall presents the enormous advantage that it can be
frequently washed; while the loss of ventilation may be ignored, if
windows and doors be properly utilised for this purpose. The
presence of poisonous pigments in oil-paints is of importance to the
workman, but not to the householder except during the painting, as
paint, unlike distemper, does not rub off the wall. Lead is the chief
poison present, as white lead (carbonate of lead). Various
substitutes for lead paints have been introduced.
Painting wood or iron-work is valuable, not only as a preservative
from the effects of the weather and the oxidising action of
the air, but also because it tends, to a large extent, to prevent the
absorption of organic matters; and its surface can be frequently
cleansed.
Paper is the material most commonly employed for covering
walls. It is more absorbent and retentive of moisture than
distemper.
Light-coloured papers should be chosen, as they are more
cheerful, and are not so likely to harbour dust. Glaring patterns
are objectionable, as they tire the eyes. The paper should not
present any surface-projection for the lodgment of dust.
In bathrooms and water-closets, the wall-surface should be
non-absorbent. Paper, unless varnished, should therefore be
avoided. The best covering for these places is glazed tiling, or
painted cement.
Not uncommonly, a new paper is pasted over an old one; and
this may be repeated several times. Under these circumstances
dangerous dirt accumulates. Before new papering is put on, the
walls should be cleared of all vestiges of the old, thoroughly washed
down, and subsequently coated with size (that is, “clear coloured”).
The sizing diminishes the absorptive power of the wall, and gives a
good surface for applying the paper.
Bed-room papers require to be more frequently changed than
those of other rooms. Bed-rooms in regular use should be
re-papered at least every two years. It is still better to use
distemper for such rooms, as this can be washed off in a few hours
with comparatively little expense, and can be made of any tint
desired.
Rooms in the basement should not be papered, as the walls
require frequent washing down and cleaning. Here also a washable
distemper colour can be used.
Various kinds of sanitary paper are now sold which are
washable, and relatively non-absorbent. Some of them require
varnishing; others do not. Such papers are certainly cleaner than
ordinary paper; but it would not be safe to trust to their non-absorptive
character. Lincrusta Walton is non-absorbent, and can
be scrubbed with soap and water; but it is expensive. Other
cheaper materials possessing the same properties can now be
bought.
Arsenic in Wall-Papers and Paints has until a few years ago
been a not uncommon source of prolonged ill-health—the cause of
which has possibly not been detected until the illness disappears,215
when the offending room is vacated for a period. Arsenical
pigments are now only rarely used for wall-papers. The symptoms
produced vary greatly, and may closely simulate those of different
diseases. In some cases repeated attacks of diarrhœa and abdominal
pain occur. Or there may be nausea, headache, frequent griping
pains, and loss of appetite. In other cases restlessness, loss
of sleep, and general malaise are the chief symptoms, with the
occasional addition of conjunctivitis (superficial inflammation of
the eye). Out of 100 cases collected and reported on by a
Committee of the Medical Society of London, diarrhœa, nausea,
and intestinal mischief occurred in 85; severe depression in 16;
conjunctivitis in 19; and cough, asthma, etc., in 9.
The severity of the symptoms produced will vary with the
amount of arsenic contained in the paper, and the length of time
daily that the patient is exposed to the fumes.
Some persons again are much less susceptible to the influence
of arsenic than others. This will explain why some escape
while occupying the same room in which others suffer severely.
More commonly, however, the exemption is due to shorter
exposure.
The most dangerous preparation occasionally employed in wall
paper printing is Scheele’s green (arsenite of copper). Emerald-green—an
aceto-arsenite of copper—is sometimes used to produce
more delicate tints. Aniline dyes, especially the red, may contain
much arsenious acid (white arsenic). The arsenic compound is
made to adhere to the paper by size or some other material. When
dry, it cracks and peels off, and minute particles get into the air as
dust. In addition, arsenic compounds easily volatilise, and become
diffused in a gaseous condition throughout the atmosphere of a
room, even when its temperature is not greatly raised. The
virulence of the arsenical colouring is in proportion to its volatility.
Arsenic seems to be much more dangerous when associated with
size. It has been shown that a mixture of white arsenic and starch
paste, or other organic substance, leads to the formation of gaseous
arseniuretted hydrogen, while this does not occur when no organic
matter is present (Dr. Fleck). Distemper frequently contains
arsenic, and as it also contains size, arseniurretted hydrogen is liable
to be given off at any time. Size is largely used for fixing colour;
thus, the proper conditions for the development of arseniurretted
hydrogen—the most dangerous compound of arsenic—are present.
As much as 17 grains of arsenic have been discovered in each
square foot of a wall-paper. Now, arsenic is sometimes given
internally for certain skin and other diseases, but the dose is only
from 1 ∕ 60 to 1 ∕ 12 grain; the capacity for poisoning of such a paper as
the above will therefore be evident.
Papers of other colours than green have been found to contain
dangerous quantities of arsenic; thus blue, mauve, red, and brown
may contain large quantities; the delicate greys often yield a
considerable amount, and some white papers are heavily loaded
with it. Arsenic is occasionally present in stockings and other216
wearing apparel, artificial flowers, toys, etc. In these cases, it may
produce irritation of the skin, and even eczema.
The presence of arsenic may be detected by the following
tests:—
(a) Reinsch’s test. A portion of the suspected paper (two or three inches
square) is cut into small pieces, and placed in a good-sized test tube; water is
added until the tube is about a third full and then one or two teaspoonfuls of pure
hydrochloric acid, and a small piece of pure copper foil. If the test tube is now
heated for a few minutes over a spirit lamp, arsenic, if present, will be deposited
as a black or dark steel-coloured coating on the copper. A mere tarnish of the
copper must not be accepted as evidence of the presence of arsenic, but an
almost complete obliteration of the colour of the copper.
(b) Take the copper covered with arsenic, dry it, and then heat it in a
perfectly dry test tube. Crystals of white arsenic, which may be identified under
the microscope, will be deposited higher up in the tube.
(c) Marsh’s test. The ordinary apparatus for developing hydrogen by the
action of diluted sulphuric acid on zinc is employed, the suspected paper being
inserted in the bottle. The hydrogen coming off is burnt, and a clean porcelain
surface is applied to the flame. If there is arsenic in it, it is deposited on the
porcelain in a black patch.
Windows are required to open directly into the external air in
every habitable room. The window area according to the model
bye-laws of the Local Government Board and the London Buildings
Act of 1894, must be at least one-tenth of the floor area, and half
of this at least must be made to open. The following rules have
also been given. (B = breadth, L = length and H = height of room.)
| Area of window |
 |
(B × L) ∕ 10 |
London Building Act |
| (B × L × H) ∕ 100 |
Gwilt |
| √(B × L × H) |
Morris |
In a room measuring 15 × 20 × 12 feet, the preceding rules
would give a superficial area of window space of 30, 36, and 60
square feet respectively. Plate glass dissipates heat less quickly
than sheet glass.
Objection may be taken to plate glass windows, in passing,
especially for shops, banks, etc., in view of the fact that they are
commonly made without any arrangement for ventilation (see also
page 148).
The hygienic necessities of Floors are that they shall be
impervious to moisture and to dust. On the ground floor the
ordinary arrangement is to provide a joisted and boarded floor
raised about a foot above the ground. Dry rot is one of the dangers
in connection with such boarded floors on the ground floor. The
chief causes which tend to induce rotting, are damp walls, lack
of ventilation, contact with mortar, damp earth, or vegetable mould,
and worst of all, alternations of damp and dryness, or wet along with
heat.
In order to avoid these dangers in connection with boarded
floors, the ends of all timbers resting on walls should have a clear
air-space around them, and communicate with the external air by217
means of perforated bricks. The larger timbers, girders, etc.,
should rest on stone templates, and the smaller joists on hoop-iron
bonds. In all cases, the timber used should be well seasoned, and
properly ventilated. The ends of oak posts, which are to be
driven into the ground, should be charred, if the timber is old,
or steeped in a solution of chloride of zinc.
The ends of the joists should be trimmed, so as not to come too
near to chimney flues.
The best plan for flooring is to place an impervious flooring
resting on the solid ground. This is more secure against rot than
the boarded floor, and affords no space for dirt and vermin to lodge.
Such an impervious floor may be formed of concrete over a layer of
asphalte, as in the well-known terrazzo flooring. This is very
suitable for corridors, pantries, etc. For living-rooms wood-block
flooring is placed over the cement, molten pitch connecting the
two. The blocks are 2 to 3 inches thick. If the wood is soft, as
deal, it must be kept clean by washing; if hard, as oak or teak,
it can be wax-polished. Parquetry consists of small pieces of hard
woods carefully fixed and polished.
For upper floors the ordinary flooring is of floor-boards
supported on wood joists, beneath which are wood laths and plaster.
The floor-boards should be thoroughly seasoned, otherwise they will
shrink, and the joints be filled with dirt. This dirt may accumulate
for years between the floor
and the ceiling of the room
below, vitiating the air and
helping to increase the
stuffiness characteristic of
dirty houses. Various plans
are adopted for uniting the
edges of floor-boards and
preventing dust from dropping
between the boards.
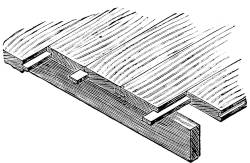
Fig. 41.
Tongued Floor.
The one most commonly
employed is the
ploughed and tongued floor
(Fig. 41). In this, both edges
of the floor are grooved so as to receive strips or tongues of iron or
wood, an equal half of each strip being in the groove of each of two
boards when they are in place. A less expensive method than
the above is to splay the ends of the boards so that they slightly
overlap each other. This is not so efficient as the above, but is
much better than simply placing the boards side to side as is
commonly done.
Solid wood floors resting on a bed of concrete are free from the
risk of harbouring dust, and are relatively fire-proof.
Oak or teak in narrow boards, made with close joints, and then
oiled and beeswaxed and rubbed to a polish, makes a good and
almost non-absorptive floor. One of the best floors is made of
concrete, with iron joists, and oak boards laid above this.
218
Carpets are commonly made to cover the entire floor of rooms.
This cannot be too much deprecated. Carpets, like curtains, are
mere dirt-traps, which become loaded with filth of every description.
This is abundantly proved when a carpet is swept, and the
dust allowed to settle on all the articles in the room. Such dust, if
examined, will be found to consist not only of mineral matter, but
also of every description of vegetable and animal impurities. The
inhalation of such dust, which may contain particles of fæcal
matter, as well as the dried expectoration from consumptive or
other infectious patients, is a not infrequent cause of infection to
healthy persons.
The substitution of a central carpet, for one covering the entire
floor, is a great improvement.
The carpet should be easily removable, in order that it and the
floor may be thoroughly cleaned at intervals.
In bedrooms, the less carpet the better. Good Chinese or
Indian matting is serviceable, as it does not retain the dust and
other impurities which are apt to become fixed in the woolly
texture of the carpet. Oil-cloth, linoleum, and similar materials
are in common use for covering halls, passages, etc. They are
particularly useful in preventing dust from gaining access to the
spaces between floor-boards.
The prevention of dust should be the great aim of the
householder, as dirt frequently carries infection. Sweeping as
ordinarily done scatters dirt over the room, and dusting with a dry
cloth fails to remove it. Mechanical sweepers, in which the dirt is
collected in a box are valuable. The best plan is to have movable
carpets, roll them up for shaking or beating at a distance from any
house, and wipe the boards with damp cloths. All wooden and
leather furniture, picture frames, etc., should be wiped down with
cloths rung out of water so as to be just damp.
219
The Varieties of Soil.—The following facts summarise what
is regarded as the relative healthiness of various sites for
dwellings. The differences between different sites may, however, be
reduced to a minimum by having the dwelling well above the
ground-level and by protecting it from dampness.
1. Granitic, Metamorphic, and Trap Rocks usually form
healthy sites for houses. The slope is generally great, and the
ground consequently dry.
2. Clay Slate resembles the last in its effects on health.
Water is, however, often scarce, owing to the impermeability of the
rocks, and for the same reason occasional floods occur.
3. Limestone and Magnesian Limestone Rocks resemble
the last in possessing considerable slope, so that the water passes
away quickly. The hard oolite is the best formation under this
head, and magnesian limestone the worst.
4. Chalk is a healthy soil when unmixed with clay, and
permeable. Goitre is not so common as in limestone districts. If
the chalk be mixed with clay, it is often damp and cold.
5. The Sandstones are healthy, soil and air being dry. If
mixed with clay, or if clay lie under a shallow layer of sand-rock,
the site may be damp. The hard millstone grit is a healthy
formation.
6. Gravels of any depth are healthy, unless they are water-logged,
as near rivers. Then a house on impervious clay may be
drier than one on gravel.
7. Sands are healthy when of considerable depth; they may
be unhealthy when shallow, and lying on a clay basis; or when the
ground water rises through them from ground at a higher level.
8. Clay, Dense Marls, and Alluvial Soils generally, are
apt to be cold and damp. Water is retained in them, and is often
very impure. Thorough drainage improves a clay soil, and a house
on a clay soil may be so constructed, as not to be damp.
9. Cultivated Soils are not necessarily unhealthy; but
10. Made Soils are always to be carefully avoided, as sites for
houses. The materials with which inequalities have been filled up
are commonly the contents of dust-bins, or some other refuse. The
gradual putrefaction of organic matters renders the air about the
houses impure. Such soils require free subsoil drainage, in order220
to keep them dry. It appears that the organic matters in soil are
gradually removed by oxidation and bacterial purification. At least
three years should be allowed before any such site is built on.
The following table places different geological formations in
their order of healthiness for the purposes of a site (Parkes):—
|
PERMEABILITY
OF WATER. |
EMANATIONS
INTO AIR. |
| 1. Primitive rocks, clay slate, millstone grit |
Slight. |
None. |
| 2. Gravel and loose sands, with permeable subsoils |
Great. |
Slight. |
| 3. Sandstones |
Variable. |
Slight. |
| 4. Limestones |
Moderate. |
— |
| 5. Sands with impermeable subsoils |
Arrested by subsoils. |
Considerable. |
| 6. Clays, marls, alluvial soils |
Slight. |
Considerable. |
| 7. Marshes, when not peaty |
Slight. |
Considerable. |
The general geological conditions have an important bearing on
the choice of a site for a house in so far as they affect the local
climate, and the difficulty of keeping the house warm and dry.
Pettenkofer expressed this in his dictum, that we take holiday for
change of soil, rather than for change of air. The character of a
soil has an important influence on humidity, radiation, evaporation,
and in fact most of the factors going to make up “climate.” The
immediate local surroundings of a house (page 201) have an even
greater influence on its salubrity than the underlying geological
formation.
The soil consists of mineral and organic matters. On the
amount and character of the animal and vegetable matters (along
with the condition of moisture and aeration), the healthiness of a
given soil depends. The presence of vegetable matter, subject to
alternate wettings and dryings, and to heat, has until recently been
regarded as the condition on which malaria depends; but it is now
known that malarial places owe their character to their being
favourable to the growth of the larvæ of certain mosquitoes (page
307); and that drainage of the soil cures malaria by removing the
ponds in which these develop. The two chief agencies at work to
rid the soil of organic impurities, are nitrification and the influence
of growing plants. The organic matters become oxidised into
ammonia, nitrites, and nitrates, and these are eagerly assimilated by
vegetation.
Nitrification is effected by micro-organisms in the soil.
Ordinary garden mould and agricultural humus contain large
numbers of micro-organisms. Their number diminishes with the
depth of the soil, and below 12 to 15 feet there are few. Apart from
the occasional presence of pathogenic (disease-producing) micro-organisms,
the most important are those producing oxidation of
organic matter, especially nitrification. This occurs at a less depth
than 4 feet from the surface of the ground. The operation of these
micro-organisms is necessary to convert sewage and other impurities
into harmless nitrites and nitrates, and it is regularly going on in all221
normal soils. That the power of purification of sewage by soil is
due to the micro-organisms in the latter, can be proved by the fact
that when the soil is baked, it loses for a time its purifying power.
The Air contained in the Soil varies greatly in amount
with the character of the soil, and with the level of the ground-water.
As the ground-water rises, the ground-air is driven out.
Thus, after a heavy rainfall a large proportion of this air will be
displaced. Variations in barometric pressure, and a rise or fall
of temperature, cause movements in ground-air. A house artificially
warmed is liable to receive air from underground, unless
means are adopted to make the floors impervious. The warmth of
the house acts as an air-pump, aspirating the colder air into its
interior. The air from cesspools or defective drains may be
similarly aspirated into the
house; and the same cause
particularly explains the
unhealthiness of houses
built on “made soils”.
Coal gas has occasionally
made its way into houses
when not laid on to them,
by the gas escaping from
leaky pipes in the street
often following the track of
water or drain-pipes until it
is aspirated from beneath
the house into its interior.
This has resulted in one
instance in an explosion, and
in others in poisoning by
the gas.
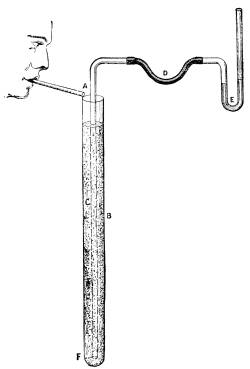
Fig. 42.
The occurrence of currents
of air in soil may be
illustrated by a simple
experiment. In Fig. 42 B
is filled with fine sand in
which is imbedded the tube
A with its open end F at
the bottom of the sand C.
The upper end of A is connected
by the rubber tubing
D with the U-shaped tube E, in which is inserted some coloured
water. When the experimenter blows on the surface of the sand
at A, the impulse passes through the sand up the tube from F,
and deflects the water in the syphon bend at E.
The amount of ground-air varies greatly. Loose sands often
contain 40 to 50 per cent., soft sandstone 20 to 40 per cent., and
loose surface-soil many times its own volume.
The nature of the air is not accurately known. It is, however,
extremely rich in carbonic acid, of which it contains from 1 to 10 per
cent. or even more. The carbonic acid is derived from the organic222
matter in the soil, by the action of bacteria, in a manner analogous
to nitrification.
The Water contained in the Soil is divided into moisture
and ground or subsoil-water. When air is present in the soil
as well as water, the soil is merely moist. Pettenkofer defines
the ground-water as that condition in which all the interstices are
filled with water, so that, except in so far as its particles are
separated by solid portions of soil, there is a continuous sheet
of water.
The Moisture in the soil varies in amount. Open gravel will
absorb from 9 to 13 per cent. by weight of water; gravelly surface
soil 48 per cent.; light sandy soils from 23 to 36 per cent.; loamy
soil 43 per cent.; stiff land and clay soils from 43·3 to 57·6 per
cent.; sandy and peaty soils from 61·5 to 80 per cent.; peat 103
per cent. (B. Latham). The moisture being derived from the
rainfall on one side, and the ground-water on the other, will vary
with the amount of these. Some soils are practically impermeable
to water, such as trap or metamorphic rocks, unweathered granite,
hard limestone, and dense clay; while others, such as chalk, sand,
sandstone, vegetable soils are permeable. Commonly the metamorphic
rocks and hard limestones present fissures, which render them
pervious. The rainfall which does not penetrate the soil flows into
the streams and rivers at once, or is re-evaporated. The amount of
percolation of rainfall is estimated by an artificial soil-gauge. Most
percolation and least evaporation of rainfall occurs from October to
March inclusive. The difference between the percolation and
rainfall is the loss caused by evaporation and vegetation.
The Ground-water forms a subterranean sheet of water, which
is in constant motion. There is first of all, an irregular rise and
fall of the water, according as it receives new additions from the
rainfall, or loses a certain amount of its substance by percolation
and evaporation; and there is, secondly, a constant movement
towards the nearest water-course or the sea. Many towns derive
their drinking-water from the ground-water, especially that in the
chalk. Thus in Brighton there are no streams; but wells are dug
in the South Downs about 150 to 180 feet deep down to the level of
the subterranean water. Then long adits are tunnelled, parallel to
the coast at or near the level of this water, which is thus intercepted
on its way towards the sea, and pumped up to supply the town. In
Munich, Pettenkofer reckoned the rate of movement of the ground
water towards the outlet as 15 feet daily. It is impeded by
impermeability, or a deficient slope of the soil. The roots of trees
also greatly impede its flow.
The level of the ground-water is constantly changing (see Fig. 7).
The alteration in level may be only a few inches either way, while
in some parts of India it is as much as 16 feet. The level is
generally lowest in October and November, highest in February
and March.
A fall in the level of the ground-water may be due to a dry
season, or to improved subsoil drainage. A rise in its level is due
to an increase in the rainfall, or some obstruction in the outflow,223
as from a swollen river. The tide may influence the level of the
ground-water at a great distance. A sudden alteration in the level
of the ground-water is a common cause of floods in mines.
The distance of the ground-water from the surface may be only
two or three feet, or several hundred feet, the difference being due
to the varying level of the nearest impervious stratum of soil. Its
distance below the surface of the soil can easily be measured by
ascertaining that of the water of a shallow well in the neighbourhood.
It should preferably not be nearer the surface than five or
six feet. Sudden changes in the level of the ground-water from
inundations render any soil unhealthy, and are even more objectionable
than a persistently high level. This is especially true in the
case of permeable soils. A sudden rising of ground-water expels
the air in the soil, together possibly with particles which may
comprise infectious material; it also washes similar impurities out
of the subsoil, and carries them into neighbouring wells. Numerous
epidemics have been traced to this source.
The Temperature of the Soil varies greatly with its
geological character, as well as with the temperature of the atmosphere.
The daily changes in the temperature of the atmosphere do
not affect the soil beyond a depth of about three feet. The annual
changes in the atmosphere will affect the soil in a varying degree,
the amount being dependent on the character of the soil as regards
conductivity and retentiveness for heat. Such annual variations do
not penetrate below forty feet, and are very small below twenty-four
feet. The temperature of the earth increases with its depth, the
rate of increase in England being stated to be about 1° Fahr. for
every 54½ feet.
In England the water of permanent springs has a fairly constant
temperature of 49° to 51° Fahr., which is the temperature of the
deeper part of the subsoil. The method of taking the daily temperature
of the subsoil at a depth of 4 feet is described on page 240.
Although the average temperature of any soil depends on the
climate, soils conduct heat in a very varying degree, and therefore
absorb unequal quantities. This has an important bearing on the
comfort of those living on a particular soil. Schübler’s experiments
give the absorbing power of the chief kinds of soil, 100 being taken
as the standard.
| Sand, with some lime |
100·0 |
| Pure sand |
95·6 |
| Light clay |
76·9 |
| Gypsum |
73·2 |
| Heavy clay |
71·1 |
| Clayey earth |
68·4 |
| Pure clay |
66·7 |
| Fine chalk |
61·8 |
| Humus |
49·0 |
It is evident from this table that sand is very retentive of heat,
while clays and humus are very cold. Green vegetation lessens the
absorbing power of the soil, and radiation of heat is more rapid,
evaporation occurring constantly from the herbage. The influence
of trees on the temperature of the soil is considered on page 228.
Damp soils are colder than dry soils because of the evaporation224
going on. Buchan finds as the result of drainage of the soil, that
(1) the mean temperature of arable land is raised 0·8° Fahr.; (2)
cold is propagated more quickly through undrained land; (3) drained
land loses less heat by evaporation; (4) the temperature of drained
land is more equable, and (5) in summer is often 1·5° to 3° above
that of undrained land.
Diseases Arising from the Soil.—The soil may be a cause
of disease: (a) indirectly and (b) directly.
Indirectly a damp soil may cause disease by acting as a means
of lowering the vitality of man and diminishing his resistance to
disease. It is in this way that it has been credited with causing
such diseases as neuralgia, catarrhs, and rheumatism. It is one of
the elements in producing a climate unfavourable to health. As to
rheumatism, see page 225.
Directly the soil may transmit the actual contagia (micro-organisms)
of disease either by means of the subsoil water or its air.
In the former case the disease-causing material gains access to the
drinking water of wells, springs, or rivers; in the latter case it may
be borne to the surface of the soil by currents of the ground-air or
by insects, and then inhaled as dust, or gain access to food.
Certain disease-producing micro-organisms have been proved to
be capable of living for some time in the soil. The chief of these
found in the soil are the bacilli of tetanus (lockjaw), of anthrax, of
malignant oedema, and of enteric (typhoid) fever. There are reasons
for thinking also that the micro-organisms causing diphtheria,
rheumatic fever, and epidemic diarrhœa, and possibly some other
diseases, may occasionally live in the soil. In some diseases as
enteric fever, cholera, dysentery and anthrax, the contamination of
the soil can be shown to be derived from a patient suffering from
the same diseases. In others, and particularly in tetanus, the same
chain of evidence is obtainable.
(1) The conditions favourable to the production of malarial
diseases have been generally considered to be the presence of
a certain proportion of dead organic matter, the exposure of the
soil to alternations of heat and moisture, with a limited access of
air, and a temperature of at least 65°F. Though most common in
marshy districts, and in recent alluvial soils, malaria may develop
in connection with any geological formation. That it may be
removed by drainage of the subsoil, is well known. The true
nature of the connection between soil and malaria is stated on
page 220.
(2) According to observations made by Pettenkofer in Munich,
attacks of enteric (typhoid) fever are connected with fluctuations
of the subsoil-water. He states his conclusions as follows:—
“Between the fluctuations of subsoil water and the amount and severity of
enteric fever there is an unmistakable connection in this wise, that the total
number of cases of and deaths from enteric fever falls with a rise of the subsoil
water, and rises with fall of it; that the level reached by the disease is not in
proportion, however, to the then level of the subsoil water, but only to the
variation in it on each occasion; or in other words, that it is not the high or low
level of the subsoil water that is decisive, but only the range of fluctuation.”
225
His observations have not been confirmed in this country; and the
coincidence between excess of enteric fever and lowness of ground-water
has been explained by the fact that under these circumstances
the water in wells is low, and the area of drainage and the consequent
risk of contamination are proportionately increased. There
can be no doubt that the most common origin of enteric fever is
from the infection of water or milk by infective matter from a recent
case of the disease. This does not exclude the fact that enteric
fever in this country is more prevalent in hot dry autumns, in which
the ground-water is low. Probably under such conditions the
contagium of the disease multiples more rapidly in the soil, in
privies and other polluted places, and consequently the risks of
infection of water and food as well of infection by dust carried
from the contaminated spot are greatly increased.
(3) In regard to cholera, Pettenkofer holds similar views. He
believes that the contagium of cholera can only be developed when
there is a damp porous subsoil to receive the infected stools from a
cholera patient; the damp porous subsoil forming a second host in
which the poison of cholera must pass through one stage of its
existence, before it is again capable of producing the disease. Such
an essential relationship of the soil is not borne out by observations
in India; and in England cholera has been repeatedly shown to be
due to contamination of food (e.g. oysters) or water by the stools of
preceding cholera patients, without the intervention of any agency
of the soil.
(4) It has been repeatedly stated that a damp soil favours the
prevalence of diphtheria. I have shown elsewhere, however, that
this is not true, and that the greatest epidemics of diphtheria have
occurred in exceptionally dry years, especially when several years
of exceptionally small rainfall have succeeded each other; and
have suggested that this may be associated with an intermediate
stage in the life-history of the diphtheria-bacillus in the soil. A
low ground-water and a comparatively high temperature of the
soil go along with deficient rainfall, and would probably favour
the multiplication of this bacillus in the soil.
(5) In rheumatic fever I have similarly shown that the
supposed connection between damp soil and this disease is erroneous,
the disease being most prevalent, both in this and other countries,
in years of exceptional drought.
(6) Epidemic or Summer Diarrhœa has been supposed to
have a special relationship with soil-temperature, Ballard having
found that the summer rise in the mortality from this disease does
not commence until the mean temperature recorded by the four-foot
earth thermometer has attained somewhere about 56°F. The soil-temperature
may be accepted as a convenient index of the conditions
causing this disease. The disease I have elsewhere shown occurs
most severely with a high temperature of the air and a deficient
rainfall, and its fundamental cause is an unclean soil, the particulate
poison from which infects the air, and is swallowed most commonly
with food, especially milk.
(7) The close connection of consumption (phthisis) with a226
damp soil has been independently stated by Drs. Buchanan and
Bowditch. Buchanan found that in the districts where improved
sanitary arrangements had led to a drying of the soil, the death-rate
from phthisis diminished; but where with sanitary improvements
the soil was not dried, the death-rate from phthisis remained in one
or two instances almost stationary. In Salisbury, Ely, Rugby, and
Banbury, the death-rate from phthisis fell from 141 to 49 per cent.
The amount of reduction in the death-rate from phthisis did not
appear to be consistently proportional to the amount of drying of the
subsoil. In a later investigation into the incidence of deaths from
phthisis in the south-east of England, Buchanan came to the
further conclusions that (a) there was less phthisis among populations
living on pervious soils than among populations living in
impervious soils; (b) less phthisis among populations living on
high-lying pervious soils than among populations living on low-lying
pervious soils; and (c) less phthisis among populations living
on sloping impervious soils than among populations living on flat
impervious soils. He, therefore, concluded that wetness of soil is a
cause of phthisis to the population living upon it. (See also page 313).
Drainage of the Soil.—There are two chief plans for rendering
the soil drier—deep drainage and opening the outflow.
Subsoil Drainage should always be carried out by drains,
separate from those for sewage. If the sewers are utilised for this
purpose, their contents when full contaminate the surrounding
soil. The subsoil drains should be composed of agricultural, i.e.
unglazed, drain-pipes laid in towns in the same trench, but above
the sewers, and they should discharge into the nearest water-course.
If it is necessary to join them with a sewer, they should not pass
directly into it, but into a disconnecting man-hole.
Opening the Outflow, in order that water may not remain
stagnant in the soil, is occasionally required. This may be done
by clearing water-courses, removing obstructions, and forming
fresh channels.
The provision of sufficient surface-drains to carry off ordinary
water and storm-water helps in drying the soil of urban districts.
Vegetation tends to diminish dampness of soil by causing
rapid evaporation, and at the same time uses up the organic matter
in the soil. Certain plants are more active in producing these
effects than others: the Eucalyptus genus, including many species,
and represented by the well-known blue-gum tree of Australia, is
noted for its power in this respect; and the common sun-flower,
which is very easy of cultivation, has a powerful influence in the
same direction.
227
The Climate of a country has an important influence on the health
and character of its inhabitants. The character of a climate
depends on four main conditions:—
- 1. The distance from the equator.
- 2. The height above the sea.
- 3. The distance from the sea.
- 4. The prevailing winds.
There are other conditions which are of subsidiary importance,
but which have great influence in modifying the climate of any
given locality. Thus:—
- 5. The nature of a surface—its aspect, shelter, slope; the
colour of the soil or rock, the reflection from rocks or sheets of
water, and the influence of vegetation.
- 6. The cultivation of the soil.
- 7. The drainage of marshes and damp soils.
- 8. The planting and clearing away of forests.
The Distance from the Equator is the most important
factor in relation to climate. The sun’s rays become less powerful
as they fall more obliquely, in travelling from the equator. This
primary factor in producing climate is largely modified, however,
by the relative distribution of land and water, and by the character
of the prevailing winds of a district.
The Elevation of a locality affects the temperature and the
barometric pressure, both falling as the height is increased. The
amount of fall varies with the latitude of the place, with its
situation in regard to surrounding districts, the degree of moisture
of the air, the presence of winds, the hour of day, and the season
of the year. It is usual to allow 1° Fahr. for every 300 feet of
ascent above the level of the sea, and one-thousandth part of an
inch depression of the barometer for every increase of one foot in
height.
Hills, Plain and Valley.—The law of decrease of temperature
with increase of altitude, is liable to great modifications, and even
subversions, from various causes. The chief cause producing such
modification of the law is the elevation in relation to the surrounding
district. Thus, in the case of rising ground, the higher parts
become rapidly cooled by radiation. The air here is likewise
cooled by contact, and becoming heavier in consequence, flows228
down to low-lying ground. Hence places on rising ground are not
so fully exposed to the intensity of frosts at night as places in
the valley.
Valleys surrounded by hills and high grounds, not only retain
their own cold and heavy air, but serve as reservoirs for the cold
air falling from neighbouring heights. One finds, in consequence,
mists in low situations, while adjoining eminences are quite clear.
The air of mountains is (1) cooler than that of lower districts
with the exception already named. (2) It is less dense in proportion
to the altitude; its pressure at the height of 16,000 feet being
only half that at the sea level. (3) Its absolute humidity is
decidedly diminished; there is some difference of opinion as to the
relative humidity. (4) The air is as a rule purer. It is generally
free from dust, and to a large extent aseptic (that is, free from
microbes). (5) The amount of ozone is commonly greater than in
lower regions. In addition to these characters, (6) the light is
intense, and (7) the direct heat of the sun is greater, and the
difference between sun and shade greater than in lower regions.
Owing to these peculiarities of mountain air, it is of great value
as a restorative. The circulation of blood is increased, nutrition
is improved, the chest expands, and the increase in its size may be
permanent.
The presence of forests and sheets of water counteracts the
effects of radiation from the earth. Thus if a deep lake fills the
basin of a valley, the cold air descending from higher levels cools
the surface water, which sinks and is replaced by warmer water
from below. In this way deep lakes are sources of heat in winter,
and places on their shores are free from the severe frosts which are
peculiar to other low-lying situations.
If the slopes of a hill are covered with trees the temperature of
its sides and base are considerably increased, as the trees obstruct
the descending currents of cold air. The frosts of winter are felt
most severely in localities where the slopes above them are
destitute of vegetation, and especially of trees. It follows that in
any given locality, the best protection against the winter cold is
ensured by a dwelling situated on a slope a little above the plain or
valley from which it rises, with a southern exposure, and sheltered
by trees planted above it. Such local conditions should always be
carefully enquired into, when a choice of site is possible, as the
temperature of one part of a neighbourhood may differ by several
degrees from that of another part near at hand. This is particularly
important in the case of invalids.
Forests tend to modify a climate, and mitigate its extremes,
whether situated on the slopes of mountains or on plains. In
America, as elsewhere, the effect of destruction of forests has been
to produce greater variation in the annual rainfall, to lengthen
periods of drought, and to increase the power of floods and cloud
bursts. Trees are heated and cooled by radiation like other bodies,
but from their slow conducting power, the periods of their maximum
and minimum temperatures are not reached for some hours after the
same phases of the temperature of the air, and the effects of radiation229
are not confined to a small surface on the soil, but distributed to
the level of the tree-tops. For these reasons, trees make night
warmer and day cooler, thus giving to forest districts something of
the character of an island climate. Evaporation occurs slowly
from the damp soil beneath trees, as it is screened from the sun,
and the trees prevent a free circulation of wind. Hence the relative
humidity and rainfall are increased. At the same time forests
mitigate the disintegrating effect of the rainfall on the soil.
Ground covered with Vegetation has a more uniform
temperature than bare soil, the effect being much the same as that
of forests, though on a smaller scale.
All growing vegetation evaporates a large quantity of water.
A plant evaporates 200 pounds of water while it forms one pound
of woody fibre; the effect of a forest must, therefore, be enormous.
At the same time, vegetation, and especially trees, retain moisture
in the soil. The water-supply of barren regions may be greatly
increased by planting trees.
The absence of vegetation leads to extreme fluctuations of temperature.
An extent of sand, for instance, raises the temperature
of the air greatly during the day, as it is a bad conductor; but at
night, radiation is very great, and the temperature falls accordingly.
Relation of Sea to Climate.—Water has the greatest specific
heat of any known substance, being four times greater than that
of the earth’s crust. On this account it takes longer to heat and
to cool than the earth. Unlike the earth, likewise, it allows free
penetration of the sun’s rays,—in clear water probably to a depth
of at least 600 feet; consequently, the surface of the water becomes
less rapidly heated. The freezing point of fresh water is 32°, while
that of sea-water is 27·5°-28·4°. Thus, the sea remains open at
a temperature at which inland lakes freeze, and has, therefore, a
greater influence in moderating winter cold and summer heat.
Another factor rendering it more competent to mitigate extremes
of temperature than lakes, is the presence of currents, causing
admixture of the water of different climates. Of these currents the
most important for this country is the Gulf Stream, an immense
stream of water which, when it leaves the Gulf of Mexico, is
travelling at the rate of four to five miles an hour, and has a
surface temperature of 88° F.
It is important to distinguish between the surface temperature
and the deep-sea temperature, the latter being fairly constant.
The whole of the depths of the sea is filled with water at or
near 32° Fahr., which in the tropics is 40°-50° below the temperature
of the surface-water.
The influence of seas on climate is so great as to lead to a
classification of climates into oceanic, insular, and continental.
An oceanic climate is least liable to violent changes of
temperature. It can only be obtained by a sea-voyage.
An insular climate presents smaller differences between the
temperature of summer and winter than the interior of great
continents, especially when the island is small and in the midst of
the ocean. In the British Islands, the prevailing winds being230
westerly, places on the east coast are less truly insular than
similarly situated ones on the west coast; and their climate
approaches more nearly that of inland countries.
A continental climate is drier and more subject to extreme
alternations of temperature than insular and oceanic climates.
Isothermal lines (lines of equal mean temperature) around
the world bend up and down, the bendings being determined by the
relative position of continents and oceans. New York has the same
mean temperature as London, though New York is as far south of
London as Madrid. This fact illustrates the fallacy in judging of
the climate of a locality by the annual mean temperature. Means,
it has been well said, are general truths but particular fallacies. One
should know the extremes of temperature, and the extremes for each
month of the year, as well as the amount and distribution of the
rainfall, and the amount of sunshine, before judging of a local
climate.
Winds are due to differences in atmospheric pressure caused
by changes in temperature and moisture. Inasmuch as the temperature
and degree of moisture of air vary with the prevailing
winds, their consideration becomes very important. Winds bring
with them the temperature of the air they have traversed: thus, in
England, south winds are warm, while north winds are cold.
Winds coming over an ocean cause less variation in temperature
than those which have passed over an extensive tract of country.
Thus, moist ocean winds are accompanied by a mild winter and cool
summer, while dry continental winds cause the reverse conditions.
The amount of moisture capable of being carried by a current of air
increases with its temperature; therefore, equatorial winds become
moister as they proceed, while north winds become drier. The
south-west winds, in the British Isles, being both oceanic and
equatorial, are very moist, while the north-east winds, being both
northerly and continental, are peculiarly dry and parching.
Owing to the atmospheric pressure diminishing from the south of
Europe northwards to Iceland, south-west winds are the most
prevalent in Great Britain; and as this diminution of atmospheric
pressure is greatest in the winter months, south-west winds are
most common at this season. The result is that the temperature of
these islands is higher than that due to mere latitude, and the
temperature on the west coast is fairly uniform from Shetland to
Wales.
Mountain ranges have an important bearing in determining the
character of the prevailing winds. If the range is perpendicular to
the direction of the winds, the latter lose the greater part of their
moisture, and the places to leeward being exposed more completely
to solar and terrestrial radiation (from comparative absence of
aqueous vapour), winter becomes colder and summer hotter. The
difference between the climates of the west and east parts of Great
Britain is largely due to this cause. In Ireland, the mountains are
not grouped in ranges running north and south, but in isolated
masses, and the difference in climate between the east and west
coasts is consequently less marked.
231
The prevailing winds have a great influence on the
rainfall. (1) Thus if the wind has traversed a considerable extent
of ocean, the rainfall is moderately large. (2) If a wind reaches
into a colder region, its saturation point is lowered, and the rainfall
is greatly increased; and if a range of mountains lies across its
path, the rainfall on the side facing the wind is greatly increased,
but diminished on the opposite side of the range. (3) If a wind
after reaching land proceeds into lower latitudes or warmer regions,
the rainfall is small, or absent. This accounts for the rainless
summers of California, North Africa, and South Europe.
The Barometric Pressure varies daily, being at its maximum
at about 9 a.m. and 9 p.m. The average range in the tropics
amounts to 0·1 inch, but in this country does not usually exceed
0·02 inches. During the year the minimum barometric pressure
usually occurs about the end of October, while the maximum is
usually at the end of May or early in June. The ordinary variations
in barometric pressure with changes of weather have little apparent
effect on health; but more extreme changes produce marked effect.
In mountain-climbing faintness and nausea may be caused at great
altitudes. At the opposite extreme, in pier-driving and laying the
foundations of bridges, men have to work in air-chambers at a
pressure of from three to four atmospheres. Then what is known
as “caisson disease” may be produced. The usual symptoms are
discomfort or pain in the ears, giddiness, bleeding at the nose,
vomiting, or even temporary paralysis. In such occupations it is
most important that on leaving the air-chambers the atmospheric
pressure should be gradually lowered.
The use of the barometer as a weather indicator is based on the
fact that moist air is lighter than dry air. Hence, if the air is
moist and rain imminent, the barometer falls rapidly. The
maximum daily range in this country is rarely greater than 3 inches.
Weather observations can be based on records kept at one spot.
Their value is greatly enhanced, when such observations are
compared with others distributed over a wide area. The wider the
area from which such observations are collated, the more accurate
the deductions that can be secured. If observations of places at
which the barometrical pressure is identical be recorded on a map,
we have a synoptic map, and the lines of equal barometrical pressure
connecting these points are called isobars. The modern development
of meteorology, enabling forecasts of weather to be made with
approximate accuracy, is based chiefly on telegraphic communication
of information, enabling isobars to be constructed.
It is found that isobars arrange themselves into seven chief
forms (1) Cyclones. (2) Secondary cyclones. (3) V-shaped
depressions. (4) Anti-cyclones. (5) Wedge-shaped isobars.
(6) Cols. (7) Straight isobars.
Each of these varieties is shown in Fig. 43, which embraces
the conditions in Europe, the eastern part of the United States,
and over the North Atlantic on a certain day.
The closeness of the isobars, i.e. the rapidity of changes in
atmospheric pressure determines the barometric gradient. The steeper232
this gradient, the greater the velocity of the wind in any given place.
The distance between two isobars is equal to a change of a tenth of
an inch in the mercury in the barometer. The direction of the wind
in a given place is from the higher to the lower isobars. This
is expressed in Buys Ballot’s law, which states that in the northern
hemisphere, if you stand with your back to the wind, the lowest
pressure is to your left and in front.
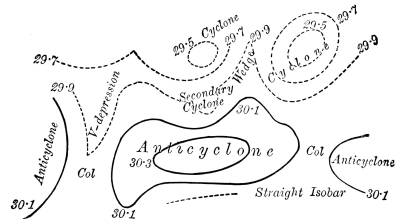
Fig. 43.
The Seven Fundamental Shapes of Isobars (after Abercrombie).
Cyclones or depressions are areas of low barometric pressure.
A cyclonic system (Fig. 43) is formed by circles of concentric
isobars. The differences between cyclones and anti-cyclones are
as follows:—
| Cyclones. |
Anti-cyclones. |
| Wind moves in the opposite direction to the hands of a watch. |
Wind moves in same direction as the hands of a watch. |
| Barometer is lowest in the centre. |
Barometer is highest in the centre. |
| Area comparatively small. |
Area comparatively large. |
| Gradient from centre to circumference steep. |
Gradient not steep. |
| Short duration. |
Long duration. |
| Velocity of wind great. |
Air comparatively quiet. |
| Weather bad; much rainfall. |
Weather fine. |
| Cool in summer; warm in winter. |
Hot in summer; cold and frosty in winter. |
Cyclones usually travel from west to east, and are always
associated with bad weather. The essential point in determining
the character of the weather, both in cyclones and anti-cyclones, is
the barometric gradient. Thus, according to the gradient, a cyclone
may mean mild wet weather, a gale, or a hurricane. The turning
point of a cyclone, just before the barometer begins to rise again, is
called the trough. Cyclones are usually oval in shape, except in the
tropics, where they are smaller and circular. The ordinary course
of events in a cyclone is shown in Fig. 44, reading it from left to
right.
233
In Secondary Cyclones, bad weather is usually associated
with a stationary barometer and no wind. They are incompletely
circular looped concentric isobars, with the lowest pressure in the
centre. They frequently follow primary cyclones.
V-shaped Depressions are angular areas, with the lowest
pressure in the centre, frequently forming between adjoining anti-cyclones
(Fig. 43). In the northern hemisphere the tip usually
points south. They usually move with great rapidity from east to
west, and are always associated with squalls or thunderstorms.
Their movement is very uncertain, and their forecast therefore more
difficult than that of cyclones and anti-cyclones.
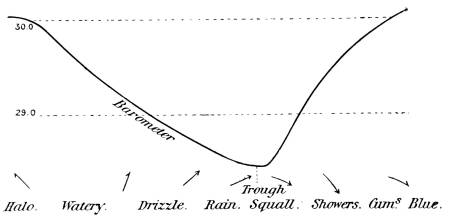
Fig. 44.
Weather Sequence in a Cyclone (after Abercrombie).
The tracing indicates the line which a self-recording barometer would have
marked. The arrows mark the shift of the wind, and the number of barbs
denote the varying force of the wind.
Anti-cyclones are associated with calm and cold in the centre,
while on the borders the wind blows around the centre, spirally
outwards in the direction of the hands of a clock. An anti-cyclone
is usually accompanied by a blue sky, dry cold air, a hot sun, a
hazy horizon, and little or no wind.
Wedge-shaped Isobars, unlike V’s, usually point north.
They are areas of high pressure moving along between two
cyclones, being really projecting parts of an anti-cyclone. The
fine weather accompanying them is only temporary, because they
are never stationary, and are generally followed by cyclonic
disturbances. At the narrow end of the wedge thunderstorms or
showers often occur, and at the wide end fog is common.
Cols or necks of relatively low barometric pressure occur
between two anticyclonic areas. Like straight isobars they are
intermediate systems. Over cols the weather is dull and gloomy;
in summer they may be associated with thunderstorms.
Straight Isobars obviously do not enclose any area of high or
low pressure. They form an intermediate condition, preceding the
formation of a cyclone; and are usually associated with a blustering
wind and hard sky.
234
Weather forecasting is necessarily somewhat difficult and
uncertain. If one is dependent on observations at a single point the
following rules are useful:—
- (a) If the barometer falls slowly and steadily bad weather will follow.
- (b) The barometer falls for rain with S.W., S.E., and W. winds.
- (c) When the barometer falls rapidly, heavy storms may be expected.
- (d) The barometer rises rapidly for unsettled weather.
- (e) The barometer rises gradually for fine, settled weather.
The Thermometer also is of great value as a weather indicator,
especially if one knows what is the average temperature at the place
of observation for each day of the year. Thus:—
- (a) A temperature continued for some time above or below the average,
indicates a probable change.
- (b) Electric storms follow unusual warmth in summer.
- (c) A low thermometer and almost steady barometer are succeeded in winter
by gales from N.N.W. or N.E.
The veering of the wind in England is also useful as an
indicator. Thus:—
- (a) When the wind, in shifting, goes round in the same direction as the
hands of a clock—i.e., from N. by E. to S., or from S. by W. to N.,—favourable
changes of weather may be looked for.
- (b) When the wind backs—that is, veers round in the opposite direction—bad
weather generally follows.
The direction of the wind is an important factor. Thus:
- (a) Settled N.W. winds bring cold and fine weather.
- (b) Continued W. and S.W. winds are followed by rain.
Clouds give useful indications. Thus:—
A mackerel sky, that is, one covered with lines of cirrus clouds, causing
halos around the sun and moon, presages rain in summer and thaw in
winter. By degrees the light clouds descend and pass into either
masses of cumulus, or into dense, horizontal stratus, which form at
sunset and disappear at sunrise. Both these kinds pass into the grey,
shapeless nimbus, which soon covers the entire sky and is followed by
rain.
When numerous observations can be synoptically studied,
forecasting becomes much more nearly certain. For this purpose
telegraphic communications are indispensable. The continent of
Europe is better placed than England for accurate forecasting.
Areas of high pressure coincide usually with large areas of land, of
low pressure with large surfaces of water. Thus England is placed
near the boundary of the usual anticyclonic and cyclonic systems,
and its chief disturbances come from the Atlantic from which early
communication is impracticable. Furthermore cyclonic disturbances
may be diverted from their course by a coastline or mountains
or by the formation of an anticyclonic area. In view of these
uncertainties, the large proportion of correct forecasts is somewhat
surprising.
The Moisture of the Air depends upon the amount of vapour
present in it, and the ratio of this to the amount which would
saturate the air at the actual temperature. The former is called the235
absolute humidity, the latter the relative humidity. The dew point is
the point at which condensation of some of the vapour in the
atmosphere occurs, either as dew, rain, snow, or hoar-frost. The
amount of moisture which the atmosphere can retain before such
condensation occurs, varies with the temperature (see page 101).
Thus the air is drier at noon than at midnight, though the amount
of vapour present in the two cases be the same; and it is for the
most part drier in summer than in winter. This refers to the
relative humidity, which is highest in cold weather. The absolute
humidity is higher in summer than in winter; it varies more in
continental than in maritime and insular climates; and there are
daily variations according to the state of the sky, the movements of
air, etc. The relative humidity is expressed as a percentage of
what would be required to produce saturation at the given
temperature. The usual relative humidity is 50 to 75 per cent. A
moist air prevents excessive changes of temperature due to radiation.
It protects the earth from too great intensity of the solar rays by
day and from too rapid loss of heat by radiation at night. The
inhalation of a dry air plays an important part in the cure of
consumption. When the air is almost saturated with moisture,
evaporation from the skin and lungs is diminished, and there is a
feeling of oppression and disinclination to work caused by the
interference with the tissue changes of the system.
Rainfall is caused by over-saturation of a column of moist
air. This may be due to the contact of the air with a cold surface,
as the ridge of a mountain or a large surface of water, or to the
impact of a colder wind.
The amount of rainfall varies greatly. In some parts there is
no rain, as in the desert of Sahara; while on the south-east slopes
of the Himalayas, which are exposed to winds laden with moisture,
it may be several hundred inches.
The latitude of a place has a great influence. As a rule the
rainfall decreases with increasing distance from the equator; but
local conditions may produce great modification, or even alterations
of this law.
The elevation above the sea-level has a varying influence. In the
Swiss Alps it is said that the rainfall increases with the elevation;
but this rule does not hold good in America.
The nearness of large surfaces of water in summer tends to
increase the rainfall, when water is colder than its surroundings,
while in winter it has the opposite effect. The neighbourhood of the
sea is for the west of England and islands adjacent, a cause of
increased rainfall.
The influence of winds on the rainfall has been already
considered. In Great Britain south-west winds more especially
increase the rainfall. In their course they have travelled over the
Gulf Stream and the general equatorial current, and have thus
received warmth and moisture. The condensation of their moisture
liberates a large amount of latent heat, thus raising the temperature
of this country. In summer, however, south-west winds are cool236
and moist, as the Atlantic is not so hot as the continents of Asia
and Europe over which other winds have travelled.
In England the average rainfall is about 33 inches, in Scotland
46, and in Ireland 38 inches. In the east of Great Britain, the
rainfall is from twenty to twenty-eight inches. On the west coasts
of Scotland and Ireland it is from 60 to 80 inches; and in some
parts of Cumberland may be about 150 inches per annum. The
annual rainfall varies greatly from the average for a number of
years. In this country it has been estimated that the maximum
annual rainfall exceeds by one-third, and the minimum annual
rainfall is less by one-third than the average rainfall of a series of
years.
The number of rainy days by no means corresponds with the
amount of rainfall. There are fewest rainy days at the equator,
where the rainfall is greatest. The rain diminishes the relative
humidity of the air, and purifies it from dust.
237
CHAPTER XXXV.
METEOROLOGICAL OBSERVATIONS.
The Royal Meteorological Society recognises stations for the
making and recording of observations of three kinds: (1) Second
Order Stations, at which observations are taken twice daily at 9 a.m.
and 9 p.m.; (2) Climatological Stations, at which the observations
are taken once daily, at 9 a.m.; (3) Stations at which one or more
elements only, e.g. rainfall, are observed. All instruments used should
have been previously verified at Kew
Observatory, so that the corrections for
index error may be known.
The Barometer used should be of a
standard kind. Five chief kinds of
barometer are in use, only the last two
of which are sufficiently accurate for
scientific purposes.
1. The Dial or wheel barometer consists of a bent
tube A B, the open end of which supports an ivory
float B. This, as it rises and falls with the mercury,
by means of the rack C turns a wheel, in the axle
of which a needle is fixed. The needle turns in
one direction, or the other as the mercury rises or
falls (Fig. 45); the dial is divided by comparing
it with a standard barometer. As the ordinary
variations of the barometer are from 28 to 31
inches, the circumference of the wheel is made
exactly 1½ inches, and thus the float B will rise or
fall 1½ inches for a rise or fall of 3 inches in the
barometer.
Fig. 45.
2. The ordinary syphon barometer (Fig. 46)
consists of a bent tube attached to a piece of wood,
and furnished with a screw v. The atmospheric
pressure acts on the mercury at d, and the difference
between the level of the mercury in the two arms
of the syphon is the height of the barometer. To
find the true height of the barometer the screw is
turned till the shorter column stands at a opposite
zero.
3. The aneroid barometer is made by exhausting
the air from a small round metal box. This box is
closed by a flexible lid of metal which, being elastic,
yields to changes in the atmospheric pressure. To
the lower end of the lid a spring is attached which
runs downwards to the floor of the box and resists
the atmospheric pressure. The movements thus
produced by variations in pressure are magnified by a rack and pinion, and so
communicated to a long index which moves over a graduated scale.
238

Fig. 47.
Fortin Barometer.
a—Attached thermometer.
b—Screw
of vernier. c—Screw
for setting level of
cistern.
The standard Kew and Fortin barometers are both cistern
barometers, the mercury in the inverted tube communicating with
the mercury in a cistern below.
4. The Kew pattern barometer has a closed cistern below, the
area of which being accurately known, the
inches on the scale are not real inches, but
inches of pressure, i.e. inches so shortened
as to compensate for the rise of the
mercury in the cistern. This compensation
is necessary inasmuch as changes in
atmospheric pressure affect the level of
the mercury in the cistern as well as of
that in the tube.
5. In the Fortin barometer (Fig. 47) the
cistern has a pliable base of leather, which
can be raised or lowered by means of a
screw. The upper part of the cistern is
made of glass, a piece of ivory indicating
the zero of the scale. Before taking a
reading, the level of the mercury must
always be set exactly to this point by
means of the screw. The Fortin is the
most sensitive form of barometer, and the
adjustment required in order to take a
reading is easily performed.
To ensure more accurate reading
of the barometer, a secondary scale
or vernier is used, which slides
upon the principal scale. This
vernier is so graduated that 25 of
its divisions correspond to 24 of the
divisions on the fixed scale. The
fixed scale is divided into inches,
tenths (·1), and half-tenths (·05).
Each division of the movable scale
or vernier is therefore shorter than
each division of the scale by 1 ∕ 25 of
·05, i.e. ·002 inch. Consequently
the vernier shows differences of two
thousands of an inch.
Method of reading Fortin’s Barometer.—First
note the reading of the attached
thermometer; next turn the screw at the
bottom of the cistern, so that the ivory point just touches
the surface of the mercury. Next adjust the vernier by
means of the rack and pinion at the side of the barometer
(Fig. 47) so as to bring its two lower edges exactly on a
level with the convex surface of the mercury. In
reading the barometer, first read off the division next
below the lower edge of the vernier. In Fig. 48 this is
29·05. Then the true reading is 29·05 plus the vernier
indication. Next look along the vernier until one of its
lines is found to agree with a line on the scale. In Fig.
48 this is at the fourth division on the vernier. But
each of the figures marked on the vernier counts as 1 ∕ 100
(·01), and each intermediate division as 2 ∕ 1000 (·002);
hence the reading of the vernier will be ·008 inch, and239
the reading of the barometer 29·05 + ·008 = 29·058 inch. If two lines on the
vernier are in equally near agreement with two on the scale, the intermediate
value should be adopted.
Certain corrections are required in the actual reading for (1)
index error; (2) temperature; and (3) height
above sea-level.
The index error is found by comparison
with a recognised standard at Kew. Correction
for temperature is required. Every barometer has
a thermometer attached, and the reading is reduced
to the standard temperature of 32° F, by means
of tables such as are given on page 32 of Marriott’s
Hints to Meteorological Observers.
The height of the cistern of the barometer
above sea-level should always be exactly obtained.
The correction necessary to reduce observations
to sea-level (i.e. mean half-tide level at
Liverpool), depends on the temperature and
pressure of the air, as well as on the altitude.
The data for this correction are given in Table
III. of Marriott’s Hints.

Fig. 48.
Scale of Barometer
(to Right) and of Vernier (to Left).
Thermometers.—The maximum thermometer
may be on Negretti and Zambra’s, or
on Phillips’ principle. In the former (Fig. 49)
the bore of the tube is reduced in section near the
bulb (A) in such a way that while the expanding
mercury forces its way into the tube, the column
of mercury breaks off on contraction, so that its
upper limit shows the highest temperature that has been reached.
The thermometer is set by holding it bulb downwards and shaking
to make the mercurial column continuous. It is mounted in the
screen horizontally (Fig. 51).

Fig. 49
Negretti and Zambra’s Maximum Thermometer.
The minimum thermometer chiefly used is Rutherford’s.
It contains spirit in which is an immersed index (A, Fig. 50).
With a falling temperature the spirit draws the index along with it;
but on rising again, the spirit passes the index, leaving it at the
lowest point to which it has been drawn. Thus the end farthest
from the bulb registers the minimum temperature. The instrument
is set by raising the bulb and allowing the index to slide to the end
of the column of spirit. The thermometer must be firmly fixed and
mounted quite horizontally.
240
Fig. 50.
Minimum Thermometer.
Thermometer Screen.—The above thermometers, as well as
the dry and wet bulb thermometers are mounted in a Stevenson’s
screen (Fig. 51). This is a doubled-louvred box through which the
air can pass freely, but the sun cannot enter. The horizontal
position of the maximum and minimum and the vertical position of
the dry and wet bulb thermometers are shown in Fig. 51.
Three additional thermometers are usually included in a well-organised
meteorological station.
A minimum thermometer placed on the grass gives the lowest
temperature on the grass, which is often considerably lower than
that of the neighbouring gravel walk. This record is chiefly useful
for agricultural purposes.
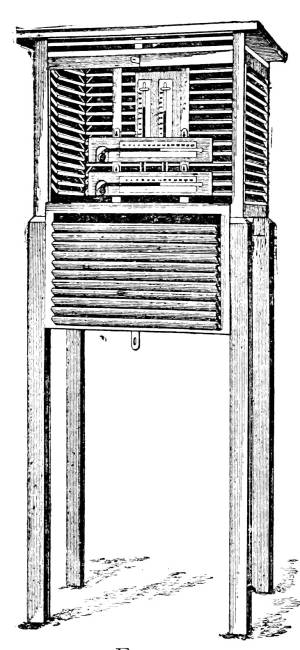
Fig. 51.
Stevenson’s Thermometer Screen.
The earth thermometer chiefly used
is shown in Fig. 52. It consists of a
sluggish thermometer mounted in a short
weighted stick attached to a strong chain,
and of a stout iron pipe which is drawn
out at the bottom to a point and driven
into the earth, usually to a depth of 4 feet.
Solar radiation is measured by black-bulb
and light-bulb thermometers in vacuo,
which are mounted on a post 4 feet above
the ground and record the maximum
temperature.
Humidity in the air is measured by
direct or indirect hygrometers. Of the
former Dines’, Daniell’s, and Regnault’s
are the best known, but as they are not
used in observations acknowledged by the
Royal Meteorological Society, the reader
may be referred to their description in
books on physics. The indirect hygrometer
which is universally employed in this
country is that furnished by the dry and
wet bulb thermometers. In frosty
weather they require much attention, and
then a Saussure’s hair hygrometer may be
used as supplementary. The general
arrangement of the dry and wet bulb thermometers is shown in Fig. 53.
The wet bulb is covered with a single layer of soft muslin,241
while a noose of six to eight strands of darning cotton connects the
neck of the wet bulb with a covered water receptacle 2 to 3 inches
distant, below and at its side. This receptacle is kept filled with
rain-water.
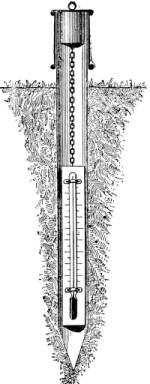
Fig. 52.
Symons’ Earth Thermometer.
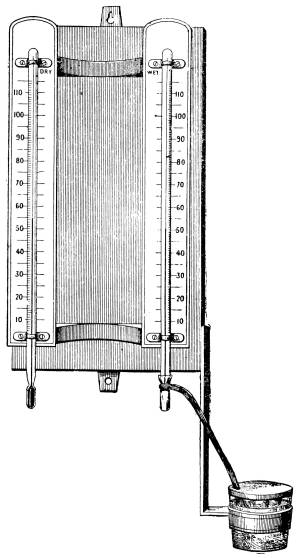
Fig. 53.
Dry and Wet Bulb Thermometers.
From the readings of the dry and wet bulb thermometers three
deductions can be made:
1. The temperature of the dew point.
2. The elastic force of aqueous vapour.
3. The relative humidity.
The dew point temperature
is that temperature
at which the outside air at
the time the observation is
taken will deposit the
moisture contained in it. It
is the temperature at which
the air is saturated with moisture. It is calculated from the readings
of the wet and dry bulb thermometers
(a) by Glaisher’s tables; (b) by Apjohn’s formula.
Glaisher’s tables are based on a series of numbers called
Greenwich or Glaisher’s factors, which he determined by comparison242
between observations made with the dry and wet bulb thermometers
and with Daniell’s hygrometer. The formula for using the factors
is as follows:—
d = D - {(D - W) × f}
where d = dew point, D = dry bulb temperatures, W = wet
bulb temperature, and f = factor.
The following examples are from Glaisher’s table of factors.
READING OF DRY BULB
THERMOMETER FAHR. |
FACTOR. |
| 55° |
1·96 |
| 56° |
1·94 |
| 57° |
1·92 |
| 58° |
1·90 |
| 59° |
1·89 |
| 60° |
1·88 |
- Thus, if D = 60°,
- W = 55°,
- then dew point = 60 - {(60 - 55) 1·96}
- = 50°·2.
The dew-point may also be obtained by Apjohn’s formula; which
for a pressure of about 30 inches is F = f - (D - W) ∕ 87
- D being dry and W wet bulb temperature,
- F elastic force of vapour corresponding to dew-point, and
- f, elastic force corresponding to wet bulb temperature (ascertained from a
table of tensions).
The elastic force of aqueous vapour, i.e. the amount of barometric
pressure due to the vapour present in the air is dependent upon the
temperature of the dew-point. It is given for every tenth of a
degree of temperature in Table VI. (p. 42) of Marriott’s Hints.
The relative humidity is a term expressing the percentage of
saturation of the air with water vapour. It is obtained from Table
VI. (above) as follows:—
| Relative |
 |
= |
Elastic force of water vapour at the temperature of the dew-point |
| Humidity |
Elastic force of water vapour at the temperature of the air (i.e. the dry-bulb reading.) |
- Thus elastic force with dry bulb = 55° is ·433 in. in the table.
- Thus elastic force with dew-point = 46°·5 is ·317 in.„
- ·317 ∕ ·433 = ·73.
- If saturation = 100, relative humidity is 73.
In Table VII. of Marriott’s Hints, a table is given which
enables the relative humidity to be found by mere inspection. Thus
if the dry bulb temperature is 58°·5, wet-bulb 51°·7, and the
difference 6°·8, the relative humidity given in the table is 62.
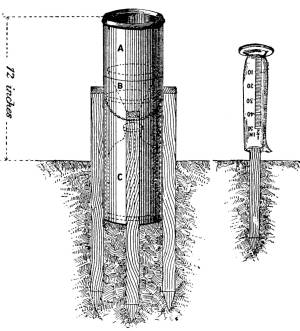
Fig. 54.
Snowdon Pattern Rain-Gauge.
A. Copper Upper Part of Gauge. B. Funnel. C. Bottle.
To the right is shown the glass measure inverted.
The Rain-Gauge is best made of copper in the shape of a
circular funnel, usually 5 or 8 inches in diameter, leading into a
bottle underneath. It must always be set in an open situation away
from trees, walls, and buildings. According to Scott no object
ought to subtend a greater angle with the horizon than 20° in any243
direction from the gauge. The rain is measured by pouring the
contents of the bottle into a glass measure, which is graduated to
represent tenths and
hundredths of an inch
on the area of the
gauge, the measure
holding half an inch
of rain on this area.
Snow is melted before
being measured.
Observations of
wind should include
its direction and
force. The direction
is observed by means
of a well-oiled and
freely exposed vane.
There are 32 points
to the compass, but
a reading to eight
points suffices. The
force of the wind
should be estimated
by Beaufort’s scale,
from 0 to 12.
Thus:—
| FORCE. |
MILES PER HOUR. |
| 0. Calm |
3 |
| 1. Light air |
8 |
| 2. Light breeze |
13 |
| 3. Gentle |
18 |
| 4. Moderate |
23 |
| 5. Fresh |
28 |
| 6. Strong |
34 |
| 7. Moderate gale |
40 |
| 8. Fresh |
48 |
| 9. Strong |
56 |
| 10. Whole |
65 |
| 11. Storm |
57 |
| 12. Hurricane |
90 |
Robinson’s anemometer is also employed, but it is not altogether
trustworthy.
Sunshine is recorded by the Campbell-Stokes burning recorder,
and the Jordan photographic recorder. Of these the former is the
more easily worked and gives more uniform results. It consists of
a sphere of glass 4 inches in diameter, supported on a pedestal in a
metal zodiacal frame (Fig. 55). The setting of the recorder should
be due south, level from east to west, and with the axis of the ring
inclined to the horizon at an angle equal to the latitude of the place,
and so that the image of the sun, when the sun is due south, shall244
fall on the meridian line marked on the ring. The sun burns away
or chars the surface of the cards inserted in the proper groove, and
so gives a record of the duration of bright sunshine.
Fig. 55.
Campbell-Stokes Sunshine Recorder.
The amount of Cloud should be estimated daily, according to a
scale ranging from 0 to 10, i.e. clear sky up to completely overcast.
The form of cloud should also be stated, as cirrus, cirro-cumulus,
cirro-stratus, cumulus, cumulo-stratus, stratus, and nimbus.
245
Certain personal factors are very important in relation to health.
The chief of these are constitution, temperament, heredity, idiosyncrasy,
age, sex, and habits.
Constitution.—Health may vary in degree without the presence
of actual disease. This fact is expressed by the use of such terms
as “perfect,” “strong,” “feeble,” “delicate,” in speaking of the
health of the same person at different times, and also as distinguishing
one person from another. The constitution is an
important factor in resisting disease, and a robust constitution may
determine recovery from a severe illness, while the patient with a
feeble constitution falls a victim to it.
The constitution of an individual is partly acquired, partly
inherited. A feeble or delicate constitution may be acquired by
unhygienic conditions, such as deficient exercise, the prolonged
breathing of impure air, unhealthy occupations, some imperfection
in diet, or dissipation.
But while many a robust constitution is enfeebled by such
conditions, a weak constitution may happily be strengthened by
careful and prolonged attention to the laws of health. This is
especially well seen in the case of those who strengthen their
muscular system by carefully-graduated and not excessive exercise.
Heredity has a great influence on health. As a rule the
children of healthy parents are robust, and on the contrary, any
“weak point” in the parents’ constitutions is liable to be participated
in by their children. Both mental and physical conditions may be
inherited. A peculiar habit of mind, as well as the same expression
of features, may be inherited.
As regards physical diseases, the influence of parents is not less
remarkable. The son of a gouty father requires to be particularly
abstemious in order to avoid his father’s disease. Certain specific
febrile diseases, e.g., enteric fever, diphtheria, and still more rheumatic
fever, are hereditary in the sense that the members of certain families
are more prone to them than others. Insanity, epilepsy, asthma,
neuralgia, and hysteria are also hereditary in the same sense, and
it is noticed that they occasionally alternate in different generations.
Cancer, consumption, certain skin diseases, and a tendency to the
early onset of degenerative diseases, appear also to occur more often
in certain families than in others.
In most cases it is the tendency to disease which is transmitted,
and not the disease itself. When an actual disease is inherited, as246
happens very rarely in tuberculosis and often in syphilis, the actual
infection is transmitted before birth from the parent.
A peculiarity of form, character, or tendency to disease has been
known to disappear in one generation and re-appear in the next;
this variety of heredity is termed atavism. The evidence showing
the inheritance of acquired characters, i.e. those which arise in
consequence of the effect of external forces on the organism is not
conclusive. Weismann believes that only those forces that influence
the germ-plasm are inherited. It must be admitted that the instances
of inheritance of acquired characters can be better explained otherwise.
Thus the long neck of the giraffe was formerly explained on
the supposition that the neck became gradually lengthened owing to
the efforts made generation after generation in reaching food; but is
better explained by Weismann on the supposition that those giraffes
which, during times of famine were able to reach higher and obtain
food from the twigs of trees would survive and pass on their
characteristics to their young, while shorter necked giraffes would be
exterminated.
The inheritance of proclivity to or immunity from attacks of
infectious diseases is a problem of great difficulty; but there is no
substantial reason for thinking that the efforts being made to
diminish the prevalence of these diseases (including consumption)
are likely to produce a weaker race or one more likely to suffer with
excessive severity from these diseases should they be introduced
after a long absence. (See also page 309).
Temperament indicates a peculiarity in constitution, causing
a liability to particular diseases, or to a special character in any
disease to which a person becomes subject. Four temperaments
are usually recognized—the sanguine, phlegmatic, bilious, and
nervous, but unmixed specimens of these temperaments are rarely
seen.
By idiosyncrasy is understood a peculiarity limited to a
comparatively small number of individuals. Four varieties of
idiosyncrasy may be described.
The first consists in an extreme susceptibility to the action of
certain things, or an extreme lack of susceptibility. Thus most
people at some time or other inhale the pollen of grasses, but only
in a few cases does it produce that troublesome and distressing
complaint—hay asthma. In certain persons a very minute dose of
iodide of potassium produces distressing symptoms; in most cases
these symptoms arise if the drug is taken for a prolonged period;
but in a few cases it may be taken for an indefinite period without
troublesome result. The case of a physician at Bath is very curious.
The smell of hyacinths in bloom always made him faint away; so
constant was this result, that before entering a room during the
hyacinth season, he always asked the servant if there were any
hyacinths in it.
The second form of idiosyncrasy consists in the production of
poisonous results by common articles of diet. Thus some people
cannot partake of shell-fish or lobsters without having severe nettlerash.
In rare instances the smallest amount of egg, or in other247
cases mutton, or pepper, or some other substance will produce
severe indigestion or nettlerash.
The third form consists in an inversion of the usual effects of
certain substances, especially drugs. Thus opium in rare cases
produces convulsions; while the aperient Epsom salts have been
known to produce constipation.
A fourth form, that of mental idiosyncrasies, may be added, as
where there is a strange preference or aversion for objects usually
regarded as indifferent. Many cases of mental peculiarity, short of
actual insanity, will come under this head; as will instances of
depraved appetite for food, etc.
Age and Sex.—According to the period of life, danger arises
from different sources. In infancy and old age extreme changes of
temperature are especially dangerous, and additional protection is
required (see also page 271). Thousands of deaths occur in the first
year of life, from substituting starchy foods for milk, the natural
food for infancy and childhood (see page 303). In childhood the
danger from bad feeding is still present, and is evidenced by the
frequency of rickets (page 28); infectious diseases claim their
thousands; and the disorders associated with dentition are common.
In youth rapid growth is proceeding, and so the food must be
abundant and nutritious. A proportionately larger amount is
required than by an adult, as the functions of the body not only
require to be carried on, but material is necessary to build up the
growing tissues.
Manhood is the period of greatest stability of health. The
health now depends on the use made of the previous periods of life,
and on the habits acquired.
With the onset of old age come various degenerative diseases.
The tendency is to death by gradual decay—a euthanasia or easy
death, which is too seldom seen. Commonly, bronchitis or
apoplexy or kidney diseases bring the scene to a somewhat
premature end.
The mortality of man is greater than that of woman at all
ages except 5—20.
Habits.—The immense power of habits in the formation of
character is perhaps duly appreciated; but their influence on
physical health is not so well appreciated; though it would be
difficult to exaggerate it. The laws of health are as inexorable
and unaltering as all other laws of nature; and whether broken
through carelessness or ignorance, the Nemesis of disease inevitably
follows. Whatever a man sows he reaps, in health as in other
matters.
Habits are easily formed; but, when once formed, not so easily
broken. They ought to be our servants; very commonly they
become our masters.
In reference to eating and drinking, habits regular as to time
and moderate as to quantity are especially important. The habit
of eating hastily and masticating the food imperfectly, is certain,
sooner or later, to produce disease. Over-eating, again, is a fertile
source of disease, especially when the excess is in animal food.248
The amount of stimulation produced by a given dose of alcohol,
gradually diminishes with its repetition; the consequence is, that
in order to produce the amount of stimulation to which the system
has become habituated, the stimulant requires to be gradually
increased. The craving for stimulants is often a sign of ill-health,
owing to disregard of hygienic laws or actual disease. Not infrequently
it is due to badly-ventilated rooms or long hours of work
without food, producing a sense of depression which food does not
immediately allay. When the cause is unknown, recourse should
be had to competent medical advice, and not to the brandy bottle.
Attention to the Action of the Bowels is a matter which is
commonly neglected. The importance of a regular habit in this
respect cannot be exaggerated; the bowels should always be
relieved at a particular time each day. Where this does not
occur the condition of constipation results. Owing to the retention
of the fæces in the intestines beyond the normal period, the
stomach and higher parts of the intestines do not perform their
functions normally; indigestion and “dyspepsia,” accompanied
by headache, flatulence, and other symptoms follow. Hæmorrhoids
(piles) are another frequent consequence. At the junction of the
small and large intestines is a dilated sac (in the cæcum). This
becomes distended when constipation occurs; inflammation may be
set up, and an operation required, or the condition is fatal. Powerful
purgative medicines are injurious to the bowels, and they tend
afterwards to increase constipation. It is better to take slow-acting
aperient remedies, and better still not to take any at all, but relieve
the condition by means of such articles of diet as stewed fruit, pears,
figs, olive oil, or brown bread. As a rule more exercise is required
in this condition, and always a prompt attention to the calls of
nature.
249
Physiological Considerations.—In the strict sense of the
word, exercise signifies the performance of its functions by any
part of the body; thus, digestion is exercise of the stomach, respiration
is exercise of the lungs, thinking is an exercise of the brain.
But the term is usually applied chiefly to muscular contraction, and
restricted to contraction of voluntary muscles. Involuntary
muscles, which are concerned in the carrying on of the unconscious
organic functions of life, are not directly controllable, and so their
growth and state of nutrition cannot be regulated. There are two
sets of involuntary muscle, which are of special importance—the
heart and the muscles of respiration. The heart contracts at least
sixty times per minute; the respiratory muscles contract about
seventeen times per minute; and this amount of exercise goes on
throughout the whole day. But although we cannot make our hearts
beat quicker by a direct volition, and cannot breathe more rapidly
than usual beyond a few seconds, yet a brisk walk will cause
increased action of the heart and respiratory muscles, as well as a
vigorous contraction of the muscles directly concerned in walking.
Going uphill is a valuable exercise for the heart. The vermicular
contractions of the intestines are to some extent also increased by
voluntary exercise, through the indirect excitation of the whole
system; thus, exercise is an important element in the treatment of
constipation.
The muscles contain about a fourth of the whole blood of the
body, and a very large share of the metabolism (page 4) of the body
occurs in them.
Hence the importance of keeping them in a healthy condition
by exercise. The great danger is of not equilibrating the muscular
and nervous functions. The ideal condition is where neither mental
nor muscular culture is neglected, but both are co-ordinated to the
production of a healthy man.
Effects of Healthy Exercise.—1. The Nutrition of the Muscles
is improved. The volume, density, and energy of the muscles are
increased.
2. The action of the lungs is increased. Dr. E. Smith found that
if the air inspired while lying down be represented by unity, the
amount inspired when erect is 1·33; when walking at the rate of one
mile per hour, 1·9; at four miles per hour, 5; at six miles per hour,
7; riding on horseback, 4·05; swimming, 4·33. Or, putting it in250
another way, under ordinary circumstances a man inspires 480 cubic
inches per minute; if he walks four miles per hour, he inspires
2,400 cubic inches; if six miles an hour, 3,260 cubic inches.
At the same time the amount of carbonic acid expired is
increased. Its amount bears a nearly constant relation to the
amount of muscular exercise, and consequently the amount of
carbonic acid eliminated in various forms of exercise affords a just
estimate of their relative value. The increased elimination of
carbonic acid, the corresponding increased absorption of oxygen,
and the absence of increase of elimination of urea are shown in the
following summary of observations by Pettenkofer and Voit:—
|
ABSORPTION OF
OXYGEN IN GRAMMES. |
ELIMINATION IN GRAMMES OF |
| CARBONIC ACID. |
WATER. |
UREA. |
| Work day |
955 |
1284 |
2042 |
37·0 |
| Rest day |
709 |
912 |
828 |
37·2 |
The above amounts are for the entire day. During actual
exercise the excess of elimination of carbonic acid is much greater.
Thus, Dr. E. Smith experimentally found that if the amount of
carbonic acid eliminated during rest be represented by one, the
amount walking at two miles an hour and carrying 7 lbs = 1·85,
the amount walking at three miles an hour = 2·64.
Alcohol diminishes the excretion of carbonic acid, and should
therefore be avoided during muscular training.
By muscular exercise the size of the lungs is increased, and
their vital capacity, that is, the amount of air capable of being
expired after a forced inspiration, is considerably increased.
Corresponding with this increase of vital capacity, exercise,
especially that in which the arm and chest muscles are systematically
developed, increases the size of the chest. A perceptible
difference in the circumference of the chest may be noticed after
only a few weeks’ methodical exercise.
3. The action of the skin is increased.—Sensible perspiration is
commonly induced, but less readily in those habituated to hard
work. Insensible perspiration is always increased.
4. The temperature of the body is not increased, so long as
perspiration occurs. Every muscular contraction involves the
production of heat; but this is counteracted by increased evaporation
from the skin, and by the circulatory current carrying the hotter
blood to every part of the body, and so rapidly equalising its
temperature. Chilblains are due to the defective circulation of the
blood, and can in most cases be cured by active exercise aided by
warmer clothing and an abundant supply of oxidisable food.
5. The Heart and Blood-vessels.—By exercise the heart’s action
is increased in frequency and force. The pulse usually increases
from ten to thirty beats per minute above the rate while at rest.251
After prolonged exercise it may temporarily fall below the normal
standard.
6. The Digestion and assimilation of food are aided by exercise,
especially when taken in the open air.
7. The nervous system is improved in nutrition and power by a
moderate amount of exercise. In fact, a certain amount of muscular
exercise is essential for a healthy mind.
8. The elimination of urea is not increased by exercise. Evidently
then it is not the metabolism of the nitrogenous substance of the
muscles which supplies the energy for muscular contraction; but of
the other oxidisable and non-nitrogenous substances (such as
glycogen and sugar) contained in them.
In practice it is found that with exercise more nitrogenous and
non-nitrogenous food are both required.
Effects of Excessive Exercise.—After prolonged exertion
muscles become exhausted. This is associated with an accumulation
in the muscles of the products of their action (especially sarcolactic
acid). Then rest becomes necessary, in order that the effete
products may be removed, and the nutrition of the muscles restored.
Long-continued over-exertion produces chronic exhaustion, which
may, if excessive, cause wasting of muscles. Exhaustion is much
more liable to occur when a small group of muscles are exercised
out of all proportion to others. Thus, in clerks, we have what is
known as the writer’s or scrivener’s palsy. The muscles of the hand,
and especially of the thumb, cease to respond to the volition of
the writer, but are seized with spasm every time writing is
attempted; and the muscles of the thumb tend to waste. A similar
condition sometimes arises in violinists, tailors, etc. The practical
inference from these facts is, that one group of muscles should not
be exercised disproportionately to the muscles of the rest of the
body, and that proper intervals of rest should be allowed.
Excessive exercise of the whole muscular system is very apt to
harm those of previously sedentary habits. A walking tour entered
on with more zeal than discretion, and not taken by easy stages for
the first few days, is often productive of more harm than good.
In the intervals of great mental labour, as with students, the
amount of exercise should not be suddenly increased, but should be
regular and moderate in amount.
Competitive exercise should be strictly regulated. The Oxford
and Cambridge crews have been said to acquire heart-disease more
commonly than the average, but this is not correct. Hypertrophy
of the heart may occur as the result of severe exercise, and this
within certain limits is not an abnormal condition. Occasionally
dilatation of the heart has been produced in weakly lads.
Amount of Exercise Desirable.—According to Parkes, the
average daily work of a man engaged in manual labour in the open
air is equivalent to the work involved in lifting 250 to 350 tons one
foot high; this is a moderate amount, 400 tons being a heavy day’s
work. The amount of muscular exercise involved in this may252
be easily known by remembering that a walk of 20 miles on a level
road is equivalent to about 353⅔ tons lifted 1 foot; and that a walk
of 10 miles while carrying 60 lbs. is equivalent to 247½ tons lifted 1
foot. (Haughton).
The amount of work done by a healthy adult per diem is stated by M. Foster
to be about 150,000 metre-kilogrammes (i.e., 150,000 kilogrammes lifted 1 metre).
Metre-kilogrammes can be converted into foot-pounds by multiplying by 7·233; into
foot-tons by multiplying by ·003229; 150,000 metre-kilogrammes therefore equal
484·35 foot-tons. This is considerably in excess of Parkes’ estimate, but in
certain laborious occupations this high amount is reached.
In addition to this amount of external work, there is the internal work of the
heart, muscles of respiration, digestion, etc. This is estimated by Parkes at
about 260 foot-tons.
The internal and external muscular work of the body together amount to
about 1 ∕ 7th to 1 ∕ 8th of the total force obtainable from the food.
Every healthy man probably ought to take an amount of
exercise represented by 150 tons raised 1 foot, which is equal to the
work done in walking 8½ to 9 miles on a level road. A certain
amount of this exercise is taken in performing one’s daily work; but
apart from this, out-door exercise should be taken equivalent in
amount to a walk of five or six miles. It is impossible to lay down
rules to suit all cases, but a less amount of exercise than that named
is probably incompatible with perfect health.
Effects of Deficient Exercise.—The muscles themselves
become enfeebled and wasted. Some wasting of muscle occurs after
a few days’ confinement to bed; and a limb confined in a splint
speedily loses its healthy, rounded contour. Oxidation processes are
diminished; less carbonic acid is eliminated, and it tends to
accumulate in the system, owing to the diminished activity of
respiration. In consequence of the diminished oxidation, the
temperature of the body is not well maintained, and the heat is not
uniformly distributed. Cold feet are a common complaint of those
who lead sedentary lives, though seldom complained of by others.
Along with the other muscles, the heart becomes enfeebled and
the circulation less perfect. Digestion is enfeebled; the appetite is
poor. The nervous system also suffers; nervous irritability is a
common result, while sleeplessness—a thing almost unknown
among those who live by the sweat of their brow—is becoming
much more common among the worried and ill-exercised inhabitants
of our towns.
Many diseases are favoured by deficient exercise, and can be
averted by systematic exercises and the concomitant increased
supply of pure air. It is often difficult to appraise the relative
merits of exercise and pure air; but there can be no doubt that both
are of extreme importance.
The prevention of consumption, even in those with a strong
hereditary tendency, is greatly helped by systematic exercises,
especially those directed to the expansion of the chest cavity. In
cases of consumption there is commonly a history of deficient
exercise or an occupation involving a cramped position, as well as
of living in an impure air.
253
Various deformities are induced by defective exercise of particular
groups of muscles. Thus drooping shoulders may be caused by
shoulder-straps confining the action of the shoulder-muscles in the
earlier years of life. Stooping is favoured by sitting in cramped
positions in school, and by the use of desks not inclined at the
proper angle. Lateral curvature of the spine is due to weakness of
the muscles of the back, and is best treated in its earlier stages by
gymnastic exercises specially directed to strengthening these
muscles. The tendency to such curvatures is greatly increased in
girls by the fact that their trunks are imprisoned in corsets as if in
splints, and so exercise of the trunk muscles is reduced to a
minimum.
Rules respecting Exercise. 1. The clothing during exercise
should not be excessive, and should not interfere with the free play of
the limbs, nor with full expansion of the chest. Flannel is the best
material to wear next the skin, as it absorbs perspiration without
becoming non-porous.
2. Avoid chill after exercise. It is well, if there has been any
perspiration during exercise, to strip and scrub the skin, particularly
about the chest and arm-pits, with a rough towel.
3. Exercise should be systematic and regular. It is important to
avoid sudden, violent, and competitive exercise. No severe exercise
ought to be undertaken without a gradual training.
4. The amount of exercise must be regulated by individual fitness. A
chain is no stronger than its weakest link. The muscles may be
stronger than the heart or lungs, and the latter may be fatally
injured by an amount of exercise which the muscles can well bear.
Hence the importance of ascertaining the condition of the vital
organs before entering on a course of training.
Another important bearing of this rule is in relation to the
exercise of growing boys and girls. When we remember that a
boy at school will sometimes grow six to eight inches in a year, it is
evident that much energy is being expended in this direction, and
that excessive gymnastic exercise can only do harm. Between the
ages of fifteen and seventeen there is usually the greatest amount of
physical development, and if there is great muscular strain at this
period, growth is interfered with, and the power of resistance to
disease may be seriously lowered.
5. Every part of the body ought to be exercised. This is done
spontaneously by the infant. Every muscle of his body acts in
sheer delight. The evils of exercise confined to particular groups of
muscles have been already described. Lawn tennis is very valuable
as affording exercise for both limb and trunk muscles.
6. Exercise should not be taken immediately after meals, as thus
digestion is interfered with.
7. Exercise should be taken, as far as possible, in the open air. A
small amount of exercise out of doors is much more invigorating
than a large amount indoors.
The Forms of Exercise taken may be divided into recreative254
and educational, though both of course may be recreative under
many circumstances.
The primarily recreative exercises, such as rowing, cricket,
football, tennis, hockey, will, it may be hoped, be never replaced by
educational gymnastics, though the latter possess a high value.
The recreative influence as well as the influence on the power of
self-control of such games as cricket and football render them of
national importance.
Educational gymnastics can be applied to exercise the muscles
of any part of the body, and can be exactly graduated to individual
requirements. Singing, speaking, and reading aloud, are forms of
muscular exercise very much neglected, and they are particularly
important, as the lungs and voice are by these means greatly
strengthened, and rendered much less liable to the inroads of
disease.
Professor Haughton has shown that the work done by a man walking on a
level surface at the rate of three miles an hour is equivalent to raising his own
weight, plus the weight he carries through 1 ∕ 20 of the distance walked.
- Thus, if W = weight of the man,
- W1 = weight carried by him,
- D = distance walked in feet,
- C = co-efficient of traction (1 ∕ 20, at three miles an hour),
then we obtain by the following formula the amount of work done, the co-efficient
of traction being multiplied by 2,240 (the number of pounds in a ton) to obtain
the result in foot-tons.
(W + W1) × D ∕ (C × 2,240)
In ascending a height, a man raises his whole weight through the height
ascended.
A regiment of soldiers marches ten miles, each carrying a weight of 60 lbs. What
amount of work is performed by each soldier?
If we assume the average weight of each soldier to be 150 lbs., and that the
march was at the rate of three miles an hour, then—
(150 + 60) × 10 × 5,280 ∕ (20 × 2,240) = 247·5 foot-tons.
In this example it is assumed that the march is on entirely level ground
that all weights are carried in the most convenient manner, and that the rate of
travel is three miles an hour. Velocity is gained at the expense of carrying
power. It has been found that the amount of work is generally inversely as the
square of the velocity. Haughton has determined from Weber’s calculations
the co-efficient of resistance for three velocities.
| VELOCITY. |
CO-EFFICIENT OF TRACTION
OR RESISTANCE. |
| 1·818 miles per hour |
1 ∕ 28·27 |
| 4·353 miles per hour |
1 ∕ 13·70 |
| 10·577 miles per hour |
1 ∕ 7·51 |
Parkes has extended these calculations to show the distance in miles255
required to be travelled at various velocities to do work equal to 300 foot-tons,
and the time required in each instance.
| VELOCITY IN MILES PER HOUR. |
CO-EFFICIENT OF RESISTANCE. |
DISTANCE FOR MEN OF 156 LBS. TO EQUAL 300 FOOT-TONS. |
TIME REQUIRED IN HOURS AND MINUTES. |
| 2 |
1 ∕ 26·74 |
12·2 |
hrs.
10 |
mins.
36 |
| 3 |
1 ∕ 20·59 |
16·3 |
5 |
24 |
| 4 |
1 ∕ 16·74 |
13·3 |
3 |
18 |
| 6 |
1 ∕ 12·18 |
9·6 |
1 |
36 |
| 8 |
1 ∕ 9·60 |
7·6 |
0 |
57 |
| 10 |
1 ∕ 7·89 |
6·3 |
0 |
38 |
The co-efficient 1 ∕ 20, corresponds very nearly to 3·1 miles per hour, and it
appears that at this rate of travel the greatest amount of work can be done with
the least expenditure of energy.
How much work is done by a man weighing 150 lbs. who walks 15 miles up an
incline 1 in 200?
The number of feet ascended in 15 miles
= 5,280 × 15 ∕ 200 = 396.
The amount of work done by the man in raising his own weight 396 feet high
= 396 × 150∕ 2,240 = 26·5 foot-tons.
The amount of work done in walking 15 horizontal miles at the rate of 3
miles an hour
= 150 × 15 × 5,280 ∕ (20 × 2,240) = 265.2 foot-tons.
Total amount of work done = 265.2 + 26.5 = 291.7 foot-tons.
Eight palanquin bearers carry an officer weighing 180 lbs. and a palanquin weighing
250 lbs., a distance of 25 miles. Assuming that each man weighs 150 lbs., what amount
of work was done by each man? (Parkes.)
- 250 + 180 = 430
- 150 × 8 = 1,200
- ———
- W + W1 = 1,630
- 1,630 × 25 × 5,280/(20 × 2,240) = 4,802·7 foot-tons.
This being the total work done, the work per man = nearly 600.3 foot-tons.
A hill-coolie weighing 150 lbs. goes 30 miles with an ascent of 5,500 feet in three
days, carrying 80 lbs. in weight. What is the work per day? (Parkes.)
Work of the ascent = (150 + 80) × 5,500 ∕ 2,240 = 564·7 foot-tons.
Work of 30 miles walk = 230 × 30 × 5,280 ∕ (20 × 2,240) = 813·2 foot-tons.
Total work = 564·7 + 813·2 = 1,377·9.
Total work per day = 1,377·9 ∕ 3 = 459·3 foot-tons.
256
Suppose a man weighing 150 lbs. in his clothes, carries a load of bricks weighing 35
lbs. up a perpendicular ladder 30 feet high, 100 times daily, what amount of work does he
do; and what will it equal in miles walked upon a flat road at the rate of 3 miles an
hour?
- (150 + 35) × 30 × 100 ∕ 2,240 = 247·8 foot-tons
- (185 × D) ∕ (20 × 2,240) = 247·8.
- Therefore D = 60,056 feet>
- = about 11·4 miles.
Suppose a man strikes 12,000 strokes in 5 hours with a 14-lb. hammer, raising it at
each stroke 4 feet, how much work does he do? Compare this with a walk of 15 miles
on a level ground at 3 miles an hour, the weight of the man and what he carries being
180 lbs.
- (a) 12,000 × 14 × 4 = 672,000 foot-lbs. of work
- = 300 foot-tons.
- (b) (180 × 15 × 5,280) ∕ (20 × 2,240) = 318·2 foot-tons.
The two amounts of work are related as 300 : 318·2.
257
CHAPTER XXXVIII.
PERSONAL HYGIENE (continued)—REST AND SLEEP.
Physiological Considerations.—Life is made up of alternations
of rest and action. The exercise of any organ is followed by
a necessary period of repose, during which the oxidised materials
produced by functional activity are removed by the blood, and
carried to the excretory organs; while at the same time fresh
nutritive material is supplied by the blood to make good the losses
thus sustained.
The only apparent exceptions to this rule of alternation of rest
and exercise are the heart and lungs, and some less important organs
acting out of the control of personal volition. But even these
organs obey the universal law. The difference is that their rest is
very frequent and momentary; the heart having to contract sixty
or seventy times per minute, rests 6 ∕ 11 of a second each second, or
more than thirteen hours in the twenty-four. The lungs and
respiratory muscles rest a shorter time than this, but probably about
three hours per day.
The necessity for rest is well shown by the sense of taste. If
salt is kept in the mouth for a considerable time, the power of
tasting it disappears, and only returns in its original strength after
several hours. The gustatory nerve has been exhausted.
The other sense-organs illustrate the same principle. Persons
are not uncommonly made deaf by the sounds of machinery.
After looking at a particular colour for some time, the nerves
receiving impressions from this colour are exhausted, and only
its complementary colour is visible.
Rest may be either partial or general.
The principle of partial rest has very useful practical bearings.
Such rest is illustrated by the student who takes a walk, or uses
methodical gymnastic exercises; a concert may provide agreeable
exercise for the auditory nerves and the part of the brain connected
with them, while allowing the over-tired intellectual part of the
brain to rest in peace; similarly, light literature may prove a
pleasing rest after severer studies.
Walking is more especially the exercise of the brain-worker.
Partial rest is the same thing as change of occupation, and by a
careful regulation of the relative amount of cerebral and muscular
work, energy can be largely economised. The horse, which exercises
chiefly his muscles, requires only five or six hours to recuperate his
energy; and our muscles require less sleep than our brain.
258
Sleep is the only form of complete and general rest. In
attaining this condition, the muscles sleep first, then the eyes close
(owing to muscular rest), and the thoughts wander; hearing is the
last sense to lose cognizance of the surrounding world; dreaming
succeeds wandering thoughts, and even dreaming may cease if the
brain repose is complete.
During sleep the brain diminishes in size, and becomes paler;
the amount of blood in the brain being diminished. Probably the
cerebral anæmia is rather a consequence of the functional inactivity
of the brain during sleep than a cause of the sleep.
During sleep the heart and lungs continue their work; the
blood is circulated and purified, the intestines continue their
vermicular contractions, and absorb food from the alimentary canal,
and the organs nourish themselves at leisure.
Two facts relating to sleep have important practical bearings.
First, during sleep metabolism is less active, and so the temperature of
the body tends to be somewhat lowered. Secondly, assimilation is
more energetic; this favours the absorption of noxious vapours, if
any are present. There is probably, therefore, slightly less danger of
remaining in a stuffy, impure atmosphere during the day than at
night.
Practical Rules Concerning Sleep.—1. Amount of sleep
required. It is impossible to lay down any fixed rule applicable to
all persons and circumstances. The amount of sleep required, like
the amount of food, varies greatly.
Habitual deficiency of sleep produces a condition of wretchedness
and prostration, with great restlessness. Prolonged watching
inevitably breaks down the constitution. Not the least evil
consequence of irregular and deficient sleep is, that sleep, when
desired, is often courted in vain.
Habitual excess of sleep produces a condition of brain less active
than usual, and less favourable for thought and action. Impressions
are received less readily, and the power of will is
correspondingly diminished.
The amount of sleep required varies with—
(1) Age.—The infant, if healthy, spends the larger part of his
existence in sleep; gradually the amount required diminishes until,
for the adult, seven or eight hours suffice. Children over two
or three years old require sleep only during the night. In advanced
life there is a tendency to revert to infantile habits, sleep occurring
in frequent short snatches.
(2) Sex.—Women have been stated to require rather more
sleep than men, but this is doubtful. The hours of sleep required
have in accordance with this view been stated to be, “Six for a
man, seven for a woman, and eight for a fool.” A reversal of
this order would more nearly approximate to the requirements of
town life.
(3) Temperament.—Those of a cold lymphatic temperament
require more sleep than sanguine or nervous people, though the
latter sleep more deeply. Frederick the Great, John Hunter, and259
Napoleon I. are said to have required only five hours’ sleep per
day; but the last of these had the faculty of taking short naps at
a few moments’ notice.
(4) The sick and convalescent require much more sleep than those
who are healthy.
(5) Habit has a very important influence. Many people appear
to sleep too much, and thus dull to some extent their mental
faculties; but on the other hand, modern life, with its nervous
strain, keen competition, and constant hurry and worry may make
a larger amount of sleep necessary than that required by our
forefathers, who invented the foregoing proverb.
(6) Occupation.—Mental work requires more repose than
physical.
2. Relation of sleep to food.—The molecular life of the tissues—that
is, the processes of nutrition—ought to be undisturbed. These
go on most perfectly when no active function, such as that of
digestion, is being performed. But while the stomach carries on
the digestive functions to only a small extent during sleep, the
intestines continue still to digest and absorb food. In accordance
with these facts, it is advisable to allow at least two hours between
the last meal of the day and sleep, especially if animal food has
been taken.
3. As absorption is increased and the temperature is lowered
during sleep, it is important to sleep in pure air, and to have warm
coverings, especially about the shoulders and arms. Many an
obstinate cough might be cured by the simple expedient of wearing
a flannel jacket at night.
4. Sleep during the night and not during the day. It should hardly
be necessary to say this, as the universal instinct of animals shows
its advisability; but, unfortunately, the habits of mankind have
commonly led to a partial reversal of the natural arrangement.
5. The room should be dark; light, like sound, is inimical to
sleep. The head should be moderately raised. The temperature of
the room for robust persons need not be artificially raised.
Sleeplessness, as a rule, occurs only when some physiological
law has been broken. To relieve it, it is essential to equilibrate
muscular and mental functions. Increase of muscular exercise is
an important element in its treatment. In addition it is advisable
not to have any severe mental work during the evening, nor to
indulge in late suppers. Sleeplessness is the bane of many men of
a nervous temperament, and chiefly attacks those of sedentary habits.
It is apt to recur, and for this reason, if for no other, narcotics
ought to be scrupulously avoided. The habit of taking such
soporifics is unfortunately becoming much more common, and is
productive of many evils. Death from accidental overdose is a
frequent calamity; and, apart from this possibility, the invalid’s
nervous system is completely ruined by persistence in the habit, his
power of will is annihilated, and he becomes the miserable slave of
an evil habit, whose end is death (see also page 54).
260
CHAPTER XXXIX.
PERSONAL HYGIENE (continued)—CLEANLINESS.
Physiological Considerations.—The skin consists of a superficial
part or epidermis, and a deeper part called the dermis or cutis.
Tubes of two kinds open on the surface of the skin, penetrating
at their deeper ends into the cutis, viz. sweat or sudoriparous glands
and sebaceous glands. The sudoriparous glands are simple tubes, the
lower ends of which lie coiled up in the dermis. Each tube when
straightened out is about a quarter of an inch long. It has been
estimated that in the palm of the hand there are 3,528 orifices of
sudoriparous and sebaceous glands on a square inch of surface;
reckoning each gland at ¼; inch long, this means 73½ feet of tubes
in this small space. Assuming that there are 2,800 tubes to every
square inch, and that the amount of surface in a man of ordinary
height and bulk is 2,500 square inches, it follows that there are
seven million pores in a man—that is, 1,750,000 inches, or nearly
twenty-eight miles.
The perspiration secreted by the sudoriparous glands is
constantly evaporating from the surface of the body. It is very
important that the orifices of these glands should be kept open in
order that the secretion may not be interfered with. Animals have
been killed by covering their skin with gelatine, and so preventing
the escape of perspiration.
The sebaceous glands are shorter than the sudoriparous, and
commonly end alongside the hairs before the latter issue from the
skin. They secrete an oily material which serves the purpose of
a natural pomade. The sebaceous secretion also keeps the general
surface of the skin unctuous and supple. The smell of the sebaceous
secretion may be unpleasant, especially in the arm-pits and some
other parts. Frequent washing is therefore desirable.
The Conditions Due to Uncleanliness are due to obstruction
of the excretory ducts, to accumulation of débris on the general
surface of the skin, and to the consequent interference with the
circulation.
1. The obstruction of the sudoriparous pores of the skin interferes
with the elimination of waste products by the perspiration; these
are re-absorbed or retained in the system; consequently more work
is thrown on the lungs and kidneys, and the equilibrium of health
is destroyed.
Sebaceous obstruction causes an accumulation of oily secretion
in the ducts. The black spots so commonly seen about the nose,261
are the blocked up orifices of sebaceous glands, and by squeezing
the nose tiny threads of fatty matter are forced out from the interior
of these glands. Pimples on the face are usually due to obstruction
of the sebaceous glands; sometimes the obstruction leads to
inflammation around the sebaceous gland (acne) which often permanently
injures the skin.
2. Accumulation of effete matter on the skin occurs, unless
frequent ablutions are performed. The epidermis is constantly
shedding its older and more superficial parts, in the form of minute
scales or “scurf.” In the absence of frequent washing, the scales
of epithelium tend to accumulate, the sebaceous secretion matting
the scales together, and rendering them more adhesive. The saline
matters of the perspiration also accumulate along with the scales
and sebaceous secretion, and in virtue of their hygroscopic properties
tend to keep the skin clammy and cold.
The obstruction of excretions and the accumulation of débris
lead to other consequences. Thus:—3. The sensibility of the skin
is dulled when the sensory papillæ are covered with dirt. The
sensations received by the skin are important in regulating the
temperature of the body. A cold external temperature should
cause a reflex contraction of the small arteries bringing blood to
the skin, thus diminishing the flow of blood and preventing undue
loss of heat. Similarly, if the external temperature is high, or the
internal development of heat is too great, these arteries dilate, and
sending more blood to the skin, cause a greater loss of heat by
radiation and conduction. Impaired sensibility of the skin leads
to imperfect action of the reflex nervous mechanism to which the
above effects are due, and consequently the dangers resulting from
sudden alterations of temperature are greatly increased.
4. The tendency to chills is increased, not only by deficiency of
the nervous tone of the skin, but also by obstruction of the pores
of the skin, and by the hygrometric action of the saline matter
collected on it.
5. Cutaneous diseases are due to, or favoured by, uncleanliness.
These are of two kinds—parasitic and non-parasitic. Acne, which
is the chief non-parasitic disease favoured by uncleanliness, has
been already mentioned.
Parasitic skin diseases are greatly favoured by the presence of
a dirty skin, which affords a suitable soil for the development of
the parasites. (See also page 275).
Uses of Soap.—Soap is produced by an action of an alkali on
an oil. The alkali displaces glycerine from the oil, and forms an
alkaline stearate, which is soap. Soft soap is chiefly stearate of
potassium; hard soap is stearate of sodium. There may also be
present the alkaline salts of oleic and palmitic acid. Soft soap is
not used for washing the skin, as it is too irritating. All soaps
contain a slight excess of soda; the greater this excess, the more
irritating is the soap to delicate skins. Hard soaps may be also
made with potash, if the fat employed be a solid one; but such
soaps are rather softer than ordinary hard soaps, and more caustic.
Cocoa-nut oil is used in making marine soaps, because, unlike all262
other kinds, it is not rendered insoluble by brine, and so will form
a lather with sea-water. Normal soaps contain from 15 to 35 per
cent. of water. “Liquoring” a soap consists in adding 5 to 25 per
cent. of soluble silicates. By this means the soap may be made to
hold 70 per cent. of water, which is obviously very wasteful.
In washing the skin, the water washes away a considerable
amount of epidermis, and the saline matters which have collected.
For the oily sebaceous secretion soap is required. The alkali in
soap combines with the oily matter, forming an emulsion which
carries away with it a quantity of the dirt which previously blocked
the orifices of the sebaceous and sweat ducts. When the skin is
rubbed by the towel after washing, the softened epithelium, and
with it any remaining dirt, are rubbed off, leaving the skin clean, and
able to perform its normal functions.
The Use of Baths.—The primary object of bathing is cleanliness.
A secondary consideration is the pleasure derived from
bathing. Baths are especially necessary for those persons who lead
sedentary lives. When the skin is kept in an active condition by
exercise, it to some extent cleanses itself. Thus, a farm labourer
who has a weekly bath, may be really cleaner than a person of
sedentary habits, who has two baths per week.
Baths are classified according to temperature as follows:—Below
70° Fahr. they are described as cold; tepid up to 85°; warm
up to 97°; and hot over this temperature. It is important in
deciding the temperature of a bath not to trust to one’s sensations;
the only accurate measure is by the thermometer. A cold morning
tub in the summer will commonly contain water at 55° to 60°;
while the same in winter will be down to 40°, or occasionally to 32°.
For purposes of cleanliness the warm bath is the most efficient,
combined with the free use of soap. The chief objection to it is
that it produces an increased flow of blood to the skin, by relaxing
the cutaneous blood-vessels, thus increasing the danger of chills if
there is subsequent exposure. The increased sensibility to cold
resulting from a warm bath may be obviated by afterwards rapidly
sponging the body all over with cold water, and then drying the
body quickly, and using the friction of a moderately rough towel.
It is desirable for both cold and warm baths to have a “bath-sheet,”
in which the person may be completely enclosed on coming
out of the bath. Drying is thus much more quickly accomplished,
and the danger of chill is minimised.
A daily morning cold bath is a most important agent in the
maintenance of robust health. The first sensation on entering a
cold bath is of shock, due to the cooling of the surface of the body.
This is followed in a few seconds by a glow, due to the blood
returning with considerable force to the skin. A cold bath ought
to be taken as rapidly as possible. If soaping the body is desired,
it should be done before entering the bath, and the stay in the latter
should be little more than momentary. In this way the best
reaction or “glow” is obtained.
If a feeling of cold and chilliness remains after a cold bath, it
has done more harm than good. This condition may often be263
avoided by quick drying and brisk friction; if after this a good
reaction is not obtained, the temperature of the water should be
increased. For those who are not very robust, the “cold tub” in
winter is to be deprecated. If the water be raised to 60° by the
addition of warm water, or in some cases even to 70°, a good
reaction may be obtained. In other cases, in which a reaction is
not experienced even after a bath of the latter temperature, a tepid
bath may be taken, and then the body rapidly sponged with colder
water.
Cold baths increase the tone of the skin, rendering it less
susceptible to changes of temperature. The tendency to “catch
cold” is diminished, the blood-vessels and nerves of the skin both
responding more readily to any stimuli.
Swimming is a valuable combination of bathing and exercise.
A sudden plunge into cold water for swimming purposes is
dangerous to those who are not hardened to it, and especially so
in the case of running water, as in rivers or the sea. Here the
water around the swimmer is constantly being changed, and each
layer of water coming in contact with him abstracts a considerable
amount of heat. Many of the cases of so-called death from
“cramp” are really due to the benumbing and depressing influence
of continued cold on the vital organs.
Swimming, under proper superintendence, ought to be universally
enforced. The exercise accompanying it serves in most cases
to counteract the depressing action of the cold water; but it is
important in all cases to attend to certain rules. The immersion
should not be prolonged; the body should be warm at the time of
entering the water; and the bath should not be taken until about
two hours after a meal; nor after prolonged fasting, as before
breakfast.
Personal Cleanliness.—Personal cleanliness involves not
only attention to the skin, which we have already considered, but
to the hair, nails, mouth, and other parts of the body.
The hair ought to be carefully brushed and combed, but it is
not desirable to use soap to it as often as to the skin. Soap
removes the sebaceous secretion from the hairs, and renders them
dry and brittle. Artificial pomades are, as a rule, unnecessary.
The nails should be cut square, and not down at the sides. It
is hardly necessary to say that they should be kept clean: they
may convey serious infection.
The mouth and all mucous orifices should be kept scrupulously
clean. A fœtid breath is not uncommonly due to the discharges
from carious teeth, or to the decomposition of food which has been
allowed to accumulate in the cavities of teeth. Such decomposing
matters when swallowed, are apt to produce indigestion; and this
also occurs from imperfect mastication of food by the bad teeth.
It is important that the teeth should be frequently cleansed, and
that all carious teeth should be “stopped” at an early period, and
tartar and other accumulations removed. Whether bad teeth,
which are so extremely common, are due to the drinking of very
hot liquids, or to the fact that the more perfect cooking of food264
gives less healthy friction to the teeth, is doubtful. Whatever the
cause, by keeping the mouth thoroughly sweet and clean, and by
having the carious teeth stopped as soon as discovered, their
vitality may be greatly prolonged. Teeth should be periodically
inspected by a competent dentist. Irregularities of the teeth may
be corrected, if they receive early attention. Carious “milk-teeth”
should receive attention from a dentist, as well as the permanent
teeth.
General Cleanliness.—Next to cleanliness of the skin, that
of the apparel is most important.
There is a general preference for colours “that do not show
the dirt”; the fact that it is still there, though not seen, being
partially ignored. Changing of apparel is commonly confined to
underclothing. It is forgotten that vests, trousers, dresses, etc.,
acquire a large amount of dirt and organic matter, and ought to be
changed and well aired at intervals.
Cleanliness in respect to bedclothes is very important. Organic
matters evolved from the skin, lungs, etc., hang about the bed-linen,
and give the bedroom the “close smell” which can be
perceived on entering it in the morning straight from the fresh air.
The beds should not be made directly after being evacuated, but
the clothes should be thrown over the bottom of the bed, the
bolsters and mattress well shaken, and every part exposed to a free
current of air during the greater part of the morning, before
re-arranging the clothes. Eider-down quilts, unless frequently
ventilated by exposure to outside air, are unwholesome. Superfluous
bedroom furniture should be avoided, as it all takes away
from the breathing-space. Bed-hangings should be reduced to a
minimum, and all excretory matters covered up during their stay
in the room, and removed as early as possible.
Cleanliness of the house is also very important as a means of
health. Dust, in however obscure a corner it rests, attracts to
itself organic matters, and forms a soil in which disease germs may
grow. Besides this, it devitalises the air, depriving it of its active
oxygen. (See also page 101).
Dust in the streets serves to carry about various diseases,
besides mechanically irritating any part it comes in contact with,
producing bronchitis, etc.
265
Physiological Considerations.—The average temperature of
the surface of the body in man is 98·4 to 98·6°. The maintenance
of a tolerably uniform temperature is an essential condition of life.
The factors governing the temperature of the body are the amount of
heat produced and the amount lost. If more heat escapes, more has
to be generated; and the source of all the heat produced in the body
is the food taken. This becomes changed by the metabolic
processes occurring in the body which produce heat.
Heat is lost, (1) by the skin; (2) in respiration, the expired air
having been heated during its stay in the lungs; (3) with the food
and drink taken, if not at the temperature of the body; (4) with the
excreta; and (5) by transformation of heat into mechanical energy.
Of the whole loss by these different channels, probably eighty to
ninety per cent. is through the skin.
The Loss of Heat by the Skin is in three different ways.
First, by conduction, when the skin comes in contact with anything
cooler than itself; secondly, by radiation into space; and thirdly, by
evaporation of the perspiration. The last cause produces a
considerable reduction of temperature, even when the perspiration
is not so abundant as to be visible, but is in the form of insensible
perspiration. The losses by these different sources vary in amount;
when one is increased, another is diminished, by way of
compensation. Thus, in very cold weather, the amount of radiation
and conduction of heat are increased; but evaporation greatly
decreases, and the diminished loss of heat in this respect counter-balances
in some degree the increased loss by radiation and
conduction.
When the external warmth is considerable, increased
evaporation occurs; while when the weather is cold, the cutaneous
arteries contract, and less blood goes to the skin, and so the loss of
heat is diminished. In most climates, however, this action of the
skin requires to be supplemented by some kind of clothing.
Requisites of Dress.—1. The first and most important
requirement is that clothing should maintain a uniform and equable
temperature in all parts of the body.
In hot climates clothes are required in order to protect the body
from external heat. In this country, they are required to prevent
the too rapid escape of heat from the body. For both these266
purposes, dress must be of a non-conducting material, in order
not to encourage transfer of heat into or from the surrounding
atmosphere.
The loss of heat by the skin may be prevented by interfering
with radiation or conduction of heat, or with evaporation from its
surface. Radiation of heat from the skin is prevented by clothing,
the dress taking the place of the skin as a radiating surface. The
amount of radiation from the dress will depend on the rapidity of
conduction of heat from the skin. The amount of conduction and
of radiation of heat will vary considerably with the material and
colour of the dress.
As regards conductivity, the two extremes are represented by
linen and fur. It is found that if the conducting power for heat of
linen = 100, then that of wool = 50 to 70. This partly explains
why woollen goods are so much warmer than linen. We shall find
that there is another explanation in the relative hygroscopic
properties of the two materials.
As regards radiation of heat, in one experiment it was found
that while a piece of linen took 10½ minutes to cool, a corresponding
piece of flannel took 11½ minutes.
Apart from the material, the colour of dress has some influence
in regulating the loss of heat. Dark-coloured materials absorb
more light and heat than lighter coloured materials; they may be
good or bad conductors of heat, according to the nature of the
material. White reflects the rays of light and heat; hence it is a
poor absorber. In summer it prevents the passage of heat inwards,
and, in winter, may prevent its passage from the body. It is thus
well adapted for both winter and summer clothing, and has the
additional advantage of being the cleanest colour.
Franklin placed a number of squares of different coloured
cloths of the same material on snow, and found after a time that
the snow covered by the black piece was most, and by the white
piece least melted. In another set of experiments, shirting materials
dyed various colours were taken, and it was found that if the rays
of heat received by white were represented as 100, pale straw
received 102, dark yellow 140, light green 155, Turkey red 165,
dark green 168, light blue 198, black 208.
The influence of colour is antagonised to a large extent by the
nature of the material; the increased heat absorbed by a dark
material may be counterbalanced by the material being a good
conductor. Also the influence of colour is only exerted superficially;
hence, although it produces considerable effect in thin textures, as
gauze, it has little influence on thick materials.
2. The dress should not interfere with perspiration. In order
that it may not do this, it should be competent to absorb
moisture easily, without its surface becoming wetted. Materials
like linen which lose their porosity and rapidly become
wetted by perspiration, cause rapid loss of heat from the
body, inasmuch as water is a better conductor of heat than
air. Pettenkofer found that while the maximum hygroscopic power
of wool (flannel) is 174 and the minimum 111; the maximum of267
linen is 75 and the minimum 41. Hence, with a flannel vest next
the skin, the liability to chill is much less than with a linen one.
There is one slightly counterbalancing drawback; hygroscopic
materials absorb moisture from the air, as well as from the skin. A
woollen coat during a damp day, without rain, increases considerably
in weight.
Waterproof clothing is injurious when worn beyond a short
period, on account of its being non-porous and consequently keeping
the body enveloped in a vapour bath composed of its own
perspiration. For a similar reason India-rubber boots are objectionable,
except for short periods; they make the feet damp, and
even sodden. Sealskin jackets are objectionable for walking, not
only because of their weight, but because they are not porous.
3. The warmth of clothing should be uniformly distributed
throughout the body. This principle is very frequently departed
from; and consequently one part may be chilled while another is
over-heated. This is seen especially in female apparel. The same
evil is seen in the short sleeves, and short and low-necked dresses of
young children. “Combination” garments for women, and sleeves
and leggings for young children are happily becoming more generally
adopted, and will diminish the diseases due to exposure to cold.
4. The clothing should not be tight; and this for three reasons.
First, because loose clothing is warmer than tight; this everyone has
experienced in the case of gloves. The retention of air in the
meshes of clothing is one of the main causes of its warmth, air
being a bad conductor of heat. The imprisonment of air in the
meshes of the material largely explains the warmth of eider-down
quilts, furs, and flannels as contrasted with linen.
Secondly, clothing should not be tight, in order to avoid interference
with the action of the muscles. Tight sleeves prevent the
muscles of the arms and chest from being exercised. Tightly laced
corsets imprison the trunk muscles, prevent their contractions, and
so lead to muscular weakness and occasionally spinal curvature.
Tight skirts similarly prevent free play of the lower limbs, leading
to a halting gait, a diminished amount of exercise, with all the evils
following deficient exercise. Tight clothing is not confined to one
sex, and in all cases leads to hampered movements and deficient
muscularity.
Thirdly, tight clothing tends to impede the functions of circulation,
respiration, and digestion. The fashion which more than any other
interferes with important functions is tight-lacing. This produces
(1) compression and displacement of the viscera; the liver and the
stomach especially suffer. (2) Respiration and circulation are
impeded, the action of the diaphragm being impeded. (3) The
muscles of the trunk being tightly encased, are incapable of
movement, and consequently tend to waste and atrophy. The
general outline of the body is altered. Instead of the waist being
elliptical, as it naturally is, it becomes nearly circular; and instead
of its circumference averaging twenty-six to twenty-seven inches, it
may be eighteen to twenty-one inches. Tight garters tend to produce
varicose veins.
268
Tight boots are injurious, as they tend to destroy the natural
elasticity of the movements, and confine them within narrow limits.
They act to some extent the part of splints. By interfering with
the circulation of blood through the feet, they cause cold feet, and
not uncommonly chilblains. High-heeled boots do not allow the
natural elasticity of the foot to come into action. They distort the
movements of the body and cause corns and bunions. Similar
effects are produced by boots which are too narrow and have pointed
toes, thus not allowing free movement of the toes.
5. The weight of the clothing should be the smallest amount consistent
with warmth, and it should be evenly distributed. The chief weight
should not be suspended from the waist, as here the parts are not
well supported by bones. The shoulders and hips should share in
the suspension of clothing, thus diminishing the danger of compression
and displacement of internal organs. In order that
garments may be as light as possible, they should be made to fit
to each limb separately, thus diminishing the amount of material
required.
6. The materials of dress should be as far as possible non-inflammable.
This may sometimes be disregarded, but is often
important, as in the nursery. In this respect, as in many others,
wool possesses great advantages. Woollen fabrics smoulder rather
than burst into flames, and thus the injury resulting from any
accident is limited. Cotton is more inflammable than linen, linen
than silk, and silk than wool. A closely woven cloth is less
inflammable than one with open meshes.
Dress materials, and more particularly muslin, have been
rendered non-inflammable by treating with a solution of ammonic
phosphate, or ammonic phosphate and ammonic chloride mixed.
The best material, however, is sodic tungstate, which, unlike the
others, is not affected by ironing. Sodic molybdate is used in
arsenals to render the workmen’s clothing non-inflammable. All
the above plans are objectionable, as the weight of the material
is increased 18 to 29 per cent., and they all wash out. To remedy
this, a “fire-proof starch,” containing sodic tungstate has been
devised.
Perfect non-inflammability is only required in certain dangerous
occupations. The plans hitherto mentioned simply prevent the
fabric breaking out into flame. The only cloth absolutely unaffected
by fire is asbestos cloth.
7. Elegance of dress, although not so important as utility, is not
to be neglected, and the two are perfectly compatible. In fact,
elegance is indirectly associated with utility, for nothing which is
awkward, or leads to obstructed movements or distortions of the
body, is really elegant. A sudden constriction, as in a very tight
waist, is not only bad from a hygienic point of view, but is also ugly.
Materials for Clothing.—The materials used are derived
partly from the vegetable world, as hemp, flax, cotton; and partly
from the animal world, as silk, wool, hair, feathers. The most
important materials are wool, silk, cotton, and flax.
269
1. Wool varies somewhat in character, according to the animal
from which it is derived. In all its varieties, however, it preserves
the character of a bad conducting and porous substance, the two
most important requisites in a dress material.
(1) Wool from the sheep is really a soft and elastic hair,
composed of fibres three to eight inches long, and about 1 ∕ 1000 inch
thick. The finer and shorter wools are used for fine cloth, the
longer and coarser for “poplins,” “worsted pieces,” etc. Flannel is
a woollen stuff of rather open and slight fabric. Wool is irritating
to delicate skins, and may be so much so, that it cannot be worn
next the skin, whether as flannel, worsted, or merino. In these
cases, it may be worn outside a linen or gauze vest, and so all its
advantages secured. It is one of the worst conductors of heat, and
ought always to be worn in winter; while even in summer, it
ensures a greater immunity from chill after perspiration than any
other material.
(2) Cashmere is made from the down found about the roots of the
hair of the Thibet goat. Imitation cashmere is made of various
materials mixed together.
Fig. 56.
Microscopical Appearance of Fibres of
A—Cotton. B—Silk. C—Linen. D—Wool.
(3) Alpaca is obtained from the fleece of the llama, alpaca, and
vicuna. It is longer than the fleece of the sheep, the fibres, which
are soft and strong, averaging six inches in length. It is commonly
made up with cotton or silk.
(4) Mohair is the hair of a goat inhabiting the mountains near
Angora. It is woven into an almost waterproof cloth, and used in
making plush, braid, etc.
2. Hair derived from the horse or cow differs from the hair
usually called wool, in the greater solidity of its structure, which
makes it ill adapted for clothing. Its chief use is in the manufacture
of felts, of which hats are made.
3. Leather is a kind of natural felt, very close and firm in its
texture. It is used in this country chiefly for boots, but in some270
colder climates also for coats, etc. It is impervious to moisture,
like sealskin, and is consequently not very healthy. The same
objection applies to chamois-leather underclothing, which is non-porous,
and consequently keeps the skin hot and clammy; also, it cannot
be washed without becoming stiff on drying. This necessitates
wearing the material after it has become impregnated with
perspiration.
4. Silk. The thread spun by the silk-worm is composed of
filaments 1 ∕ 2000 inch wide, and is the strongest and most tenacious
of textile fabrics. Its thread is three times as strong as a thread
of flax of the same thickness, and twice as strong as a thread of
hemp.
Its fibres are round like those of linen, but softer and smaller;
it gives an agreeable sensation of freshness to the skin even more
than linen. It is a worse conductor of heat than cotton or linen.
Its great disadvantage for wearing next the skin, apart from its
expense, is that it irritates delicate skins. Satin is silk so prepared
as to form a smooth, polished surface.
Velvet is a silk fabric of which the pile is due to the insertion of
short pieces of silk thread under the weft or cross-thread. Cheaper
kinds are made, containing a certain proportion of cotton.
Crape is made of raw silk gummed and twisted to form a gauze-like
fabric. Taffety, moire, brocade, and plush are made of silk
alone or combined with cotton.
5. Cotton consists of the downy hairs investing the seeds of the
gossypium plant. The threads of which it is composed are flat,
ribbon-like, and twisted, about 1 ∕ 800 to 1 ∕ 2000 inch wide. Owing to
its flat fibres with sharp edges, it is apt to irritate delicate skins;
linen is preferable for dressing wounds for a similar reason.
Cotton is warmer than linen, being a worse conductor of heat.
It also absorbs moisture better, not becoming wet so soon; but it
lacks the “freshness” which makes linen materials pleasant to
wear. Calico, fustian, jean, velveteen, and muslin are the chief
cotton fabrics.
6. Flax is formed from the fibres of the flax plant. Linen is
made from it. Cambric and lawn are very fine and thin linen
materials. The fibres of linen are round and pliable; thus it is
smooth and soft, and peculiarly agreeable to the skin. It is, however,
a good conductor of heat, and consequently “it feels cold” to
the skin. Furthermore its pores quickly become filled with
perspiration, which escapes rapidly, thus chilling the body.
7. Mackintoshes are valuable as a temporary protection against
external wet. Worn for more than a short period, they produce
great heat and a sense of closeness, owing to retention of the
perspiration. The best form of mackintosh is one having a cape,
with a space for evaporation between it and the rest of the garment.
The Amount of Clothing required varies with circumstances.
1. Health; those of robust constitution require less than the feeble.
The more active are digestion and assimilation, the less is the
amount of clothing required. If heat is preserved by clothing, less
food is required. Thus a distinct saving of food is effected by warm271
clothes. Warm clothes are the equivalent of so much food that
would have been required to keep up the temperature of the body,
if the clothes had not been worn. Thinly clad persons under
conditions of starvation die more quickly than those who are better
protected.
2. Clothing requires to be adapted to climate and season. In
winter and in cold climates the amount of clothing must be
increased, and warmer materials chosen. In the changeable
climate of Great Britain, it is difficult to adapt the character of
one’s dress to the requirements of the weather. Clothing ought,
however, not to be changed according to the calendar, but according to
the weather. The tendency is to assume summer clothing too early
in the spring, and to continue it too far into the autumn. According
to Boërhave, winter clothing should be put off on Midsummer day,
and resumed the day after. This, although rather exaggerated, may
serve to impress the caution required. The same authority says
that only fools and beggars suffer from cold, the latter not being
able to procure sufficient clothes, the former not having the sense
to wear them.
3. Age. Those at the two extremes of life are specially susceptible
to cold. The mortality of infants during the first three months
of life is nearly doubled in winter. Bronchitis and pneumonia
prove fatal chiefly at the two extremes of age.
The younger a child the larger is its surface as compared with
its bulk, inasmuch as the area of a body varies as the square of its
dimensions, while its mass varies as their cube. Thus a cube 1 inch
each side has 6 square inches of surface to 1 cubic inch of bulk,
while a cube 10 inches each side has 600 square inches of surface to
1000 cubic inches in bulk. Similarly a child 1 ∕ 10 the size of its
mother, besides its feebler powers of producing heat, has ten times
as much surface in proportion to its size by which heat is lost.
After the age of thirty-five, it is better to exceed than to be
deficient in clothing. A degree of cold that would act as a useful
tonic to the robust and middle-aged, produces serious and even
fatal depression of the vital powers in children or aged people.
For the same reason it is advisable to discontinue cold baths as age
advances.
A very pernicious delusion is prevalent, that children ought
to be “hardened” to the influences of cold, and that too much
clothing “makes them tender.” Excessive clothing may possibly
increase the tendency to “catch cold,” owing to its exciting
perspiration, or owing to the fact that the extra clothing is
commonly thrown off at irregular intervals—witness the effects of
wearing a scarf round the neck occasionally. But to suppose that
children can be hardened by exposure of arms and legs, and other
parts of their bodies, is irrational. A large amount of heat is lost
from these bare surfaces, and apart altogether from the danger
of chill, more food must be taken to compensate for this loss of
heat, and keep up an equable temperature. Also if the food taken
is expended in preserving the warmth of the unprotected body, less
material is left for the purpose of growth. From these causes it272
frequently happens that children remain stunted in growth, even if
latent disease is not actually developed by the extra strain on their
resources.
The children of the very poor are often pointed to as demonstrating
the power of hardening. It is forgotten how many of
these poor children have perished under the hardening system, and
that the good health of those remaining is in spite of the hardening.
Poisonous Dyes in Clothing.—These, like poisonous wall-papers,
were formerly much more common than at present, and, as
in wall-papers, the poisonous agent has most frequently been arsenic,
large quantities of which were formerly used in the preparation of
certain dyes. Occasionally such poisonous pigments are still
employed.
The means of detecting arsenic in any fabric or wall-paper are
given on page 216.
273
Parasites (Greek, para, upon, and siteo, I feed), in the broadest
sense of the word, are living organisms, which derive their nourishment
from other living organisms. They may belong to the
vegetable or animal kingdoms, and may live on the skin, in the
alimentary canal, or in some one of the internal organs. Some, like
the fungus causing ringworm, feed on the living tissues of the
animal infested; others, like tape-worms, on the partly digested
food; while other parasites, like fleas, only pay temporary visits to
the surface of the body, for the purpose of obtaining food.
Vegetable Parasites.—Vegetable parasites all belong to the
class of fungi, and more accurately to the two lowest divisions of
this class which have been provisionally formed, viz.—Schizophyta,
and Zygophyta. The Schizophyta include two orders, Schizomycetes
and Saccharomycetes.
BACTERIA.
Bacterium is the generic name given to the micro-organisms
belonging to the schizomycetes, whether a bacillus (rod-shaped),
coccus (rounded), spiral-formed (spirillum or vibrio), or filamentous
(leptothrix and spirochœta). All these are destitute of chlorophyll
and multiply by fission.9 They are all extremely small, the width
usually not exceeding 1 µ = 1 ∕ 25000 inch. Various names are given
to them, which are synonymous, thus: germs,
- microbes,
- micro-organisms,
- microzymes,
- bacteria (singular bacterium).
When they cause disease they are called contagia. They multiply
rapidly, and may reach maturity in 20 to 30 minutes. One
bacterium may, under favourable conditions, become 16,000,000
in 24 hours.
Methods of Examination.—Until Koch discovered the
method of cultivating bacteria on solid media, the science of
bacteriology remained in its infancy, as it was impracticable to
obtain pure, i.e. unmixed cultures of a given bacterium. Koch hit
on the idea of mixing minute portions of cultivations of bacteria274
which were growing in liquid broth with liquefied gelatine, and then
spreading the mixture on glass plates, and allowing it to solidify
under cover, so that no atmospheric bacteria could contaminate
the growth. When this was done, individual bacteria formed
individual “colonies” scattered over the gelatine, and these could
be identified by sub-culturing and other methods, for the details of
which books on bacteriology must be consulted.
The food supply of most bacteria is vegetable or animal refuse.
Some of them have a most useful purpose in nature, that of breaking
down complex organic substances and reducing them to a simpler
form. Thus bacteria play an essential part in purifying the soil,
and in the operations in sewage tanks and on sewage farms (pages
192, 195 and 220). A thimbleful of ordinary garden soil which has
received a periodical manurial dressing contains one to three million
bacteria. Certain bacteria have been found to be capable of
exercising an opposite effect, i.e. fixing the atmospheric nitrogen and
building it up into the nitrogenous tissues of plants. Thus the
nodules on the roots of leguminous plants consist of bacteria living
in symbiosis with the protoplasm of the plant and supplying it with
nitrogen in an assimilable condition. Pure cultures of these bacteria
have been put on the market as nitragin, for enriching land poor
in nitrogen. Thus a fairly complete cycle of nature is secured, and
by rotation of plants (legumes alternating with other seeds),
manures, especially nitrogenous manures, may be partially saved.
The souring of milk is caused by the bacillus lactis. This souring
is an indispensable preliminary to the making of cheese, and the
bacillus can now be used in pure culture to hasten the natural
process. The peculiar aroma of good butter is due to a bacterium
which has been isolated; and it can now be supplied in pure culture
for butter-making, thus obviating bad butter.
Certain bacteria are disease-producing or pathogenic. The
largest of these is the Bacillus of Anthrax, a disease common in sheep
and oxen, and sometimes communicated to man. This bacillus is
1·2 µ thick and 6 to 8 µ long. When an animal dies of this disease
it should be buried without cutting the skin. When exposed to the
air this bacillus forms minute spores, very difficult to destroy. They
may live for several years in pits in which animals dying from
anthrax have been buried. Butchers have died when inoculated
through cracked fingers when dressing the carcase of a cow
which has had anthrax. Similarly men handling the hides of
such animals may be inoculated, either with a form of disease
in which rapid blood-poisoning is produced, or with a malignant
carbuncle, from which recovery is possible if it be treated
promptly. Wool sorters of mohair wool are very liable to
suffer from a fatal form of pneumonia due to the dust from wool
derived from animals which have died from anthrax (page 107).
This disease gives a good instance of possible attenuation of virus,
of which another example is seen in small pox (page 293). Pasteur
grew anthrax bacilli in broth at a temperature of 110° Fahr.
At this temperature the bacilli multiplied by division, and no
spores were formed. By repeatedly sub-culturing after the bacilli275
had become old (i.e. by putting minute quantities of the growth
into fresh broth) and exposing to air, he obtained anthrax
bacilli which were only slightly virulent, only producing slight
constitutional disturbance when inoculated, i.e. injected under the
skin of sheep, and yet protected them against ordinary infection
by anthrax. Other methods of attenuation of virus have been
discovered. For instance the growth of the bacillus in the presence
of a feeble antiseptic, or passing it through the circulation of an
animal which is relatively insusceptible to the particular bacillus
has this effect.
Other important pathogenic bacteria will be considered later
(pages 298 to 398). It is only necessary here to mention that
suppuration, erysipelas, puerperal fever, and a number of forms of
blood-poisoning are due to the invasion of the system by cocci. A
single round cell (commonly not more than 1 ∕ 25000 inch in diameter)
is called a micrococcus. When in pairs as in the micro-organism
causing pneumonia they are called diplococci; when in chains,
streptococci (i.e. twisted); when in masses, staphylococci. When cocci
and other micro-organisms are kept out of wounds, healing occurs
without suppuration; this is the principle of the antiseptic and
aseptic methods of treating wounds (pages 106 and 110). The
question of immunity is discussed on page 288.
Saccharomycetes occur in fermenting substances, as in the
fermentation of saccharine solutions. The only organism belonging
to this order, which is associated with diseased conditions, is the
Sarcina Ventriculi. This is found occasionally in the vomit or even
in the urine of some persons.
The Zygophyta occur as thread-like growths, forming a
mycelium. This is composed of jointed branching tubular cells, in
which minute spores are produced. Each spore, when liberated
from its tube, is capable of producing another mycelium, and thus
the growth spreads. The spores may be carried through the
atmosphere, thus producing infection at a distance. They have an
average diameter of 6 µ = about 1 ∕ 4000 inch.
The following are the chief Zygophytous parasitic diseases:—Thrush
is associated with the growth of a minute filamentous
fungus, the oidium albicans. It is common in babies, who are
improperly fed, and in old people, or in persons exhausted by any
chronic disease. Small white patches collect on the tongue and
neighbouring parts, and these are often followed by the formation
of minute ulcers. When it occurs in children, the food must be
carefully attended to, and feeding bottles frequently scalded, etc.
Ringworm is due to the growth of a large spored or a small
spored fungus (known under the names of Microsporon Audouini;
Trichophyton megalosporon endothrix, Trichophyton megalosporon ectothrix)
which attacks the skin. It is most difficult to eradicate when it
occurs in hairy parts, as the growth penetrates to the roots of the
hairs, and continues to live here long after it has been destroyed on
the general surface of the skin. The fungus spreads on the skin in
gradually enlarging circles, forming rings with a slightly raised
margin. It is extremely contagious, being especially apt to spread276
in schools. The spores may be carried about by means of hats or
bonnets, by gloves, towels, razors, and other means. The disease
often remains undetected for some time; and many cases, especially
where the scalp is affected, remain contagious after they have been
apparently cured.
The removal of ringworm, as of all other skin parasites, is
effected by some local parasiticide. Prolonged treatment, including
the pulling out of diseased hairs, is required for ringworm of the
scalp. A special cap should be worn, when the patient mixes with
others.
Favus, or “scald-head,” is due to the growth in the skin of a
minute fungus called the Achorion Schönleinii, which invades the same
parts as those affected by ringworm, but differs in its mode of formation
of spores; yellow cupped discs from ¼; to 1 ∕ 3 inch in diameter
being produced. It is very rare in England, and almost confined
to persons (especially children) who are kept in a filthy condition.
It is a common and fatal disease in mice. The treatment is similar
to that of ringworm.
Tinea versicolor is caused by the growth in the epithelial cells
of the skin, of a fungus called the microsporon furfur, which, unlike
the two last, does not invade the hair or nails. It forms light
brown patches covered with a horny scurf, which gradually spread,
until nearly the whole trunk may be covered. It does not attack
children, and never affects uncovered parts of the body. It chiefly
occurs in those who do not take frequent baths, and who perspire
freely. It can be removed by daily washing with soap and water
and rubbing with a rough towel, followed by the application of a
weak carbolic lotion.
Animal Parasites.—Animal parasites are found on the skin or
in internal organs or in the blood or lymphatic vessels. The
following are the most common:—
The Acarus Scabiei is a minute animal not unlike a cheese mite,
which causes the disease known as scabies or the itch. It is probably
never more than 1 ∕ 77 of an inch in length. The female has
eight legs, with terminal suckers on the four front legs and hairs on
the hind legs. The male is smaller than the female, and in the
adult condition the two hindmost legs have suckers, as well as the
four anterior. It remains on the surface of the skin, while the
female burrows deeply in the substance of the epidermis. At the
bottom of the oblique burrow it deposits ten to fifteen or more eggs,
which hatch in a fortnight and then commence similar operations on
their own account. Scabies generally starts between the fingers,
whence it rapidly spreads. The disease is acquired from some
patient suffering from the disease, or by contact with his apparel.
It may become very severe when suspicion as to its parasitic
character has not been entertained. Formerly it was called “the
seven years’ itch,” from the great difficulty in curing it before its
true cause was discovered.
The irritation caused by the insect produces eczema, and this
may be thought to be the only disease present, unless careful
examination is made for the burrows of the insect.
277
To remove this parasite, first the skin is softened, the superficial
epidermis is removed, and the burrows are laid bare, by the daily
use of hot baths with soft soap, and subsequent rubbing with flesh
towels. Then some parasiticide, such as the well known sulphur
ointment, is rubbed into all the affected parts of the skin. A few
days’ perseverance in this treatment usually suffices for a cure.
The patient’s clothes and bed clothes ought also to be thoroughly
purified by boiling or by steam disinfection or by baking in an oven;
otherwise he may become re-infected.
The Larvæ of several insects have been found embedded in the
skin. In the ox, the larva or bot of the gadfly produces a troublesome
disease, a large boil being formed under the skin as the larva
grows. This larva has, on rare occasions, attacked human beings.
Rare cases are recorded where other larvæ have become developed
in men, in all upwards of twenty separate kinds of insects having
been recognized. The treatment consists in removing the parasite.
The Chigoe, commonly known as the jigger or sand-flea, is a
minute parasitic insect, found in the West Indies and northern
parts of South America. It is so small as to be scarcely visible;
but the impregnated female possesses a proboscis, by means of
which it penetrates the skin generally near the nails and there
develops a bladder the size of a pea, which sets up severe inflammation.
To get rid of the intruder, the orifice by which it entered must
be dilated with a needle, until large enough to admit of its extraction,
without rupturing the cyst.
Several species of Fleas infest the human frame. They are
propagated by means of eggs, the worms from which enclose themselves
in a tiny cocoon before assuming the adult form.
Three varieties of Lice occur on the human skin. The first
(pediculus capitis) infests the head, especially of children, and
multiplies with astonishing rapidity, the female laying altogether
about fifty eggs. The other two varieties are the body louse
(pediculus corporis) and the crab louse (pediculus pubis).
Strict attention to cleanliness is the best means of getting rid of
fleas and bugs. A wash made of carbolic acid and vinegar painted
over bed crevices is very efficient. Lice may be removed from the
head by cutting the hair short, and carefully cutting out any hairs
to which nits are attached. The nits are cemented to the shafts of
hairs. Washing the hair with methylated spirit or paraffin is also
helpful in removing them. Afterwards the use of white precipitate
ointment will prevent their re-appearance.
The Trematoda or Flukes furnish two human parasites, viz.
the liver-fluke (Distoma hepatis), and the Bilharzia hæmatobia.
The liver-fluke occasionally produces jaundice in man. In sheep it
is the cause of the disease known as the “rot.” The Bilharzia
hœmatobia is chiefly found in Egypt, and the Cape Colony. It is
about a quarter of an inch long, and infests the blood vessels, more
particularly of the kidneys; setting up severe irritation and the
discharge of blood. It is probable that the eggs of this parasite are
received in drinking water or on salads, though occasionally inoculation
may occur through the skin when bathing.
278
The family of Nematoda possesses numerous parasitic members.
The common thread worm (Oxyuris Vermicularis) is one of the most
common of these. The female is 1 ∕ 3 to ½ inch in length, and inhabits
chiefly the lower bowel. The ova, which are from 1 ∕ 490 to 1 ∕ 1100 inch
in diameter, often gain access to drinking water, or are carried by
flies, or received on salads, etc. The injection of salt and water
into the bowel, and treatment tending to improve the general health,
are the proper remedies.
The round worm (Ascaris Lumbricoides) inhabits chiefly the
small intestine; hence medicines for its removal require to be given
by the mouth. The female is from 10 to 14 inches long; the ova,
of which each female discharges on an average 160,000 daily, are
from 1 ∕ 340 to 1 ∕ 440 inch in diameter.
The whip-worm (Trichocephalus Dispar) is a smaller nematode,
which is rarely met with in this country. The Dochmius Duodenalis
is met with chiefly in Italy and Egypt. It sucks the blood in the
intestine, causing dangerous anæmia. The Strongylus Gigas is
chiefly found in the kidneys of the ox, dog, etc., and is very rare in
man. It resembles a very large round worm. In the kidney it
produces severe disorders. How it gets there is not known.
The Trichina Spiralis has been already described (page 23).
The Filaria Dracunculus (Guinea Worm) seems to gain access
into the stomach along with water, or possibly in some cases, by
perforating the skin. It burrows among the tissues, especially of
the legs, and attains a length of several feet. It causes large boils
and sores, and through these the eggs escape and pass into water.
Here the embryo which has escaped from the egg meets with a
fresh water crustacean (cyclops), enters its body, undergoes larval
growth, and is swallowed with its host by a man, in whom it
burrows and undergoes its next stage of life.
The embryos of three species of Filaria infest the blood of
man, chiefly in the tropics. One embryonic species is found in
the blood of infested patients by day, one by night, and one during
both day and night. The length of the embryos varies from 1 ∕ 75 to
1 ∕ 125 inch, and its width from 1 ∕ 3000 to 1 ∕ 3500 inch. The night embryo,
which is the most common, is produced by the Filaria Bancrofti.
This adult worm infests the lymphatic system of man, sometimes
reaching a length of three to four inches. Its embryos may
obstruct lymphatic vessels, causing obstruction of the flow of chyle
(hence originates chyluria), and elephantiasis, in which enormous
swelling of the legs and other parts ensues.
The nocturnal migration into the lymphatic vessels, and thence
into the blood of the embryo of the F. Bancrofti, is an adaptation
to the nocturnal habits of a particular mosquito (culex pipiens or
ciliaris). When the mosquito bites an infested person, his proboscis
removes some embryo filariæ, which are quickly transferred to its
stomach. Some of these escape digestion, develop within the
mosquito, and when the mosquito dies in water they bore their way
out, and are subsequently swallowed by man.
It is essential, therefore, in order to prevent this disease to boil
or efficiently filter all drinking water, and to prevent the access of279
mosquitoes to water. Persons infested with filariæ should sleep
inside mosquito nets, in order that they may not, when bitten by
mosquitoes, spread the disease.
Tape-worms are found infesting the alimentary canal of man.
Each has a double phase of existence. In the first, the characteristic
head, or scolex, along with a bladder-like body, lies embedded
in the solid tissues of an animal; in the second, the strobilus or
tape-worm, occupies the alimentary canal of another animal. The
tape-worm consists of a number of flat segments, each of which is
capable of producing a large number of eggs, from each of which
a six-hooked embryo is developed. The segments escape from the
alimentary canal, and their ova are discharged and scattered broad-cast.
These eggs are swallowed by another animal, the hooked
embryo escapes from its case, migrates into the solid tissues, and
there produces a scolex. When the host is eaten by another animal
or by man, the scolex enters the alimentary canal, loses its bladder-like
body, and developes a chain of segments. It follows from the
above that two distinct hosts are necessary to complete the cycle of
existence of these creatures, one being commonly a herbivorous,
and the other a carnivorous animal. Thus:—
| Cystic Form. |
|
Tape-worm Form. |
| Cysticercus Cellulosæ in the muscles of the pig |
becomes |
Tænia Soluim in the alimentary canal of man. |
| Cysticercus Bovis in the muscles of the ox |
„ |
Tænia Mediocanellata in the alimentary canal of man. |
| Cænurus Cerebralis of the sheep’s brain |
„ |
Tænia Cænurus in the alimentary canal of the dog. |
| Echinococcus of man, etc. |
„ |
Tænia Echinococcus in the alimentary canal of the dog. |
The cysticercus cellulosæ has been already described (page 23).
The cystic form of the dog’s tape-worm (echinococcus) is a most
dangerous parasite for man. When the egg of the dog’s tape-worm
is swallowed by man, the embryo escaping from this egg burrows
from the alimentary canal, and forms large cysts, chiefly in the
liver, but occasionally in the lungs, brain, and other organs. For
the removal of these, surgical interference is required. This form
of cyst is commonly known as a hydatid. It is most frequently
seen in Iceland and Australia, though not uncommon in this
country. Its frequency depends largely on the number of dogs,
and on the facility with which the ova of their tape-worms can
gain access to water.
The adult Tape-worms are usually derived in man from eating
meat containing the cystic form. The cysticercus of the pig produces
Tænia Solium; that of the ox, the Tænia Mediocanellata.
These are the two most common forms of tape-worm in man.
The minute head of T. Solium has four suckers and a double row of
hooklets, 28 in number; while the head of T. Mediocanellata has
four suckers but no hooklets. The segments of T. Solium are280
smaller than of T. Medioc., and the structure of the segments of the
two is somewhat different.
Preventive Measures.—In avoiding the various Entozoa
described, it is important (1) to carefully avoid all underdone meat.
The eating of smoked sausages, or of meat which is not cooked
throughout, is a common source of tape-worm and of trichinosis.
(2) All vegetables should be thoroughly washed: this is especially
important in the case of water-cress, lettuce, etc., which are eaten
raw.
(3) If the purity of the water is not ensured, it should be boiled
or filtered through a Pasteur-Chamberland filter (page 98), especially
in tropical climates, and where many dogs are kept. Dogs should
be kept out of the kitchen, lest ova accidentally gain access to
articles of food.
(4) The possibility of flies and mosquitoes acting as carriers of
parasitic disease must be remembered, and precautions taken.
281
CHAPTER XLII.
THE RÔLE OF INSECTS IN SPREADING DISEASE.
Insects are now known to be important agents, (a) as
carriers and (b) as intermediate hosts of disease-agents.
The common domestic fly (Musca domestica) is the unwelcome
companion of man in nearly every country. The eggs are usually
laid and the larvæ undergo their development in excrement, but the
female sometimes selects meal, bread, or fruit for the purpose. In
practice, however, one of the best means of diminishing the number
of domestic flies is to insist on the daily removal of all manure,
especially horse manure, and to sprinkle the manure receptacle in
the interval with lime. The fly may obviously be the means of
conveying infected material from place to place. Anthrax has been
ascribed to this cause. Nuttall has proved experimentally that flies
are able to carry the infection of plague, and that they die of the
disease. The presence of enormous numbers of flies in cholera
times has been noted. Experimentally, flies caught in cholera wards
have been found to harbour the cholera spirillum. It is probable
that they play a serious rôle in spreading the infection of cholera.
Hence all infectious dejecta (stools and urine) should be covered
until finally disposed of, and food should be protected against flies.
The same remarks apply for enteric fever. Flies fed with pure
cultures of the bacillus of enteric fever pass these bacilli in their
dejecta in a still virulent condition. In camps, especially in
connection with large armies, there is the strongest reason for
believing that flies carry infection from latrines to food. Flies have
been known to feed on the expectoration of consumptive patients
and it is possible therefore that they may thus infect food.
The bed bug (Cimex lectularius) has been stated to be capable
of conveying by its bite the infection of plague and other diseases
from an infected to a healthy person; but Nuttall’s experimental
results were entirely negative.
Fleas (pulex) probably do not play any part in spreading
anthrax. Experimentally, anthrax bacilli die off rapidly in fleas. In
India, persons who had handled rats dead of plague frequently
acquired the disease. This was explained by Simond on the
supposition that the fleas abandoned the dead rat for the human
subject. The rats which appeared to have caused plague in
man were stated to have died but a short time before; and the
handling on the day after their death of rats dead of plague was
stated to be safe because the rats’ fleas had then deserted the dead282
rat. It is assumed that the flea injects the poison of plague under
the skin. On the contrary it is to be remembered that the fleas
infesting rats and mice belong to a different family from that which
attacks man. Whether this is a usual means of conveying plague
may therefore be regarded as still doubtful. That rats convey
plague to man is certain; whether fleas act as an intermediary
remains somewhat uncertain.
The Mosquito family (Culicidæ) has been found to be an
important if not the sole means of spreading certain serious diseases
to man. To this family belong all true gnats or mosquitoes; but
the only two genera which have been proved to be able to cause
disease are Culex and Anopheles. The culex may usually be
distinguished by the fact that when alive and at rest its head is
below the level of the thorax and abdomen, thus giving the insect a
hump-backed appearance, while the body of the anopheles under
the same circumstances is all in a line.10 The anopheles is more
slender and its head smaller than that of the culex. The anopheles
usually confines its blood-sucking operations to the evening and
night. During the day it remains in dark corners. It lays its eggs
usually in a natural pool or pond on the ground, on the surface of
the water. In about two days a minute larva is hatched out. This
grows rapidly, assumes the pupa form, from which the perfect insect
emerges. The female insect alone is blood-sucking. In about 20
days after birth, it lays from 150 to 200 eggs. Its relation to malaria
may be gathered from the following historical sketch. In 1880
Laveran found in the red blood corpuscles of malarious patients
minute bodies which he regarded as not bacteria, but a very low
form of animal life, possessing amœboid movements. These grew
at the expense of the blood corpuscles, deposited a dark pigment,
and often assumed the appearance of a “rosace,” a rounded body
with little spherules at its circumference. Golgi in 1889 observed
differences between the rosaces of tertian and quartan fever, and
found that the periods of occurrences of the fever corresponded with
the times of maturation of the rosaces. It was concluded therefore,
that the rosaces caused the fever by shedding their sporules into the
blood. These sporules when thus shed were found to attach
themselves to, and grow in, other red blood corpuscles. It is now
known that there are three species of the parasite, in one of which
the parasites are crescentic in shape. The examination of a drop of
blood from a patient now enables a doctor to recognise which of
these three forms of malaria he is dealing with.
Laveran observed that certain forms of the parasite presented
“flagella,” i.e. filaments exhibiting very active movements. Manson
having observed that flagella were not found in blood first drawn,
but only appeared after a little time had elapsed, conceived the idea
that the function of these must be that of spores. Having previously283
observed that a microscopic worm, filaria, is drawn with the blood
into the stomach of a kind of mosquito (page 278), and finds in the
latter a secondary host, he concluded that a similar cycle of events
might occur in malaria. Ross tested this theory, and by causing
mosquitoes bred in bottles from the larva to bite persons affected
with the crescent form of malaria, after repeated unsuccessful
attempts, was eventually able to find in comparatively rare
mosquitoes which had thus bitten a malarious patient, small
rounded bodies embedded in the wall of the stomach. These were
watched and found to present appearances identical with those of
the parasite of malaria. Similar pigmented bodies were subsequently
found in other mosquitoes.
The malarial parasite belongs to the Protozoa, of which it is
one of the smallest members. Man is its intermediate host, and the
anopheles its definitive or final host. In the red blood corpuscle of
man it is a unicellar organism, from 1 µ to 8 µ in diameter. It has
two methods of reproduction, endogenous by spore formation and
exogenous or sexual. The former occurs in man; the latter in the
mosquito. Without the latter, the parasite being unable to pass
from man to man, would die with its host. In endogenous multiplication
spores are formed which separate from the original
parasite and gain access to other red blood corpuscles. The large
pigmented spheres and the crescent bodies require to enter the
stomach of the anopheles to attain full development. In the
anopheles the crescents become spherical, flagella are shot out,
having a length of 4 to 5 times the diameter of a red blood corpuscle.
These represent the male element, while other spheres without flagella
are the females. By the fusion of these two a fertilised cell is
produced (the travelling vermicule), which now assumes the shape of
a spear-head and is actively mobile. The travelling vermicule
pierces the stomach wall of the mosquito and develops into a zygote.
If an infected mosquito is examined on a succession of days under
the microscope, the following stages can be traced. The zygote
consists of pigmented spheres 7 to 8 µ in diameter, lying in the
muscular fibres of the mosquito. These grow, and become surrounded
by a capsule. Smaller spheres form and subdivide, bud-like
processes develop on their surfaces; these gradually become sickle-shaped
and protrude into the body cavity. They increase in size.
until they attain dimensions of from 40, to 60 µ. Eventually they
rupture, and the sickle-shaped bodies (sporozooites) escape and are
carried in the body fluid of the mosquito to its salivary glands.
These sporozooites are about 14 µ long, and human infection is
caused by them. They have been traced as far as the end of the
proboscis of the mosquito. (See also page 307).
284
The prevention of disease depends largely on a knowledge of its
causes. Disease may be due to a personal life not in accordance
with physiological laws; or to some cause or causes acting
ab extra. With advance of knowledge the number of diseases
which can be proved to be caused by a contagium vivum introduced
from without is steadily increasing. We have already discussed
the influence of habits, of clothing, exercise, sleep, and food on
health, and have shown how errors in these respects may lead to
disease. It now remains to consider more particularly the
prevention of diseases, due to the introduction into the system of
contagia.
In the study of such diseases three chief factors require
consideration: (1) the contagium itself; (2) conditions of environment,
as climate, soil, season, weather, etc., which may favour or
impede its spread; and (3) personal conditions which similarly
influence it. Of these age, heredity, fatigue, injury, diet, and race
are specially important.
The first two groups of diseases given in the Registrar-General’s
classification of causes of death are (1) Specific Febrile
or Zymotic Diseases, and (2) Parasitic Diseases. The objection
to the word “specific” is that, although in most instances diseases
in this group are “specific” in the sense that they are caused by
a particular microbe, e.g. tetanus, anthrax, tuberculosis, in a few
instances the same lesions may be caused by several microbes, e.g.
septicaemia (blood-poisoning), pneumonia. “Zymotic” was the
name given by Farr, in view of the analogy of the febrile process to
that of alcoholic fermentation. In both there is the introduction of
a living germ or germs; in both a period of “incubation” in which
nothing can be observed; then follows the active disturbance; and
in the disease, as well as in the fermenting liquid, the process is
stopped, when the microbes have multiplied to a certain extent,
a temporary or permanent protection being the result. The best
name for the diseases in this group is “Infective.” Parasitic
diseases, like ringworm, scabies, or trichinosis, are also infective;
but for convenience may be described separately as “parasitic.”
The relation between the words “infectious” and “contagious”
requires explanation. A disease like measles or small-pox, which
can be transmitted from person to person, without immediate
contact between the two, is termed infectious. In these cases the285
infection is conveyed by mucus expectorated or by dust blown
about, or carried in apparel, etc., from the first patient. Such
diseases may also, of course, be communicated by direct contact.
If direct contact between the sick and well is indispensable for
the transmission of a disease it is called contagious. There is no
such hard line in nature, although some diseases can be more easily
communicated than others. The term contagious is usually applied
to parasitic diseases like ringworm and scabies, but even these
can be communicated by means of infected articles as well as
persons. The word contagious should be abandoned for all the
acute febrile diseases. The word infective is used to include all
specific febrile diseases, however spread. This word, therefore,
includes not only infectious and contagious diseases, but also
diseases spread by inoculation, i.e. injection of the infection under
the skin. Thus malaria is not infectious from patient to patient;
but can be inoculated by the mosquito.
Infective Diseases are either acute or chronic. Of acute
infective diseases small-pox and enteric fever are typical examples;
of chronic, tuberculosis and syphilis.
It was formerly supposed that in certain diseases the contagium
or infective agent grew in external noxious matter, a miasm being
produced; while in other diseases contagion was only produced
direct from patient to patient; and others originated in either way.
Hence the classification of infective diseases into (a) miasmatic,
(b) contagious, and (c) miasmatico-contagious diseases. This
classification has now been abandoned. Thus influenza and ague
were formerly thought to be miasmatic; but the former is spread
by personal infection; the latter by inoculation of the contagium by
an infected mosquito.
Bacteriology has thrown an immense light on the causation
of infective diseases. A large number of these have been proved
to be caused by bacteria, and by analogy we infer the same thing
for many others. Koch has laid down the following postulates
as necessary before it can be stated that a particular disease is
directly caused by a given microbe:—
(1) The microbe shall be demonstrated in the diseased tissues or blood of
man or an animal suffering or dead from the disease.
(2) The microbes shall be isolated from these and cultivated in suitable
media until obtained in pure culture. That is to say, matter containing the
microbe, taken from the infected source, must be cultivated in artificial media
outside the animal body, under conditions excluding the possibility of the
introduction of other microbes, until pure cultures of these microbes are obtained,
and these microbes must be transplanted from generation to generation, until
it is certain that no trace of non-living matter derived from the original animal
body remains in the culture.
(3) A pure culture of the microbe, thus obtained, shall, when introduced
into the body of a healthy susceptible animal, reproduce the disease in question.
(4) The microbe in question shall be found in the animal so affected.
Kanthack adds a further condition, that
(5) The toxins and poisonous substances obtained from the artificial cultivations
shall agree chemically and physiologically with those obtained from the
diseased animal.
286
All the preceding conditions have been fulfilled for anthrax,
diphtheria, and tetanus; and the first four conditions have been
fulfilled in regard to tuberculosis, glanders, gonorrhœa, malignant
œdema, and actinomycosis. In enteric fever and influenza the first
two conditions have been met; but inoculation experiments (3) have
failed. In leprosy and relapsing fever the first condition is met,
but (2) has failed.
In the following diseases the specific microbe has not been
isolated, though from analogy it is believed that each of them is
caused by such a microbe:—
- Measles.
- Rubella (German measles).
- Typhus fever.
- Scarlet fever.
- Varicella (chicken pox).
- Variola (small-pox).
- Whooping Cough.
- Mumps.
- Hydrophobia, etc.
Erysipelas occupies a special position. It is a specific disease
due to a microbe, which, when it attacks other parts than the skin,
may produce abscesses, boils, or blood-poisoning.
Bacteria are either saprophytes, i.e. they can grow on dead
organic or even inorganic matter; or parasites, i.e. they are
dependent for their existence on a living plant or animal which they
invade. There are two varieties of each of these, obligate and
facultative. An obligate parasite can develop only within a
living host; while a facultative parasite can, according to
circumstances, lead either a parasitic or saprophytic form of
existence. The fact that certain contagia are completely, and
others only partially, parasitic brings out important differences in
their life-history. Thus, so far as we know, the contagia of scarlet
fever, measles, small-pox and hydrophobia do not multiply outside
the body. Hence there is a reasonable prospect of annihilating
them by measures of disinfection and isolation. The position of
diphtheria is doubtful. It may have a saprophytic phase of life.
The contagium of tuberculosis, as well as of erysipelas, may have a
life outside the host, though to what extent is doubtful. Cholera and
enteric fever, although generally communicated by infection, appear
sometimes to be communicated by contagia grown in saprophytic
life, remote from preceding cases.
The infection caused by bacteria may be local or general. Thus
in tetanus and in diphtheria the invading bacteria usually remain
at their original point of invasion (under the skin in tetanus, in the
throat in diphtheria). In anthrax always, and often in enteric
fever, they are present in the general circulation. In both instances
the symptoms of disease are due chiefly to the toxic products or
toxins formed by the bacteria. These toxins are enzymes,
ptomaines, tox-albumins, etc. The specific toxins of anthrax,
diphtheria, and tetanus have been identified; and by this means
the possibility of neutralising them is created.
The channels of infection, i.e. of invasion of contagia, are the
skin and the mucous membranes, particularly of the digestive and
respiratory tracts.
287
The Incubation Period of an infectious disease is the interval
elapsing between the receipt of infection and the earliest development
of symptoms. The period of incubation of the chief infectious
diseases is shown in the following table:—
| DISEASE. |
BEGINS USUALLY
ON THE |
BUT MAY POSSIBLY BE AT
ANY PERIOD BETWEEN |
| Scarlet fever |
4th day. |
1 and 7 days. |
| Diphtheria |
2nd day. |
2 and 5 days. |
| Small-pox |
12th day. |
1 and 14 days. |
| Chicken pox |
14th day. |
10 and 18 days. |
| Typhus fever |
12th day. |
1 and 21 days. |
| Enteric fever |
14th-21st day. |
1 and 28 days. |
| Cholera |
1st-3rd day. |
A few hours and 10 days. |
| Measles |
12th-14th day. |
10 and 14 days. |
| Rötheln (German measles) |
14th day. |
12 and 18 days. |
| Mumps |
19th day. |
16 and 24 days. |
| Whooping cough |
14th day. |
7 and 14 days. |
| Influenza |
2nd day. |
2 and 6 days. |
The period of incubation is several weeks in hydrophobia and
syphilis, and may be several years in leprosy.
Following the period of incubation, come the premonitory
symptoms, which usually are somewhat sudden in onset. For the
chief symptoms of onset see page 318.
Persons vary in susceptibility to attack by different infective
diseases. The intensity of an attack depends on the condition of
the patient, and on the number and the virulence of the particular
microbes infecting the patient. In certain families attacks of
particular diseases are more severe, and attacks are more liable to
occur than in others.
It has been shown in certain diseases that the cells and the
fluids of the body have a protective effect against infection. This
protective action varies in different persons, and in the same person
at different times. The cells of the body (phagocytes) swallow up
and destroy a certain number of bacteria. This action is called
phagocytosis. It is overcome when the dose of contagium is
excessive, or when the vitality of the individual is lowered,
especially the local vitality at the part attacked. Thus children
with “weak throats” are particularly prone to scarlet fever and
diphtheria.
The protection afforded by one attack of an infective disease
against its recurrence varies greatly. A second attack of small-pox
is very rare, of scarlet fever less uncommon, of diphtheria common.
In erysipelas, influenza, pneumonia, and rheumatic fever, second
or even more numerous attacks are common.
Immunity against an infective disease may be natural, but is
more often acquired by an attack of the disease in question. This
latter immunity is active, and is due to the formation in the tissues of
the immunised person or animal of substances produced by the
reaction of these tissues to the stimulus of the contagium. Thus a
pig when it has recovered from an attack of swine-plague has288
produced what are called in German antikörpers, and its tissues are
now a medium unfavourable to the growth of the bacillus of swine-plague.
If the serum of the protected pig is injected under the skin
of another pig, the latter acquires passive immunity against swine-plague,
which is not so persistent as active immunity.
Active Immunity can be produced (1) by an attack of an
infective disease, or (2) by artificial inoculation (under the skin) of
the contagium of the disease, producing a milder attack of the
disease. This may be done (a) by inoculating small doses of a virulent
contagium, as in the inoculation of small-pox from a previous patient;
or (b) by inoculating an attenuated virus, as in vaccination. Inoculation
of small-pox virus usually produced a milder attack than
infection by ordinary means; but patients thus inoculated were a
great source of danger to other persons. In vaccination the virus
of small-pox is employed, which has become attenuated by passing
through the calf. In its passage, it has lost the power of producing
anything beyond a vesicle at the point of inoculation. The principle
of protecting by attenuated virus was extended by Pasteur, who
was able to render animals resistant against anthrax, swine-fever,
and quarter-evil, and hens against fowl-cholera, by inoculating them
with attenuated cultures of the contagia of these diseases. Haffkine
has applied the same method on a large scale for cholera.
The above are methods of bacterial vaccination. Salmon and
Smith have shown that artificial active immunity can be produced
also by (3) toxin-injection. They artificially cultivated the hog-cholera
bacillus in broth. This broth was then sterilized, the bacilli
being killed, but their products remaining. By injecting pigeons
with this sterilized broth they made them resistant to subsequent
infection by the bacillus itself, thus proving that immunity can be
produced by chemical as well as by biological means. The immunity
was proportional to the dose of the toxin absorbed. By
gradually increasing the dose, it was found practicable to confer
immunity, not only against doses of toxin that would otherwise have
been fatal, but also against bacterial infection by the particular
bacillus used in manufacturing the toxin.
Passive Immunity.—Behring and Kitasato found that if the
toxin (free from the bacilli) of tetanus be injected into an animal in
increasing doses until it becomes immune against infection by the
bacilli of tetanus, the blood serum of the animal in question injected
into white mice confers the same immunity on them. The protection
thus conferred is only temporary. Exactly the same procedure has
been adopted for diphtheria, and it is now found that by injecting
anti-diphtheritic serum into children who are exposed to the infection
of diphtheria, they can for several weeks be prevented from
developing the disease. This is of great practical importance, as
meanwhile the source of infection can have been removed. Furthermore,
the protective serum is also curative, and by its means diphtheria,
if early treated, can be reduced from a dangerous to an insignificant
disease.
Various theories have been propounded to explain immunity.
Pasteur supposed that the special pabulum or food of the bacillus of289
the given disease became exhausted; but this does not fit in with
the immunity that can be produced by toxins and anti-toxins.
Chauveau supposed that certain bacterial products are retained in
the body, rendering it unsuitable for further growth of the particular
bacillus; just as more than 14 per cent. of alcohol in a saccharine
solution prevents further fermentation. This does not explain all
the facts. Metschnikoff concluded that the fight of the leucocytes
and phagocytes of the body against weaker bacilli, gave the power
of fighting and overcoming a more virulent bacilli, and that these
properties of the cells were transmitted to later generations of body
cells. This theory fails to explain the acquired immunity against
toxins as well as against bacteria which occurs. The discovery
that the blood and other normal tissue fluids possess some power of
destroying bacilli has relegated the phagocytal theory to a secondary
position.
Natural Immunity varies in different animals. Thus enteric
fever, scarlet fever, and measles are not known to occur except in
man. Tuberculosis, anthrax, hydrophobia (called rabies in the dog),
glanders and tetanus are common to man and certain other animals.
Man, cattle and pigs frequently suffer from tuberculosis; goats,
sheep, horses, and dogs are practically immune to it.
Epidemic and Endemic Diseases.—Infective diseases may
occur sporadically, in epidemics, or in pandemics, i.e. epidemics spread
over a number of countries. The word epidemic is used here to
mean specially prevalent, and not to apply only to infective diseases.
Thus there may be an epidemic of arsenical poisoning from
contaminated beer.
Certain infective diseases are endemic or topical, i.e. they have
special homes or centres, from which they occasionally spread as
epidemics. Yellow fever, cholera, and malaria belong to this group.
In a minor degree enteric fever, epidemic diarrhœa, and tuberculosis
may be described as endemic.
Each infective disease has a special seasonal incidence. Of
these the most important are the autumnal group, viz.
| Epidemic Diarrhœa, |
maximum |
prevalence |
in |
July and August. |
| Enteric Fever |
„ |
„ |
„ |
November, but excessive, Aug. to Dec. |
| Erysipelas |
„ |
„ |
„ |
Nov. to Dec. |
| Diphtheria |
„ |
„ |
„ |
Nov. and Dec., excessive, Sept. to Dec. |
| Scarlet Fever |
„ |
„ |
„ |
Oct., excessive in Aug. to Dec. |
| Of other infective diseases |
| Small-pox has its |
„ |
„ |
„ |
May, but is excessive Jan. to June. |
| Whooping Cough |
„ |
„ |
„ |
Dec. to May. |
| Measles commonly has two seasonal maxima, in June and December with intervening minima. |
290
Causes of Epidemics.—Measles recurs in the large towns of
England every alternate year. Other infective diseases occur at less
regular intervals. The recurrence of epidemics is not solely due to
personal infection and the accumulation of a population at susceptible
ages. There are longer cycles of the causes of which but little is
known. Thus scarlet fever has been shown by Longstaff and
Gresswell to become epidemic chiefly in dry years; and I have
shown that diphtheria and rheumatic fever become widely epidemic
under the same conditions, diphtheria becoming so only when a
series of dry years occur in immediate succession.
291
Acute Infectious Diseases are characterised by certain definite
characters.
1.—They are usually infectious or contagious. It is preferable to
use these two terms as interchangeable. The modes in which
infection is received vary greatly with different fevers.
(1) Some can only be propagated by inoculation—the introduction
through an abraded surface of a minute quantity of the poison;
as in glanders and hydrophobia. Others, again, may be introduced
in this way, but are usually acquired in another manner, as scarlet
fever, small-pox.
(2) Some are carried through the atmosphere. The contagium
of small-pox can be carried as far as any, while that of typhus fever
only traverses a few feet. The atmosphere acts as a conveyer
of infection, and the infectious matter must necessarily, in most
instances, be in the condition of dust to enable it to be wafted by
currents of air or disturbed by the movements of persons in an
infected room.
(3) Clothes, books, and furniture are not uncommonly carriers
of infection. An old letter, or a lock of hair, has even after many
years’ concealment in an enclosed space produced infection on being
brought to light. Woollen articles convey infection more easily
than calico, and dark clothes better than light coloured. A fever
nurse’s clothes should never be woollen, but some washable
material.
(4) Drinking water and food often form a vehicle for infection.
Milk and water are the two usual sources of infection; but uncooked
food, especially oysters and mussels, fed in sewage-polluted
estuaries, may produce the same effect. Cholera, enteric fever,
dysentery, and summer diarrhœa are the chief diseases from this
source; but scarlet fever and diphtheria occasionally have a similar
origin. Milk may be infected from having been handed by an
infectious patient; or it may possibly convey infection of the
disease from which the cow at the time is suffering, e.g., tuberculosis
(see also page 312). Water may be contaminated with sewage or
the excreta of a single infectious patient.
2. They retain their specific character and origin. Small-pox never
produces scarlet fever, nor vice versâ; and it is found universally
that all the specific fevers “breed true,” each one retaining its
identity. More than this, a previous case of the same fever can292
nearly always be detected on careful examination. Overcrowding
and other insanitary conditions diminish the resistance to infection,
and may increase its virulence.
3. The behaviour of contagia, when received into the system, is
characteristic of these diseases. There is first of all a period of
latency or incubation, during which no symptoms are manifested
(see page 287.) The incubation period is followed by the characteristic
symptoms of the particular fever, which disappear in a variable
period, leaving the patient, as a rule, more or less insusceptible to a
second attack (see page 288).
Throughout the progress of the disease, except in the period of
incubation, the patient is able to communicate his disease to persons
about him who have not been rendered safe by a previous attack.
The way in which he thus communicates his disease varies in
different cases. In scarlet fever, the throat and skin are the chief
sources of contagion; in influenza, whooping-cough, and measles,
the secretions from the respiratory passages; in hydrophobia, the
saliva; in enteric fever and cholera, the vomit and stools.
Prevention of the Spread of the Chief Acute Infectious
Diseases.—We may divide these into three classes. (1) Those
which are infectious by contact with the patient or by the atmosphere
around him. (2) Those in which the intestinal and renal
evacuations are almost alone infectious; as enteric fever and cholera.
(3) Those in which inoculation through an abraded surface is
generally if not always necessary to produce infection.
SMALL-POX OR VARIOLA.
The contagium of small-pox is very tenacious of life. All
parts of the body, and all secretions and excretions contain it. As
in typhus it adheres to every article in the room, but unlike typhus
is possessed of great vitality, and if not exposed to the air may be
active after many years. There is considerable evidence indicating
that the contagion of small-pox may occasionally be conveyed aerially
for a considerable distance, for even a quarter or half a mile from
hospitals in which small-pox patients are isolated. Whether this is
the aerial convection of infection, or in part at least due to carelessness
of persons connected with the hospital in their movements to
and fro, may remain an open question; but such hospitals in the
midst of towns are in practice a mistake; and in London small-pox
has been found to be more manageable since its small-pox patients
were all conveyed to extra-urban hospitals. The means for the prevention
of small-pox are (1) Isolation of infectious patients. (2)
Disinfection of all infected articles. For particulars under these
two heads, see pages 319 and 324. They must be carried out most
rigidly in this disease. (3) Vaccination and re-vaccination.
Inoculation of small-pox virus was largely practised as a
means of ensuring a comparatively mild attack, until it was made
illegal in 1840. Sometimes, however, the attack thus produced was
fatal, and every case of inoculated small-pox became a new focus of293
infection, and a source of high mortality, especially among young
children.
Vaccination. About the year 1795 Dr. Edward Jenner was
informed by a milk-maid that she could not take small-pox, as she
had already contracted the natural cow-pox during milking. Many
had previously heard this same statement made; but Jenner was
the first to put the matter to the test. He took the lymph or virus
from a woman who had accidentally acquired cow-pox (vaccinia)
from a cow, and inoculated a boy with it. Some months later he
inoculated the same boy with small-pox, and a second time five
years afterwards, without producing small-pox on either occasion.
Many other experiments were made, all confirming these results;
and in 1798 Jenner published his results.
The practice of vaccination gradually became more general,
and was followed by a progressive decrease in the mortality from
small-pox.
Cow-pox or vaccinia is small-pox modified and mitigated by its
passage through the system of the cow, and not a spontaneous
disease of the cow. By its passage through the cow it has become
attenuated and altered. Instead of a general eruption all over the
body, there are vesicles only at the point of inoculation; and
vaccinia, unlike small-pox, is not communicable from person to
person except by inoculation. Furthermore it is in the vast
majority of instances an extremely mild ailment, not involving more
than a few days discomfort.
Objection is taken to vaccination for small-pox on the ground
that serious diseases such as syphilis, erysipelas, and tuberculosis
may be inoculated at the same time. With lymph obtained from
healthy children this is impossible. Most of the cases of infection
described have been in reality hereditary disease, the local irritation
of vaccination serving to call into activity the morbid tendencies of
the child. The risk of such infection is infinitesimal; it may be
reduced to zero by moderate care and attention to detail. With
modern antiseptic methods, it is very rare for a vaccination sore to
“go wrong.” Erysipelas may be inoculated from dirt getting into a
vaccination sore, as it may be into any other sore; but with cleanliness
this need not occur; and in fact very seldom does occur. The
risks are so small as to be negligible; and if the protection afforded
is one tithe of what is claimed for it, no parent is justified in withholding
this protection from his infant. The law as to vaccination
requires that every infant shall be vaccinated within six months of
its birth, domiciliary visits for this purpose being made by the
public vaccinator. The obligation can only be avoided by a statement
on oath before a magistrate by the parent of conscientious
objection to vaccination.
Does Vaccination protect against Small-Pox? The
registration of deaths for the whole country only began in 1837, and
before this period death-rates from small-pox in terms of the
population cannot be accurately stated. Since that time there has
been less or more vaccination, so that it is difficult to obtain a true
comparison between periods with and without vaccination. Some294
indication of the facts in London prior to 1801, when the first
English census was taken, may be obtained from the fact that in
1796 (two years before the date of Jenner’s “Inquiry,”) small-pox
reached its highest point, causing 18½ deaths out of every 100 total
deaths from all causes. In the præ-vaccination period small-pox
was 9 times as fatal as measles, and 7½ times as fatal as whooping-cough
(McVail), while since vaccination has been practised it has
sunk to an insignificant position, when compared with these diseases.
Dr. Guy found that in London there were in 48 years of the
seventeenth century ten epidemics, in the whole of the eighteenth
century 19 epidemics, and in the nineteenth century no epidemic
during which the deaths from small-pox caused one-tenth or more
than one-tenth of the total deaths from all causes in any year. The
worst year under obligatory vaccination in London was 1871, in
which barely 4½ per cent. of the total deaths was due to small-pox,
a proportion which was exceeded in the eighteenth century ninety-three
times.
In Sweden, the highest death-rate before vaccination (1774-1800) was
7·23 per 1,000 inhabitants, the lowest 0·31; under permissive vaccination
(1801-1815) the highest 2·57 per 1,000 inhabitants, the lowest
0·12; under compulsory vaccination (1816-85) the highest 0·94 per 1,000
inhabitants, the lowest 0·0005. It has been stated that these results,
which might be extended by quotations from the statistics of other
countries, have been obtained not by vaccination, but by improved
sanitation, including in this term not only improved housing and
better water and food supply but also increased means of isolating
the infectious sick. Improved housing may by diminishing overcrowding
aid in diminishing the spread of this disease. Whether in view
of the immense increase in the proportion of the population
which lives in towns, it can be said that this has occurred is
doubtful. Hospital isolation undoubtedly prevents the spread of
infection when promptly effected. But a large share of the improvement
in small-pox mortality occurred before either hospital or
home-isolation of small-pox patients was generally enforced. There
is no reason for supposing that impure water or food, or nuisances
about houses have any connection with the origin or spread of
small-pox, any more than they have with the origin or spread of
measles or whooping-cough; which still remain as prevalent as in
the past. Further light can be thrown on the subject by an
examination of the age-incidence of small-pox, and of its attack-rate
and severity in vaccinated and unvaccinated respectively.
The age incidence of deaths from small-pox has, since
1847, when returns classified according to age became available,
undergone a remarkable alteration. Prior to 1870 the small-pox
deaths in infants nearly always formed 20 per cent. or more of the
total mortality from this disease, between 1870 and 1890 they did
not greatly exceed 10 per cent. of the total, while since 1890 they
have again begun to form an increasing proportion of the total
small-pox mortality. At ages 1-5 the change is even more remarkable.
Before 1870 deaths at these ages nearly always exceeded 30
per cent. of the total; since 1870 they have varied between 5 and 14295
per cent. of the total; and since 1890 they have, like the proportion
of deaths under one, again increased. At the higher ages the
proportion of deaths has correspondingly increased, so that the
curves of age incidence have become curiously inverted.
The lowered birth-rate can only account for a small portion of
this transference of the chief mortality due to small-pox from
childhood to adult life.
Furthermore it must not be supposed that the only change
which has occurred is that the deaths which formerly occurred in
childhood now occur in adult life. The death-rate at all ages has
greatly declined. The only explanation which in my judgment
satisfactorily explains this remarkable change in age-incidence of
small-pox mortality is the fact that vaccination protects children
from small-pox and that the protection diminishes, though it never
entirely disappears, with advancing years. This conclusion is
confirmed by the evidence obtained as to the proportion of vaccinated
and unvaccinated attacked, and as to the severity of the attacks
occurring when a community is invaded by small-pox.
Attack-rate among Vaccinated.—If the protective effect of
vaccination, like that of a preceding attack of small-pox, wears off,
it will not be expected that no attacks of small-pox will occur
among the vaccinated. For evidence of immunity from attacks we
must examine the records as to revaccinated persons exposed to
infection. During the six years 1890-95, out of a staff in the
London small-pox hospitals varying from 64 to 320, the percentage
attacked by small-pox was nil, except in 1892 when it was 1·4, and
in 1893 when it was 1·9.
Taking the experience of towns in which during recent years
epidemics of small-pox have occurred, the following attack-rates
have occurred. By attack-rate is meant the percentage number of
attacks occurring among persons living in infected houses. By
fatality is meant the number dying out of 100 attacked.
|
ATTACK RATE UNDER 10 YEARS OF AGE. |
ATTACK RATE OVER 10 YEARS OF AGE. |
| VACCINATED. |
UNVACCINATED. |
VACCINATED. |
UNVACCINATED. |
| Dewsbury |
10·2 |
50·8 |
27·7 |
53·4 |
| Leicester |
2·5 |
35·3 |
22·2 |
47·6 |
| Gloucester |
8·8 |
46·3 |
32·2 |
50·0 |
Severity (Fatality) among Vaccinated.—The experience of
the same three towns comes out as follows:—
|
FATALITY RATE UNDER 10 YEARS OF AGE. |
FATALITY RATE OVER 10 YEARS OF AGE. |
| VACCINATED. |
UNVACCINATED. |
VACCINATED. |
UNVACCINATED. |
| Dewsbury |
2·2 |
32·1 |
2·6 |
18·7 |
| Leicester |
0·0 |
14·0 |
1·0 |
7·8 |
| Gloucester |
3·8 |
41·0 |
10·0 |
39·7 |
296
In view of such results as the above it is not surprising that
the Royal Commission, in their majority report, summed up the
advantages of vaccination as follows:
“(1) That it diminishes the liability to be attacked by the disease.
“(2) That it modifies the character of the disease, and renders it (a) less
fatal, and (b) of a milder or less severe type.
“(3) That the protection it affords against attacks of the disease is greatest
during the years immediately succeeding the operation of vaccination. It is
impossible to fix with precision the length of this period of highest protection.
Though not in all cases the same, if a period is to be fixed, it might, we think,
fairly be said to cover in general a period of nine or ten years.
“(4) That after the lapse of the period of highest protective potency, the
efficacy of vaccination to protect against attack rapidly diminishes, but that
it is still considerable in the next quinquennium, and possibly never altogether
ceases.
“(5) That its power to modify the character of the disease is also greatest
in the period in which its power to protect from attack is greatest, but that its
power thus to modify the disease does not diminish as rapidly as its protective
influence against attacks, and its efficacy during the later periods of life to modify
the disease is still very considerable.
“(6) That re-vaccination restores the protection which lapse of time has
diminished, but the evidence shows that this protection again diminishes, and
that, to ensure the highest degree of protection which vaccination can give, the
operation should be at intervals repeated.
“(7) That the beneficial effects of vaccination are most experienced by
those in whose case it has been most thorough. We think it may fairly be
concluded that where the vaccine matter is inserted in three or four places, it is
more effectual than when introduced into one or two places only—and that if
the vaccination marks are of an area of half a square inch, they indicate a better
state of protection than if their area be at all considerably below this.”
SCARLET FEVER.
Scarlet Fever and Scarlatina are the same disease. It is
extremely infectious, the contagium retaining its virulence for
protracted periods. It occurs in epidemics at irregular intervals.
During recent years the type of scarlet fever has become greatly
attenuated, and this constitutes one of the difficulties of prevention,
as the mild form of the disease is apt to be overlooked. The
fatality per 100 persons attacked varies greatly with age. It is
highest in children under four, rapidly declining with increasing
age. Hence the importance of protecting children from attack in
early life. Two results follow from the wise precautions taken to
prevent attack early in life. (a) With each successive year of life
the liability to attack, when exposed to infection, diminishes; (b)
the danger of the attack if it occurs and its liability to be fatal
becomes rapidly less with greater age. The most common mode of
infection is by contact with a previous patient. Outbreaks due to
milk infected by a scarlatinal patient also occur. Infected cream
has also been known to convey infection. In a milk outbreak the
patients would be found chiefly among the customers of a special
dairyman, the cases occur almost simultaneously, except secondary
cases which may be infected from the first. The simultaneous
occurrence of two or more attacks in one house, especially if the
same thing happens in a number of houses should throw suspicion297
on the milk supply. It has been suggested that scarlet fever may
originate apart from human infection, from a special disease of the
cow, but the evidence on this point is inconclusive.
The duration of infection is usually reckoned until the desquamation
of the skin is complete, i.e. about six or seven weeks from the
onset of the attack. Occasionally it is more protracted even though
desquamation is complete, infection appearing to persist in discharges
from the nose and ear and in sore places inside the nostril and
possibly in other parts. The period of greatest infectivity is in the
earlier part of the disease, when the throat is inflamed. The
common notion that the disease is most infectious during the later
period, that of desquamation, is erroneous. The micro-organism
causing scarlet fever has not certainly been identified. The
measures of prevention are those common to infectious diseases
(page 317).
MEASLES.
Measles is an extremely infectious disease, before as well as
after the rash appears on the fourth day of the disease. The
infectivity of the catarrhal stage constitutes one of the main
difficulties in preventing its spread, as measles may be unrecognisable
at this stage. The common notion that measles and
whooping-cough are comparatively harmless infantile complaints
will be dissipated by a study of the comparative death-rate for the
five years 1891-5 per million persons living in England and
Wales:—
England and Wales.—Death Rates per Million of Population.
| Small-pox |
20 |
| Measles |
408 |
| Scarlet fever |
183 |
| Typhus fever |
4 |
| Enteric fever |
174 |
| Whooping-cough |
398 |
| Diphtheria |
253 |
| Diarrhœa |
630 |
It is a mistake also to suppose that measles and whooping-cough
are only serious when neglected. Such neglect greatly
increases the likelihood of death from bronchitis or pneumonia; but
the diseases themselves, especially measles, are frequently fatal
during the acute early stage. More children are attacked with
measles under the age of five than at any other age, and the
greatest number between two and four years of age. The greatest
fatality is in the second year of life, when it may be 24 per cent. of
those attacked, as compared with between two and three per cent.
in the fourth year of life, and a trifling amount at higher ages.
These facts explain the folly of allowing children to have an
infectious complaint when another child in the house is attacked,
“to have it over at one trouble.” Such action is pregnant with
evil results. (1st) Severe cases occur, in which a fatal result
ensues; and even where death does not occur, the child may be left
weakly and very prone to become tuberculous. (2nd) Every
additional case forms a new centre of infection. It is like the old
practice of inoculation for small-pox; the individual is protected,
but becomes a source of danger to all around him. If there is only298
one case of measles in a family the risk to neighbouring households
is much smaller than where several children are infected. (3rd)
Every year that a child’s attack can be delayed, increases his
chance of recovery if he is subsequently attacked, and diminishes
the likelihood of his being attacked.
The duration of infection should be reckoned as at least three
weeks. The contagium of measles does not appear to hang about
rooms with the persistence of that of scarlet fever, and less stringent
disinfection is required.
WHOOPING COUGH.
Very few people have reached adult life without having suffered
from this disease, as well as measles. This is chiefly due to the
carelessness in mixing infected with healthy children. One frequently
hears the peculiar and characteristic cough of a child with whooping-cough,
in public assemblies, in railway trains, or in the out-patient
rooms of hospitals. The contagium of whooping-cough is conveyed
chiefly by the expectoration, which becoming dry, may be scattered
like that of phthisis, as dust. Clothing conveys infection easily;
visits to infected children should, therefore, be prohibited to all
who have to mix with susceptible children.
The duration of infection should be reckoned as at least six
weeks from the first recognisable symptoms. It may be longer
than this.
DIPHTHERIA.
This disease has become increasingly prevalent in the last ten
years after a period of only slight prevalence for about twenty-five
years. I have shown that epidemics of diphtheria occur during a
succession of years of protracted drought. Diphtheria is more
common in girls than boys, possibly owing to their more affectionate
habits; and occurs chiefly under ten years of age, the fifth year of
life being that of greatest prevalence. Unlike the acute infections
hitherto considered, the bacillus causing diphtheria has been
identified and cultivated in the laboratory (called the Klebs-Loeffler
bacillus or diphtheria bacillus). Direct infection from patient to
patient is probably more common than indirect infection by clothes,
etc., though the latter occurs. The infection may hang persistently
about a house and its belongings, in the absence of complete purification.
When diphtheria is prevalent slighter sore throats occur,
sometimes before true diphtheria is detected. This led to the theory
that under conditions of overcrowding, especially in schools, there
occurred in the micro-organisms causing these sore-throats “the
progressive development of the property of infectiveness.” Possibly
these were slight non-typical attacks of diphtheria. Such attacks
occur also during epidemics of diphtheria, and unless specimens
(“throat swabs”) from these sore-throats are examined bacteriologically,
are likely to spread diphtheria by attendance at school,
etc. Aggregation in schools seems to intensify the contagium of
diphtheria. The practices of kissing, of transferring sweetmeats299
from mouth to mouth, of cleaning slates with saliva, are common
means of spreading it. Effluvia from foul drains and sewers have
been commonly held to cause diphtheria. If they aid in producing
it, it is rather by lowering the vitality and causing ordinary sore
throat. Sore throats and catarrhs make the subjects of them much
more prone to diphtheria (see also page 117). Damp houses have
been stated to favour the development of diphtheria. Probably
they do so in the same way as effluvia from drains. It is likely
that the diphtheria bacillus has a saprophytic stage of existence in
the soil, as indicated by its excessive prevalence in dry warm
years. Besides direct infection from patient to patient and indirect
infection by fomites (i.e. in clothing, etc.), milk occasionally causes
epidemics of diphtheria. The infection has been usually caused by
the handling of the milk by an infectious person. In certain
outbreaks no human contamination of the milk could be discovered;
and it has been surmised that an analogous disease in the cow may
cause diphtheria in man. This is still a moot point. Fowls, cats,
and other animals are the occasional victims of diphtheria, and may
convey it to man.
The duration of infection in diphtheria is usually less than six
weeks; but it may be much more protracted. In some instances
long after all naked-eye evidences of diphtheria has disappeared,
bacteriological examination may still show the presence of the
diphtheria bacillus for two or three months; in rare cases even
longer. The protection afforded by one attack of diphtheria against
a second is slight and only temporary. The means of prevention are
isolation and disinfection as for other infectious diseases. Two
additional means are available (a) bacteriological diagnosis; (b)
prophylactic injection of antitoxic serum. Many sore throats
without membrane on the throat are due to the diphtheria bacillus.
Even if membrane be present there may be doubt as to whether the
case is true diphtheria. Hence the importance of bacteriological
examination.
The patient’s throat is swabbed with cotton-wool which has been rolled
around a metal probe and sterilised. The wool is then smeared over sterilised
and solified blood serum in a test tube. It is then incubated over night at a
temperature of 37° C. Next morning the minute growth that has occurred on
the surface of the blood serum is spread on a microscopic cover-glass, appropriately
stained, and examined microscopically. If diphtheria bacilli are present,
they can be recognised by their form and arrangement. The same means
enable us to ascertain when a patient has recovered, whether he is fit to be
released from isolation.
Antitoxic serum has been found to be a valuable prophylactic
and curative agent.
The serum is obtained as follows: Sterilised broth is inoculated with
virulent diphtheria bacilli, and grown at 37° C. for a week or more. The broth
is then filtered through a Pasteur filter. The filtrate contains toxine free from
bacilli. Some of this toxine is injected under the skin of a horse. A few days
later the dose is repeated, gradually increasing amounts being injected until
injection of further quantities of the toxine is found experimentally not to increase
the antitoxic value of the horse’s blood serum. Next the horse is bled. Its
serum is found to have acquired the power of protecting a guinea-pig against300
doses of the toxine of diphtheria which would otherwise be fatal. Ten times the
quantity of the horse’s serum which will protect a guinea-pig (of 250 grammes
weight) against ten times the minimum fatal dose of the toxine is called an
antitoxic unit.
The treatment of diphtheria in man by the antitoxic serum thus
obtained has proved to be remarkably successful. Furthermore, if
a susceptible person who has been exposed to the infection of
diphtheria, is injected with a small dose of antitoxic serum, he
becomes temporarily immune, and does not fall a victim to
diphtheria. This is a most important point especially for young
children, who may already be incubating a disease which but for this
prophylactic injection might occur and prove fatal.
TYPHUS FEVER.
This disease was formerly known as spotted or jail-fever, and
for many ages has been the scourge of prisons and armies, and all
collections of people living in overcrowded and insanitary districts.
The history of typhus is the history of human misery. It is
essentially associated with filth, overcrowding, and destitution; but
when once established by these conditions, it can be carried by
infection to others who live amidst healthy surroundings. It
generally occurs in winter, when overcrowding is greatest. With
free ventilation, the disease cannot be carried more than a few feet.
It can be transmitted by clothing. The micro-organism causing it
has not been discovered. With the clearance of the rookeries of our
great towns, it is rapidly decreasing, and appears likely to become
extinct. The means of prevention, in addition to the abatement of
nuisances, including overcrowding, are isolation and disinfection
(pages 319 and 325).
RELAPSING FEVER.
This disease was formerly common in this country, but except
in some parts of Ireland has entirely died out. It is caused by a
micro-organism (Spirillum Obermeieri) which can be detected in the
blood. Inoculation of this will produce the disease in man or in
monkeys.
Epidemics of relapsing fever commonly follow in the track of
typhus fever; overcrowding and filth being especially associated
with typhus, and starvation with relapsing fever, hence its name of
“famine fever.”
ENTERIC OR TYPHOID FEVER.
Enteric fever causes its highest death-rate in early adult life,
though it is not peculiar to any age. Eberth in 1880 discovered the
Bacillus typhosus in the spleen and other organs of enteric fever
patients. This is commonly known as Eberth’s bacillus, and is the
cause of enteric fever. A few years later it was isolated and can
now be grown on agar or gelatine in laboratories. It closely
resembles other bacilli which are normal inhabitants of the human301
intestine; but can be distinguished by certain tests. It is a small rod,
rounded at its ends, and from 2 to 4 µ long and three times as long
as broad. In the living state it is freely motile, and possesses a
number of minute cilia or flagella. Apart from other means of
distinction between it and other bacilli, Grüber’s serum reaction
enables its identity to be ascertained. The bacillus suspected to be
the Bac.-typhosus is cultivated in broth in the bacteriological
laboratory. A small quantity of blood is taken from the finger of a
patient known to be suffering from enteric fever. The serum is
separated from the blood corpuscles of this blood by a centrifugalising
machine. A drop of the blood serum is diluted with 100 drops of
broth culture of the suspected bacillus. If the latter is not the
Bac.-typhosus, the individual bacilli when a drop of the mixture is
examined under the microscope, will continue to move about freely;
if it is the Bac.-typhosus, the bacilli will adhere together in “clumps”
and become immobile. Conversely a valuable means of ascertaining
whether a suspected case is really suffering from enteric fever is
secured, as this blood added to 30 times the amount of a pure
culture of the Bac.-typhosus in broth will cause the latter to “clump”
within half-an-hour (Widal reaction). Higher dilutions are usually
unnecessary.
The chief means of spread of enteric fever is by the urine
and fæces; and nurses who have to empty bed-pans unless
very careful to wash their hands afterwards, using the nail-brush,
are very liable to become infected, probably when eating food
afterwards. The urine in a considerable proportion of cases,
contains the typhoid bacilli, and it is therefore most important that
care should be exercised in the cleansing of all chamber utensils,
and that the urine as well as the fæces should be rigidly disinfected
(see page 331). The infectivity of enteric fever has been underrated
in the past. When patients with this disease are nursed at home by
relatives who do not appreciate the full importance of the necessary
precautions, it is rather the rule than the exception for them to fall
victims to its infection. Probably, sometimes the infection has been
scattered as dust, owing to small particles of fæces or of urine
having become dried on bed linen. The most absolute cleanliness
is essential in nursing this disease. In hot climates there is reason
to believe that infective dust may be blown about from privies.
Insanitary local circumstances are an important means of spreading
enteric fever. It is more prevalent where there are privies than
where there are pail-closets; and more prevalent where there are
pail-closets, than where water-closets are the rule. Defective drains
or soil-pipes are frequently found in houses in which enteric fever
originates, and there can be little doubt that the former are at least
partially responsible for the latter. The exact link is doubtful.
Probably infective dust is blown or aspirated into the room and is
inhaled.
The most common cause of enteric fever is infected food or water.
Of foods milk not infrequently has been the means of spread of
enteric fever. Large epidemics have been traced to this source.
Usually this has arisen by washing the milk cans with or wilfully302
adding contaminated water to the milk. Water, whether added to
milk or taken independently, must have contained the specific
contagium (the Bac.-typhosus) of enteric fever, to enable it to cause
enteric fever. Hence water from a contaminated stream is more
likely to have produced this effect than well-water, unless a patient
has had enteric fever in the house to which the well is attached, and
his dejecta have contaminated the water. Surface waters or spring
waters may be contaminated with sewage (as at Maidstone) or
deep well waters through fissures (as at Worthing) and thus
widespread epidemics be produced. After floods, rivers and wells
are most likely to contain the specific contagium of enteric fever, as
at such times surface impurities from middens, etc., are apt to be
washed into the water. (See also pages 91 and 224).
The means of prevention of enteric fever are the discovery and
removal of the cause of an outbreak, and the isolation of each
patient and disinfection of all discharges. An early means of
diagnosis is secured by Widal’s reaction. This is especially useful
in cases not presenting characteristic clinical symptoms. The
recognition of a disease or at least the suspicion of its presence is
an indispensable first step for the taking of precautionary measures.
CHOLERA.
Cholera, which was formerly so prevalent, now seldom occurs
in this country, and at each successive visit to England its inroads
have become less serious. At its last visit in 1893 it scarcely
obtained a footing in the country. Thus in the epidemic of 1854 in
England it caused 1080, in that of 1866 it caused 672, and in that of
1893 only 45 deaths per million of population. In this country at
least it is chiefly spread by infected water and foods, especially by
infected water; and the preceding figures form an excellent testimony
to our improvement in this respect. For particulars of the
Hamburg outbreak, see page 93. In its mode of prevalence and
propagation it is very similar to enteric fever, being infectious by
means of the evacuations. The means of prevention are the same as
for enteric fever. Cholera was shown by Koch to be caused by
what is known as the comma bacillus or spirillum of Asiatic cholera,
so called because of its curved shape. It is from 1·5µ to 2·6µ long
and ·5µ broad. For the supposed connection of enteric fever and
cholera with movements of the ground-water, see page 225.
SUMMER OR EPIDEMIC DIARRHŒA
is a most fatal disease among infants in the third quarter of each
year. It is chiefly a disease of urban life, and occurs to a preponderant
extent among the children of the artisan and still more of the
unskilled labouring classes. It is much less abundant in towns
which have adopted the water-carriage system of sewage than in
those retaining the conservancy methods of removal of excrement
(page 198). Towns with the most perfect domestic and street303
scavenging arrangements have the least epidemic diarrhœa. An
impervious soil favours a low diarrhœal mortality; while persons
living on porous soils usually have much diarrhœa. I have shewn
elsewhere that given two towns equally placed so far as social and
sanitary conditions are concerned, their relative diarrhœal mortality
is proportional to the height of the temperature and the deficiency
of rainfall of each town, particularly the temperature and rainfall of
the third quarter of each year. In other words there is a general
inverse relationship between rainfall and diarrhœa and a direct
relationship between temperature and diarrhœa. Thus wet and cool
summers are adverse to diarrhœa. Ballard concluded that the
summer rise of diarrhœal mortality does not commence until the
mean temperature recorded by the 4-foot earth thermometer has
attained somewhere about 56° F., no matter what may have been
the temperature previously attained by the atmosphere. This is a
convenient index, as the summer warmth does not immediately
cause diarrhœa. All the above facts point to the conclusion that
the fundamental condition favouring epidemic diarrhœa is an
unclean soil, the particulate poison from which infects the air, and
is swallowed most commonly with food, especially milk. Thus,
diarrhœa, like enteric fever, is a “filth-disease.” As the contagium
appears to gain entrance by food, the following card of precautions
which is distributed each year in the poorer districts of Brighton
may be reproduced here:
HOW TO PREVENT DIARRHŒA.
During the summer a large number of infants die from diarrhœa. Scarcely
a single baby who was being suckled dies from this cause. It is evident,
therefore, that in the prevention of this very fatal summer disease, precautions
as to food are most important.
Attention to the following points would save many infants’ lives:—
1. Do not wean your infant during the hot months of July, August and
September. To begin artificial feeding during hot weather is very dangerous.
2. If feeding by hand is absolutely necessary, carefully follow these
directions:
(a) All milk should be boiled before being given to the infant.
(b) The infants’ food must be prepared fresh each time. (For particulars see
below.) Milk and water, and still more “pap” or patent foods, if
left two or three hours, “go bad,” and are then very highly dangerous
to the infant.
(c) All jugs or other utensils used for storing milk must be scalded out and kept
absolutely clean. They should be covered to prevent access of dust.
(d) The feeding bottle must be thoroughly scalded after each meal, and the tube
thoroughly cleansed. It is best to use alternately two boat-shaped
bottles without tubes. If the bottle smells sour, something is not clean,
and the infant will suffer.
3. Decomposing refuse, such as decaying vegetables, bones, fish-heads, &c., is
a fertile source of Diarrhœa. It should be burnt, and not placed in the dust-bin.
4. Scrupulous cleanliness of the house, especially of the rooms where food is
stored, is most important. Dust in every form is dangerous to health, and for
removing it wet cleansing is preferable to dry. Thus washing and scrubbing are
safer means of cleansing floors, &c., than sweeping.
5. Report to the Sanitary Office, Town Hall, any smells or choked closet or
drain.
304
DIRECTIONS FOR PREPARATIONS OF INFANTS’ FOOD.
For a Child
aged |
Mix and then boil |
For each Meal |
| Under 6 weeks |
 |
1 part fresh milk |
 |
4 tablespoonsful. |
| 2 parts water |
| 1 teaspoonful cream |
|
Mix and then boil |
| 6 weeks old |
 |
1 part fresh milk |
 |
8 tablespoonsful. |
| 1 part water |
| 2 teaspoonsful cream |
|
Mix and then boil |
From 3 to six
months old |
 |
2 parts fresh milk |
 |
8 tablespoonsful. |
| 1 part water |
| 2 or 3 teaspoonsful cream. |
The infant should be fed at regular intervals only, at first every two hours, the
interval being gradually increased.
The infant should be fed slowly.
If the milk as prepared above disagrees, freshly boiled barley water should be
used instead of water.
The addition of cream is necessary because cows’ milk is poorer in cream than
mothers’ milk, and because it is very often made poorer still by mixing with
separated milk before sale. Deficiency of cream causes rickets. A little sugar may
also be added to the milk, but this must not be regarded as a substitute for the
cream.
TETANUS.
Tetanus or lockjaw is not infectious, but is conveyed to man by
the inoculation of a wound by dirt or earth which contains the
tetanus bacillus. For this reason it is more apt to follow injuries
to the hands or feet. Extreme cleanliness of wounds is the only
practicable preventive means. Little is known of the history of the
tetanus bacillus outside the body; and as to what soils contain it
most abundantly. Wounds contaminated by horse manure appear
to be especially dangerous.
GLANDERS.
Glanders is common in horses. It attacks the mucous
membrane of the nose, causing ulceration. It is extremely
infectious. Farcy is a more chronic form of the same disease, in
which the so-called “farcy-buds” are produced. Its prevention can
best be ensured by killing both actually diseased and suspected
animals, if the latter give a reaction to mallein. Mallein is a
product allied to tuberculin, obtained from cultivations of the
bacillus of glanders. It sets up febrile reaction in glandered, but not
in healthy horses. Further preventive measures are the temporary
closing of public drinking fountains for horses, and the thorough
cleansing and disinfection of stables. Men, especially grooms, are
sometimes infected by the horse, and the disease is commonly
fatal.
305
HYDROPHOBIA.
Hydrophobia is the disease in man which is caused by the bite
of a dog or other animal suffering from rabies. It is seldom if ever
communicated otherwise than by inoculation. The incubation period
in the dog varies from three to six weeks, and in man is usually
about the same; but occasionally it is much longer, occasionally
even more than a year.
At the Pasteur Institute, Paris, patients who have been bitten
by rabid dogs are treated by the inoculation of an attenuated virus
of rabies derived from rabbits, with promising results.
Dogs only acquire rabies from dogs or other animals already
rabid. So far as is known, it does not arise de novo. Hence the
necessity for an extensive area of muzzling when cases of rabies
occur. The enforcement of this plan has greatly reduced the
amount of hydrophobia in this country in recent years. There has
been much misplaced sympathy with dogs on this score. In the
dog the symptoms of rabies occur in three stages: a premonitory
stage, in which the dog’s habits change, he becomes morose and
quiet, and dribbles; a second stage, in which he has paroxysms of
fury, his voice is high-toned and croupy, and he cannot swallow
water; and a third or paralytic stage, in which his jaws drop, he
drags his hind legs and soon dies.
ERYSIPELAS.
Erysipelas occurs on various parts of the skin. It is caused by
the inoculation through an abraded surface of a virulent form of
the same streptococcus that commonly causes suppuration. It
occurs chiefly in debilitated subjects. Some persons are specially
prone to it, and may have many attacks. Erysipelas, like scarlet
fever, occurs most in years in which there is deficient rainfall; and
is probably conveyed by dust. It may spread, though exceptionally,
from case to case.
YELLOW FEVER.
Yellow fever never occurs in England, except when imported
from the West Indies or other countries in which it is endemic. It
clings to seaport towns in hot countries; and as a permanent
disease is only found when the mean winter temperature is at least
68°-72° F. A frost always stops an epidemic of this disease. The
germs of this disease are communicated by mosquitoes, which act
as an intermediate host. Dr. J. W. Lazear, although isolated from
yellow fever cases, died of it seven days after submitting to the
puncture of an infected mosquito, thus proving the communicability
of this disease, and entitling himself to an honoured position among
scientific martyrs.
PLAGUE.
Plague is an Eastern disease, which occasionally shows a
tendency to become widely epidemic. It is due to a rod-shaped
bacterium, averaging ·8 µ to 1·6 µ in length, which does not form306
spores. In its characteristic form patients suffering from this
disease have inflammatary swellings (buboes) of various glands: hence
the name Bubonic Plague. In other cases it simulates ordinary
pneumonia, typhus or septicaemia; or the patient may be so
slightly ill as to be able to walk about. It appears probable that
the bacillus enters the body through cracks or other lesions of the
skin, possibly also by inhaling infective dust. The rat is an
important factor in the spread of plague. Very commonly plague
has been widely prevalent and fatal among them before it attacks
human beings. Rats also bring it in ships from infected ports.
Hence one of the most important preventive measures is to kill all
rats on board ship, before the cargo is unloaded. This has been
done by sulphurous acid fumigation in the holds. The use of
carbonic oxide gas will probably be found practicable for the same
purpose. Manson has put the importance of this point tersely as
follows: “To prevent cholera the tea-kettle, malaria the mosquito
net, and plague the rat-trap.” Flies may carry the infection (page
281). It has been suggested that the fleas of rats carry the infection
(page 281).
ANTHRAX.
Anthrax is a very fatal disease in cattle and sheep, occasionally
in pigs. Butchers may inoculate themselves with it when dressing
a diseased carcase; the tanners similarly when handling the hide;
and woolsorters may inhale it when sorting wool derived from a
diseased animal (page 107). To prevent the latter, suspected wool
must be disinfected by steam, and special arrangements made for
carrying off the dust produced during sorting.
PUERPERAL FEVER
occurs after childbirth. It is caused like erysipelas by the
inoculation of septic material. This may be conveyed by dirty
instruments (syringes, etc.), or by dirty hands. Hence the
importance of extreme cleanliness of hands, finger-nails, and all
articles used during and after childbirth.
RHEUMATIC FEVER
has been commonly attributed to a damp condition of the
atmosphere and soil. I have elsewhere shown that this is a
mistake, probably arising from the fact that these conditions
produce what are called “rheumatic” pains, though they have no
true relationship with acute rheumatism (rheumatic fever). I have
shown that rheumatic fever occurs chiefly in very dry years, the
excess of prevalence in such years being sufficient to justify the use
of the term “epidemic.” There is strong reason to believe that
rheumatic fever is an infective disease, derived, not from other
patients suffering from the same disease, but from some outside
micro-organism which is ordinarily saprophytic. It follows the
rule that when the lesion produced by an infective disease is deep307seated
(in the joints in this instance), no infection can be communicated
to other persons. Some families are much more prone to
rheumatic fever than others.
INFLUENZA
is, like diphtheria, a somewhat mysterious infectious disease. Like the
latter it almost disappeared for a series of years, and then again
became epidemic in 1889. The previous epidemics of influenza in the
19th century had occurred in 1803, 1833, 1837-8, and 1847-8. The
causes of this recurrence of influenza are unknown. It is spread
from person to person by direct infection, the infection being
conveyed by the mucous discharge from the nose, throat, and lungs.
Pocket-handkerchiefs probably are largely responsible for conveying
the infection as dust. The disease is particularly fatal to the old;
and these should not expose themselves to possible sources of
infection, as in public places of assembly, during an epidemic.
Every patient attacked with the disease should remain indoors for
at least ten days. This is in his own interest, as he thus minimises
the risk of such dangerous complications as pneumonia; and it is
his duty in the interest of the rest of the community. Many lives
might have been saved, had not influenzal patients “struggled
about” during the early stages of the disease.
MALARIA.
Malaria, or Ague, is a generic name given to a disease caused
by the invasion of the body by the plasmodium malariæ, discovered
by Laveran in 1880. It occurs in two chief types, remittent fever
and intermittent fever. For many generations it has been regarded
as due to a marshy condition of the soil, associated with decaying
vegetable matter, and a moderately high temperature. It is now
clear that these conditions are necessary, only because they are
necessary for the life of the mosquito. The well-known danger
of being out of doors at night in a malarious country is explained by
the nocturnal habits of the mosquito. The higher salubrity of the
upper stories of houses is explained by the fact that the mosquito
does not rise high from the ground; and of high-lying localities
by their greater dryness. The value of the mosquito net, of smoke,
and of fire as protections from malaria are due to their keeping
mosquitos at a distance. The mosquito clings to the puddle or
swamp where she was born, and where she will deposit her eggs.
Hence the special danger of the immediate vicinity of such
collections of water. Thus the prevention of malaria resolves
itself chiefly into means for preventing the development of certain
species of Anopheles (page 282). The conditions necessary for the
multiplication of these are (1), an atmospheric temperature from
75° to 104° F.; (2) collections of fresh or slightly brackish water;
and (3) the presence in these of low forms of animal and vegetable
life. We have already described the cycle of life of the plasmodium
malariæ (page 282). Man is the chief, if not the only source,308
from which the mosquito derives this parasite. In native communities
the young children, even when apparently not ill with malaria, nearly
always harbour these parasites in their blood corpuscles. Hence
the importance of Europeans having their dwellings as remote as
possible from native houses. Mosquitos do not travel far.
Instances of the prevalence of malaria in the absence of
mosquitos are not substantiated. The outbreaks of malaria
where the soil has been disturbed after long lying uncultivated,
probably mean the formation of puddles favourable to the breeding
of the larvæ of mosquitos.
The necessary preventive measures are classified by Manson as:
- 1. Suppression of mosquitos.
- 2. Prevention of infection of mosquitos.
- 3. Prevention of infection by mosquitos.
The suppression of mosquitos involves the draining or
filling in of swamps and ponds, the cleansing and canalisation of
sluggish streams, and the afforestation of hills to prevent floods.
Cultivation of rice and other plants entailing the prolonged flooding
of land should be restricted to fields remote from dwellings. Subsoil
drainage is helpful. The “painting” of stagnant waters with
petroleum, which should be renewed every week or two, frees
water for a considerable time from the larvæ of mosquitos.
Eucalyptus and other balsamic trees may help to dry up pools, &c.
The prevention of infection of mosquitos is secured by
insisting on all malarial patients using mosquito nets. This
prevents the access of mosquitos. At the same time patients
should vigorously and persistently take quinine, which kills the
malarial parasites in the blood, and thus diminishes and finally
removes the danger to other persons produced by the intermediation
of the mosquito.
The prevention of mosquito bites is secured by rendering
the house mosquito-proof by filling in all openings by fine wire
gauze, and by having mosquito curtains to all beds; also by
fumigating the rooms occasionally with the dried flowers of the
chrysanthemum, by strict cleanliness of rooms, and by flushing
them with sunlight. The proof of the mosquito theory as to the
causation of malaria has been recently supplied by two test
experiments. (a) In the first, a number of mosquitos which had been
fed on the blood of malarious patients were sent to London from
Rome. These were allowed to bite Dr. Manson’s son, who had
never previously had malaria. A few days later he had a
characteristic attack of fever. Malarial parasites were found in
his blood. He recovered in a week’s time after free dosage with
quinine, and the parasites disappeared from his blood. He suffered
from a slight relapse about a year later. (b) On a fever-haunted spot
in the Roman Campagna a wooden hut was built, and Drs. Sambon
and Low, and three others took up their abode here during the
malarious season, the only precautions taken being the use of
mosquito nets and wire screens in doors and windows. They
went about the country daily, but were always home before sunset.
They all remained at the end of the season free from malaria.
309
Consumption and other Tubercular Diseases.
Consumption (also called phthisis or phthisis pulmonalis) in the year
1899 caused a recorded death-rate of 1,336, and tubercular diseases
of other parts of the body a death-rate of 575 per million of the
population. In the same year the chief infectious diseases, including
small-pox, measles, scarlet fever, whooping cough, typhus, and
enteric fever, and diphtheria, were together responsible for a
death-rate of 1,248 per million. In the five years, 1861-65, the
mean death-rate from consumption was 2,527 per million, so that
a reduction of nearly 50 per cent. has apparently occurred.
Notwithstanding this great decline, consumption and other
consumptive diseases, which may together be classed under the
name tuberculosis, still cause more deaths than all the acute
infectious diseases put together. Its importance is emphasised
by the fact that between the ages of 20 and 45, one-third of all the
deaths of males, and between one-third and one-fourth of all the
deaths of females occurring at these ages are due to consumption
of the lungs.
Formerly great stress was laid on the hereditary character
of consumption. It would appear, however, that what is inherited
is simply an increased vulnerability of tissues. Judging by the
analogy of other animals it may be said that infants are rarely, if
ever, born tuberculous. Bang examined 6,000 head of cattle with
the tuberculin test (see page 311), and found that in calves under
6 months old only 10·7 per cent. reacted, i.e. showed evidence of
tuberculosis, between 6 and 12 months old 18·7 per cent., 1 to 2
years 23·2 per cent., and over this age 31·3 per cent. reacted; from
which it may be inferred that the infection is nearly always received
after birth.
The real cause of tuberculosis was shewn by Koch, in 1881, to
be the tubercle bacillus. This is a minute bacillus, the length of
which is from a quarter to half the size of a blood corpuscle.
These bacilli, obtained from tuberculous growths in the body,
Koch was able to cultivate on glycerine agar at blood-heat outside
the body. By sub-culturing he obtained pure cultures, and after
growing the bacilli for as long as fifty-four days, he inoculated
various animals, producing tuberculosis in every case, while in310
similar check experiments in which all the conditions were the
same, barring the absence of bacilli, no tuberculosis resulted.
The tubercle bacilli are easily distinguished from most other
bacilli by the fact that after being stained by aniline dyes, such as
carbol-fuchsin, the colouration is not removed when the preparation
is soaked in dilute acid. By this means the presence of tubercle
bacilli in the sputum (expectoration) of a phthisical patient is
easily discovered, and a valuable means of early recognition of the
disease secured. This is most important, as in its early stages
consumption is an easily curable disease. Tubercle bacilli are
discharged from the lungs in consumption of the lungs, from
the bowels in consumption of the bowels, and so on. Hence
the essential necessity for disinfecting these discharges. Such
discharges while in a moist condition have, unless they are actually
swallowed, little or no capacity for evil. It is when they become
dry that they become dangerous. Thus the expectoration of a
consumptive patient spat on to the floor or deposited in a pocket
handkerchief is, so long as it remains moist, perfectly innocuous.
What is evaporated from the wet surface is simply steam, harmless
as the steam escaping from the domestic tea-kettle. But when it
becomes dry, then comes danger. Dust is formed, which contains
the living tubercle bacilli, and with the mere shaking of the
handkerchief or the disturbance of the dust on the floor these are
inhaled, and often cause consumption. The person thus infected
may be a new patient. Often also it is the consumptive patient
who is thus re-infected. Consumptive patients tend to recover.
But if the patient’s disease is daily recruited by fresh doses of the
tubercle bacilli inhaled with the dust of previous expectoration,
fresh centres of disease are produced, and thus the patient is
unwittingly helping to cause his own death.
The infectious character of tuberculosis has been long
suspected. In the 18th century, in Naples, there were enactments
insisting on the isolation of consumptive patients and disinfection
of their furniture, books, etc. We now know, however, that these
were counsels of panic, and that for practical purposes the infection
may be regarded as confined to the sputum. The expired breath is
free from infection except during coughing. That the sputum is
infectious can be easily proved by feeding guinea-pigs or the
domestic fowl on it. These rapidly become affected by generalised
tuberculosis. The simple character of the precautions against
infection which are required may be gathered from the following
copy of a card which is given to consumptive patients in
Brighton:—
Precautions for Consumptive Persons.
Consumption is, to a limited extent, an infectious disease. It is spread
chiefly by inhaling the expectoration (spit) of patients which has been allowed
to become dry and float about the room as dust.
Do not spit except into receptacles, the contents of which are to be destroyed before
they become dry. If this simple precaution is taken, there is practically no danger of
infection. The breath of consumptive persons is free from infection.
311
The following detailed rules will be found useful, both to the consumptive
and to his friends:—
1.—Expectoration indoors should be received into small paper bags and
afterwards burnt.
2.—Expectoration out of doors should be received into a suitable bottle, to
be afterwards washed out with boiling water; or into a small paper handkerchief,
which is afterwards burnt.
3.—If ordinary handkerchiefs are ever used for expectoration, they should
be put into boiling water before they have time to become dry; or into a
solution of a disinfectant, as directed by the doctor.
4.—Wet cleansing of rooms, particularly of bedrooms occupied by sick
persons, should be substituted for “dusting” and sweeping.
5.—Sunlight and fresh air are the greatest enemies of infection. Every
patient should sleep with his bedroom window open top and bottom, a screen
being arranged, if necessary, to prevent direct draught; and, if possible, occupy
a separate bedroom. The patient need not fear going out of doors in any
weather, if warmly clad.
N.B.—The patient himself is the greatest gainer by the above precautions,
as his recovery is retarded and frequently prevented by renewed infection derived
from his own expectoration.
6.—Persons in good health have little reason to fear the infection of consumption.
Over-fatigue, intemperance, bad air, dusty occupations, and dirty
rooms favour consumption.
The most common source of infection is undoubtedly the dried
expectoration. Infection may, however, probably be derived from
infected food, as milk or meat.
The danger from meat is much less than that from milk,
because the former is more generally cooked than the latter, and
because the diseased portions of the former would be at least partially
removed before it was sold. The conditions under which the
meat derived from tuberculous cattle should be destroyed are given
on page 24. The abolition of private slaughter-houses, the general
establishment of public abattoirs, and efficient meat inspection
would do much towards aiding in eliminating tuberculous cattle
from herds; because it would no longer be found remunerative to
keep tuberculous cows until they become seriously diseased.
The danger from infected milk is probably very great. This
has been repeatedly proved experimentally for bovine tuberculosis
by experimenting on calves and pigs. A cow may suffer from
tuberculosis of its udder, and yet go on freely secreting milk. The
milk from such an udder readily produces tuberculosis in calves or
pigs drinking it; but if another animal be fed during the same
period with boiled milk obtained from the same udder, it remains
well.
The presence of tuberculosis in cattle can be determined with
almost complete certainty by the tuberculin test. A glycerine
extract of pure cultivations of tubercle bacilli (filtered so as
to be free from bacilli) was found by Koch to contain substances
which, when injected into guinea-pigs suffering from
tuberculosis, produced a febrile reaction, and appeared likely
to cure the disease. So far as man and larger animals are312
concerned the hope of cure by this means has not been realised;
but as a means of diagnosis, i.e. detection of tuberculosis, injection
of a small quantity of tuberculin under the skin, has been
found most valuable. If the cow thus injected is suffering
from the slightest tuberculosis, it becomes feverish for a few
days; if it is healthy no “febrile reaction” occurs. By using
this test tuberculous cattle can be detected, they can then be kept
in separate sheds, the former sheds cleansed and disinfected; the
milk of these cattle kept separate from that of the rest of the herd,
and boiled before being drunk, or the infected cattle sent to the
butcher. If the disease is strictly localised the carcases can be
utilised for food, after careful destruction of all diseased portions.
If these means were generally adopted, tuberculosis might gradually
be eliminated from the cattle of the entire country, and a serious
source of loss to farmers, as well as of danger to children drinking
the infected milk, removed. The presence of tubercle bacilli in
cow’s milk is detected by microscopic examination and by injection
of small quantities of the suspected milk into guinea-pigs. The
proportion of infected samples found when examinations of milk
supplies have been made in different towns has varied from 10 to
over 50 per cent.
It is probable that tuberculosis is conveyed by cow’s milk only
when the tuberculous disease affects the udder. But inasmuch as
the udder of a tuberculous animal may become tuberculous very
rapidly and without being detected for a considerable time, it is
evident that no tuberculous animal of any kind should be allowed to remain
in any cowshed where milch cows are kept.
Recently (July, 1901) Koch has thrown doubt on the identity
of bovine and human tuberculosis, which was previously accepted,
because (unlike some other observers) he has been unable to
produce in 19 cattle, on which he experimented, tuberculosis by
mixing with their food expectoration of consumptive persons, or
inoculating under their skins similar material. Even if these
negative results should subsequently be confirmed, the converse
proposition does not follow, that bovine tuberculosis cannot be
communicated to man; and apart from this possibility milk
containing the bacilli of bovine tuberculosis cannot be regarded
as wholesome.
The boiling of milk destroys tubercle bacilli. So does a
temperature considerably below 212° F. In Denmark, where
butter and cheese are manufactured on a large scale, and the raw
milk is collected in central dairies, a law was passed in 1898,
obliging every proprietor of a dairy to heat all skimmed milk, or
butter milk, to a temperature of 85° C. (185° F.) before returning
it to the farms. Pasteurization, i.e. the heating of milk in a special
apparatus to a temperature of 70° C. (158° F.), and keeping it at
this temperature for 30 minutes kills tubercle bacilli. If it is
rapidly cooled, the nutritive value and taste of the milk are not
spoilt. It is safer, however, to go beyond this point, and the use
of an apparatus like the Aylmer or Sentinel Sterilizer can be
recommended. More recent experiments have made it doubtful313
whether tubercle bacilli in milk are always killed in milk at a
temperature of 70° C., the pellicle formed on milk when it is
heated appearing to shield the bacilli from the effect of the heat.
Hence it is desirable that “no sterilizer should be looked upon
as thoroughly efficient for the purpose in which a temperature of
at least 85° C. (185° F.) is not attained.”
The following test may be used to determine whether milk has
been efficiently pasteurized:—
Natural milk contains a ferment or enzyme, which is destroyed at a
temperature of 176° F. This enzyme splits up hydrogen peroxide (H₂O₂) into
water and oxygen, but this effect is not produced in milk heated above 176° F.
Take one drop of a dilute aqueous solution of hydrogen peroxide, add it to one
teaspoonful of the milk. Next add two drops of a watery solution of paraphenyldiamine.
A dark indigo colour is produced with uncooked milk, no change of
colour if the milk has been pasteurized. The same test can be used for
determining whether butter has been made with pasteurized milk.
Infection is not the sole determining cause of tuberculosis.
Certain conditions of environment may determine whether the
infection will succeed in “taking root” or not. Of these the
following are important:
The nutrition of the individual if defective favours infection.
Probably one chief reason why consumption has declined nearly
50 per cent. in the last 50 years is the better, more varied and
more abundant food of the population.
Improved housing of the population has greatly helped in the same
direction. Tubercular diseases increase with density of population,
and are most prevalent in overcrowded tenements. Probably
overcrowding chiefly acts by favouring direct infection, but it must
also lower the health and power of resistance of the individual
against infection.
The drying of the subsoil has been regarded as a chief cause of
the reduction of consumption (page 226). It is probable, however,
that the wet soil merely predisposed to consumption, because it
was commonly associated with cold and wet houses, which would
favour catarrhs, and open the way for the infection of consumption.
The dryness of the house is a most important matter. If a damp
soil means also a damp house it must favour consumption and
other chest affections. Damp air, like water, rapidly abstracts heat
from the body. Compare, for instance, the discomfort of sitting
clad in water at a temperature of 65° F. with the comfort of sitting
clad in a dry room at the same temperature! The domestic fowl
is naturally immune to anthrax; but by being kept for a few hours
with its feet in cold water, it can be rendered susceptible to inoculation
with this disease.
The effects of breathing foul air are clearly shewn by the varying
death-rate from phthisis in different occupations. Thus, if the
comparative mortality figures of agriculturists be represented as
100, that of a commercial clerk = 176, of a draper = 200, of a tailor = 211,
of a printer = 244, of a bookbinder = 246.
The effects of breathing dust-laden air are even more marked.
Thus, if the comparative mortality figures from phthisis of314
agriculturists = 100, that of a coal-miner = 166, of a mason = 215,
of a chimney-sweep = 249, of a file-maker = 373, of a cutler = 407,
of a potter = 453. The last figure probably also shows the influence
of alcoholism, which greatly favours tuberculosis.
Means for Preventing Tuberculosis.—The means for
preventing tuberculosis from infected milk and meat have been
already indicated. They comprise—
(a) Means of eradicating tuberculosis from cattle;
(b) Means of preventing harm from tubercle bacilli in milk
or meat;
Under the first head improved conditions of housing of cattle,
greater air-space, improved ventilation, a larger proportion of
out-door life are important. The use of the tuberculin test, the
separation of healthy from diseased cattle; the disinfection of sheds
occupied by infected cattle are also essential.
Under the second head come efficient sterilization of suspected
food (see p. 13), and the rejection of diseased meat. (See p. 24).
The most important measure against tuberculosis is the
prevention of infection from patients with consumption. Under
this head are comprised the following steps:—
A. Means of ascertaining the existence of the disease—
- 1. Bacteriological diagnosis.
- 2. Notification of cases, voluntary or compulsory.
B. Direct preventive measures—
- 1. Law against expectoration in places of public resort.
- 2. Disinfection and cleanliness.
- 3. Isolation.
- 4. General sanitary improvement.
C. Education of the public and of patients as to the importance of
the preceding measures.
The gratuitous examination of suspected sputum is now being
undertaken in certain towns. The earlier the infectious condition
of expectoration is detected, the sooner can the necessary precautions
be taken.
The notification to the medical officer of health of all cases of
consumption I have repeatedly advocated. This is already carried
out for the chief acute infectious diseases, and although the
difficulties of acting on the information received in regard to a
chronic disease like consumption are considerable, they can be
overcome with tact and discretion. Voluntary notification is
already practised in Brighton, Manchester, and a few other towns.
Notification gives increased and more exact opportunities of
preventing phthisis by—1. Enabling disinfection and cleansing
of affected rooms to be effected; 2. Enabling instructions to be
given to the patient and his relatives as to the exact precautions
required; and by 3. Facilitating the removal of the insanitary
conditions of home and work which may have caused the case
or favoured its untoward progress.
315
The following scheme of measures of disinfection was prepared
by Drs. Niven and Newman and myself for the National Association
for the Prevention of Consumption, and is issued by them in
pamphlet form:—
In preventing a consumptive person from spreading the disease, two sets of
preventive measures are required:—1st, The removal or destruction of the
infective matter already disseminated by the patient’s discharges, especially
by his phlegm; and, 2nd, the prevention of future dissemination. For the
latter purpose the main object is not to permit any discharge to become dry
before being destroyed. Before the consumptive person has learned the personal
precautions which must be taken, and up to the time when he has been trained
to carry them out carefully, he has probably distributed a considerable amount
of infective matter. This is especially liable to accumulate in a dangerous
form at home, where the space is small, and light and ventilation are defective.
Infective particles will be found in greater abundance on and near the floors,
on ledges, and in room-hangings. But the personal clothing and bedclothes
will also have become infected. Hence it is necessary to disinfect the floor,
walls, and ceiling of the rooms occupied by the patient, as well as the furniture,
carpet, bedclothes, &c.
When this has been done, if the personal precautions advised are carried
out by the consumptive, further disinfection should not be needed.
It is, however, difficult to make sure that personal precautions are fully
carried out, and rooms should therefore be subsequently cleaned at least once
in six months, the floors being scrubbed with soft soap, the furniture washed,
the walls cleaned down with dough, and the ceiling whitewashed.
Confined workshops in which a consumptive has worked for some time
should be cleansed, and a notice in reference to spitting should be suspended
in all workshops. The latter precaution should also be observed in all public-houses
and common lodging houses, both of which require special attention
to cleansing.
Disinfection of rooms which have been occupied by consumptive patients
may be secured in various ways, but the following are the practical rules which
must underlie any methods adopted:—
1. Gaseous Disinfection of Rooms, or “Fumigation,” as it is termed, by
whatever method it is practised, is inefficient in such cases.
2. In order to remove and destroy the dried infective discharges, the
Disinfectant must be applied directly to the infected surfaces of the
room.
3. The Disinfectant may be applied by washing, brushing, or spraying.
4. Amongst other chemical solutions used for this purpose a solution of
Chloride of Lime (1 to 2 per cent.) has proved satisfactory and
efficient.
5. In view of the well-established fact that it is the dust from dried
discharges which is chiefly infective, emphasis must be laid upon
the importance of thorough and wet cleansing of infected rooms.
6. Bedding, carpets, curtains, wearing apparel, and all similar articles
belonging to or used by the patient, which cannot be thoroughly
washed, should be disinfected in an efficient steam disinfector.
7. After all necessary measures of Disinfection have been carried out,
the essential principle governing the subsequent control of a case
of consumption is that all discharges, of whatever kind (especially
expectoration from the lungs), should under no circumstances be
allowed to become dry.
Besides measures of disinfection and cleanliness, the patient
must be placed under the best conditions for overcoming the
disease. The same measures tend to prevent infection. Thus
abundant food, an open-air life, sleeping with bedroom windows
widely open, avoidance of dust, abundance of sunshine, are all316
important. The importance of sunlight in the prevention of consumption
can scarcely be exaggerated. Koch found that tubercle
bacilli were killed in from a few minutes to some hours, according
to the thickness of the layer in which they were exposed to the
sunlight. He found that even ordinary daylight produced the same
effect, if it lasted long enough; cultures of tubercle bacilli dying in
from five to seven days if exposed at the window in compact
masses. These experimental facts emphasise the importance of
abundant open space about dwelling-houses (see p. 203), the
provision of a large window-area (see pages 202 and 216), of
staircase ventilation, and lighting, &c.
Scrofula means a tubercular affection of the lymphatic glands.
It occurs most commonly in the neck. The infection is usually
received from some neighbouring mucous surface, as from the
throat, being derived from dried expectoration or diseased milk.
The same indications as for the prevention of phthisis hold good
for scrofula.
317
CHAPTER XLVI.
NOTIFICATION AND ISOLATION.
We are confident from the actual discovery of the micro-organisms
causing certain infective diseases, that the other diseases of an
analogous nature are similarly caused by living contagia. On this
supposition, action is taken for the prevention of these diseases.
This action comes under a number of different heads, which may be
classified as follows:—
1. Means for the early recognition of the infectious
character of a disease. The bacteriological aids to recognition
in diphtheria (page 299), enteric fever (page 301), and phthisis (page
310) have been already mentioned. It is important to call in medical
aid when any suspicious symptoms arise, even when these symptoms
do not appear to be urgent. If an infectious disease is not recognised
in its early stage, it may be easily overlooked, and the patient cause
a serious epidemic. The following hints for teachers are in Brighton
sent with each circular letter as to excluding infected children from
school. The list is not exhaustive, but may aid in drawing attention
to suspicious symptoms. The only safe rule when in doubt is to act
as though a case is infectious until a skilled opinion can be obtained.
HINTS AS TO INFECTIOUS DISEASES.
As infection is sometimes spread by means of children attending school while
suffering from undetected infectious diseases, the following hints may be useful to
the teacher:—
1. Any scholar having a sore throat should be sent home and regarded as
infectious until the throat has been examined by a doctor.
If a scholar has enlarged glands in the neck, and especially if he or she is
very pallid, the suspicion of possible diphtheria should be entertained. Many
slight cases of diphtheria escape detection.
2. Any scholar suffering from a severe cold, with sneezing, redness of the
eyes and running at the nose, should be sent home. It may mean an influenza
cold or the commencement of measles, and both are infectious. This recommendation
is particularly important when measles is known to be prevalent.
3. A child with a violent cough, especially if it is severe enough to cause
vomiting or nose-bleeding, should be suspected of whooping-cough, and sent
home, even if the characteristic “whoop” is not heard.
4. Slight cases of scarlet fever sometimes escape notice, and the patients
are sent to school with the skin on the hands, etc., freely “peeling.”
5. In any of the above instances, or any other case of suspicion, the
Medical Officer of Health, on receiving a confidential intimation, will be glad to
make an investigation.
318
SYMPTOMS OF ONSET OF SCARLET FEVER.
Sudden onset.
Usually vomiting.
Always headache.
Feverish, with dry, hot skin.
Sore throat.
Red rash on chest in a few hours.
MEASLES.
Severe “cold in the head” for 72 hours before the blotchy rash appears.
Measles is extremely infectious in this preliminary stage.
Consider every severe influenza cold as possibly measles.
DIPHTHERIA
may be very indistinct.
Languor and sore throat.
Glands under and behind jaw are enlarged.
Patient very pallid.
White or yellow patches seen on examining inside throat.
Whenever doubtful, send the scholar home.
WHOOPING COUGH.
In a child under seven, a severe cough should always be regarded as possibly
whooping-cough, although no “whoop” has yet been heard.
2. The notification of all cases of infectious diseases to the
medical officer of health, is clearly a means to an end, that of
securing that the preventive measures to be next named are
effectively carried out.
3. Means for the production of an artificial immunity.
This is only practicable at present for two diseases of this country,
small-pox by means of vaccination (page 293), and a temporary
immunity against diphtheria by a dose of antitoxic serum (page 299).
Apart from these means, any measures for improving the health of
a child tend in the same direction. Enlarged tonsils, “adenoids”
at the back of the nose (causing the child to snore at night and to
breathe through his mouth), discharges from nostrils or ear, and
similar conditions should receive early medical attention.
4. Isolation: preventing the conveyance of the contagium
from the sick to the healthy.
5. Disinfection, i.e. destruction of the contagium of the
disease.
The Infectious Disease (Notification) Act, 1889, and the corresponding
London Act of 1891, impose a dual duty of notification
(a) on every medical practitioner attending on or called in to visit
an infectious patient, as soon as he becomes aware of its nature;
and (b) on the head of the family to which the patient belongs or
the nearest relative. The intimation must be sent by each of these
to the local medical officer of health, the practitioner being paid a
small fee for his trouble. Usually notification by the householder319
is only enforced when no doctor is in attendance. The diseases to
which this Act applies are small-pox, cholera, diphtheria, membranous
croup11, scarlet fever, erysipelas, and the fevers known by
any of the following names: typhus, typhoid, enteric, relapsing,
continued or puerperal. The list of notifiable diseases may be
extended by resolution of the Local Authority.
The enforcement of notification is most important for the public
health. (a) It enables the medical officer of health to take immediate
steps to prevent the spread of infection, by enforcing proper
isolation of the patient, efficient disinfection, and by preventing the
attendance of children from infected houses, at school, etc. (b) It
enables the links of evidence connecting a series of cases to be
identified, e.g. cases due to a common milk supply, or attendance at
a particular school. (c) It has a valuable educational effect on all
concerned in the cases.
ISOLATION.
Both the patient and his attendant need to be isolated in diseases
like scarlet fever, diphtheria and small-pox. The rule is less absolute
in enteric fever. In the following description the standard of
requirements taken is that of the most dangerous infectious disease,
small-pox. The first point to decide is whether the patient may
be safely isolated at home. For small-pox this ought never to be
allowed in a town. For other diseases, this may be permitted, if
the following conditions can be fulfilled.
For Isolation at Home a couple of rooms are required,
preferably on a higher floor or in a detached wing of the house.
The w.c. used for the dejecta of the patient must not be used by
any other members of the household. All linen, towels, handkerchiefs,
etc., should be immersed in actually boiling water containing
some washing soda, before leaving the sick-room. Other articles
to be washed, if they will be deteriorated by soaking in boiling
water or a chemical disinfectant, must be tightly wrapped in bundles,
and covered with a clean wet sheet saturated with a strong disinfectant
solution (page 331). Solid and liquid excreta, expectoration and other
discharges must be treated as described on page 331. The nurse
should not eat her meals in the patient’s room. She should wear a
cotton dress to be changed before going out for a walk. Her
hands must be thoroughly washed and brushed after handling or
helping the patient, particularly in enteric fever. It is advantageous
if the nurse has previously had the patient’s complaint. Attention
on the part of the nurse to minute detail is essential, especially in
view of the possibility of receiving infection from infected articles
as well as directly from the patient. The measures required for the
subsequent disinfection of the sick-rooms and of clothing, bedding,
books, etc., are given on page 332.
The use of hospital isolation has rapidly increased in recent
years, thus releasing private families from a serious burden. The320
number of beds which a Local Authority should supply for their
district is usually stated as one for every 1,000 inhabitants, but in
poorer districts this does not suffice. The site of the hospital should
be well removed from houses. There must be a minimum zone of
40 feet between all infected buildings and the boundary walls, and
the same distance between neighbouring buildings. A wall at least
6 ft. 6 in. high should enclose the hospital site. The hospital is
divided into separate detached pavilions for the treatment of
different infectious diseases. A floor space of 156 square feet
should be allowed for each bed. The height of the ward
should be about 13 feet, its width from 24 to 26 feet, and the
total cubic space for each patient should be 2,000 cubic feet for
scarlet fever, 2,500 for diphtheria. The lavatories and water-closets
are separated from the main ward by a cross-ventilated lobby. In
an isolation hospital every surface should be washable; all corners
should be rounded off, and all projections on which dust can lodge
avoided. The proportion of window space should be about 1 square
foot to every 70 cubic feet. Special isolation pavilions are required
for cases of doubtful diagnosis. The ventilation and warming of
wards must be carefully regulated. Cross-ventilation by windows
open on opposite sides of the ward can be maintained in nearly all
weathers. The temperature of the ward should be maintained at
55°-60° F.
Ambulances are usually provided by the Local Authority for
the removal of infectious patients. The ambulance should be
cleansed and disinfected after each journey. The use of private
conveyances for infectious patients is forbidden, except under special
limitations.
The hospital isolation of small-pox is beset with special
difficulties. There is a considerable body of evidence indicating
that small-pox may be aerially carried from patients in hospitals to
people living within a zone of half a mile, or possibly further.
Without accepting the view that aerial dissemination of small-pox
to considerable distances from the patient frequently occurs, it still
remains true that, either by this means or by errors in the administration
of small-pox hospitals, they do frequently constitute a source
of danger to persons living in the vicinity. The Local Government
Board recommended that a Local Authority should not contemplate
the erection of a small-pox hospital. (a) On any site where it would
have within a quarter of a mile of it as a centre either a hospital,
whether for infectious diseases or not, or a workhouse, or any
similar establishment, or a population of 150-200 persons; (b) on
any site where it would have within half a mile of it as a centre a
population of 500-600 persons, whether in one or more institutions
or in dwelling-houses.
QUARANTINE.
This term has been chiefly employed to denote the limitation of
the movements of vessels coming from infected ports, for a term321
which, as the name indicates, was formerly forty days, but is now
shorter. It may be conveniently employed, however, to signify the
restriction of the movements of all persons who have been
apparently exposed to infection, or who continue to live in infected
dwellings. In this sense we may speak of:
- 1. Domestic Quarantine.
- 2. Scholastic Quarantine.
- 3. National and International Quarantine.
Domestic Quarantine, to a varying extent, is desirable for
the members of a family of which one member has been attacked by
an infectious disease. For small-pox every member of a household
should be kept under strict watch until sixteen days have elapsed
since the last contact with the case of small-pox, or until successful
vaccination has been secured. For enteric fever this strict watch
would be unnecessary, but the remaining members of the household
should be warned to call in a doctor on the first symptom of malaise.
Quarantine is specially indicated for certain occupations.
Thus if the child of an out-door labourer had been removed to a
hospital with scarlet fever, it would be unnecessary to keep the
latter away from work during the following week. If, however, he
were a milk-carrier, or a tailor, or an assistant in a sweet-stuff shop
this would be a desirable measure.
The Quarantine of School Children is more necessary than
that of adults, because the former are more susceptible to infection.
Children are kept from school:
(a) Because the infectious patient still remains in the house.
In this case the healthy children must be kept from school until the
patient has ceased to be infectious and disinfection has been
thoroughly carried out; and for a further period longer than the
longest known period of incubation of the disease in question
(page 287), a margin being left for contingencies. It would probably
be 8 plus 2 weeks for scarlet fever.
(b) Children are kept from school for a period exceeding the
longest period of incubation when the patient has been removed to
hospital.
The table on page 322, modified from the Author’s School
Hygiene, is introduced as furnishing a convenient summary of the
subject.
Objection is sometimes taken to the exclusion of children under
the above circumstances from school, on the ground that they
continue to mix with others in the street or in neighbouring houses.
Clearly, however, in a school-room, a suspected child may
communicate infection to children coming from widely scattered
streets, while out-of-doors the danger is comparatively slight, and
among neighbours the danger is very limited in area.
It is assumed in the following table that all infected articles have
been disinfected before the termination of the period of quarantine.
322
| DISEASE. |
DURATION OF INFECTION. |
DATE AT WHICH SCHOOL ATTENDANCE MAY BE RESUMED. |
DURATION OF QUARANTINE OF CHILDREN EXPOSED TO INFECTION. |
| Scarlet fever |
From 5 to 8 weeks; ceases when all
peeling of the skin has been completed, and when the child is
free from discharge from the nose or ear or sore places. |
Not less than 8 weeks 14 days. from the beginning
of the rash, and then only if no sore throat or sore places. |
14 days. |
| Diphtheria |
At least 21 days; often much longer.
Absence of infection should be confirmed by bacteriological tests. |
Not less than 2 months, and not
then if strength not recovered, or if any sore throat or any
discharge from nose, eyes, ears, etc. |
12 days. |
| Small-pox and Chicken pox |
About 4 to 5 weeks |
When every scab has fallen off. |
18 days. |
| Measles |
From 3 to 4 weeks; when all cough and
branny shedding of skin has ceased. |
Not less than 4 weeks from beginning of rash. |
21 days. |
| Rötheln (German measles) |
2 to 3 weeks |
From 3 to 4 weeks from beginning of rash. |
21 days. |
| Mumps |
About 21 days from the beginning. |
4 weeks from the beginning. |
24 days. |
| Whooping cough |
6 weeks from the beginning of
whooping, or when the cough has quite ceased. |
In about 8 weeks |
21 days. |
| Typhus and enteric fevers |
4 to 5 weeks |
When strength sufficient. |
28 days. |
| Influenza |
2 to 3 weeks |
1 month |
10 days. |
School Closure is occasionally required to prevent the further
spread of an infectious disease. This can be enforced on the order of
any two members of the Local Sanitary Authority acting on the advice
of the medical officer of health. This ought to be only occasionally
necessary if notification of infectious diseases is strictly enforced,
and if suspicious individual children are excluded from attendance
at school. In diphtheria school closure may occasionally be
rendered unnecessary by systematic bacteriological examination of
the throats of children who had been exposed to infection (see page
299). School closure is more useful for country than for town
schools, as in the former the homes of children are more remote
from each other, but it is occasionally necessary for both. For
measles school closure is specially indicated in Infants’ Schools.
We have already seen that this disease is chiefly fatal when caught
at a tender age (page 297). The early closure of Infants’ Schools,
and particularly of the Babies’ Class is therefore indicated. It is
unfortunate that the attendance at school of children under six years
of age is encouraged. Such children have more severe and more323
frequently fatal attacks of diphtheria, scarlet fever, measles, and
whooping-cough; and these are frequently acquired at school.
International Quarantine was originally enforced against
plague; but in many countries has been extended to other diseases,
as cholera, yellow fever, typhus fever, small-pox and leprosy. In
England cholera is the only disease in connection with which it has
been in the past enforced. It has now been entirely abandoned.
It consists in the compulsory isolation at the port of entry of all
persons who have come from an infected district, or have been in
contact with a case of the infectious disease against which
quarantine is enforced, for a length of time which will enable it to
be determined whether the persons detained are or are not
incubating the disease. If this measure could be strictly enforced,
and if infectious diseases were conveyed only by infectious persons,
quarantine would undoubtedly be effective. But in practice
quarantine cannot be enforced in Europe; and as it cannot be
efficiently enforced it forms an ineffective and irrational derangement
of commerce. Thus if plague prevailed in France it would
be impracticable to detain for ten or twelve days every person
entering England. Furthermore, in this instance, infection is
brought by rats as well as persons; and measures effective for the
latter do not prevent the former from importing infection. Because
of its impracticability and of the disorganization of commerce which
would be associated with any attempt to enforce it, England has
abandoned quarantine and other countries are gradually following
its example. England bases its action on the ground that (a)
sanitation is the true chief means of defence, especially against cholera. It
does not trust to this alone but to this along with (b) medical
inspection at the ports, (c) and subsequent medical supervision of persons
landed from suspected vessels. By these means a watch can be
kept over persons who have been in contact with infection.
Regulations are issued at intervals by the Local Government
Board requiring the disinfection by steam of all rags and similar
materials imported from towns in which small-pox, cholera, etc., are
prevalent.
324
By disinfection is meant the destruction of the active cause of each
infectious disease. A disinfectant is therefore synonymous with a
germicide. Disinfectants must be distinguished from deodorants or
deodorisers, such as charcoal, and from antiseptics, which are
antagonistic to the growth of bacteria, without necessarily killing
them, e.g. common salt. Disinfection may be effected by chemical
or physical means.
CHEMICAL DISINFECTANTS.
A chemical disinfectant should fulfil the following conditions:
1. It must be an efficient germicide. 2. Its germicidal power should
not be destroyed by the fæcal or other polluting matter, with which
the bacteria of infection are associated. 3. For many purposes, it
must not be destructive to or liable to stain the skin, or fabrics, or
other articles to which it is applied. 4. It should preferably not be
a virulent poison; and should be moderately cheap. The search
for a completely non-poisonous disinfectant is a chimera.
There are three great classes of chemical disinfectants.
1. Oxidising agents, as the halogens (chlorine, etc.) and
permanganates.
2. Deoxidising agents, as sulphurous acid (SO₂) and formic
aldehyde (CH₂O).
3. Other disinfectants, which act by coagulating protoplasm or
otherwise, as carbolic acid, corrosive sublimate.
The number of disinfectants is legion. Only the chief ones
can be mentioned and their chief properties described. It is a good
rule to eschew the use of all disinfectants of which the exact
composition is not given; and all disinfectants which are described
by “fancy” names, which are not descriptive of their composition.
A. Oxidising Agents.
Chlorine has been most commonly used as chloride of lime
(CaCl₂, Ca(ClO)₂). This is somewhat unstable in composition.
A solution of sodium hypochlorite containing 10 per cent. of
available chloride is preferable. Chloride of lime for sprinkling on
decomposing matter should contain at least 10 to 15 per cent. of325
available chlorine. Sulphuretted hydrogen and other offensive
gases are decomposed by it. Thus
SH₂ + Cl₂ = S + 2HCl. Its chief action is as an
oxidising agent.
Thus H₂O + Cl₂ = 2HCl + O.
A large excess must be used in disinfecting, otherwise the
chlorine may simply oxidise fœcal or other organic matter, and not
effectually destroy contagia.
Methods of Use—(a) As a gas, by the action of hydrochloric
acid on strong chloride of lime. The molecular density of chlorine
is 35·5, of formic aldehyde 15, of sulphurous acid 32; and the rate
of diffusion of gases being inversely to the square-root of their
densities, clearly chlorine does not compare favourably as a gas
with formic aldehyde. The bleaching effect of chlorine on coloured
articles of apparel is a disadvantage.
(b) As a liquid: used thus chlorine is very efficient, applied
either as a spray or brushed on walls and other surfaces. Delépine
found that a solution of one part of chloride of lime in 100 parts of
water applied to wall paper impregnated with tubercular matter,
disinfected it in a few hours, or in a few minutes if the layer of
infected matter was not thick.
Bromine, Iodine, and Euchlorine (a mixture of chlorine and
Cl₂O₄) are efficient disinfectants.
Iodine Trichloride (ICl₃) was found by Behring to share
with corrosive sublimate (HgCl₂) carbolic acid and cresol mixed
with acids (see page 327), the halogens (Cl, Br, and I), and chloride
of lime a superiority over other disinfectants in their power of
killing anthrax spores in a short time.
Permanganates have been largely used as disinfectants, but
their value is small. Impure sodium manganate (Na₂MnO₄) with
much common salt (NaCl) containing some permanganate is known
as “Condy’s Green Fluid.” “Condy’s Red Fluid” consists of
permanganate and sulphate of soda. To be of any use it must be
employed in 5 per cent. solution. It stains fabrics brown, and it
exhausts its feeble disinfecting power in first oxidising decomposing
organic matter.
B. Deoxidising Agents.
Sulphurous Acid (SO₂) acts chiefly as a reducing agent on
organic matter. It is used chiefly as a gaseous disinfectant, and
for this purpose is generated (a) by burning 1 lb. of sulphur for
every 1,000 cubic feet of space in the room (which will equal 1·12
per cent. of SO₂). The windows and chimney of the room are first
closed; the sulphur is placed in a saucepan supported over a
bucket of water, and its ignition is aided by a small quantity of
methylated spirit. The door of the room is then sealed, and the
room left until the next morning. (b) Carbon disulphide may be
burned in a benzoline lamp. (c) SO₂ liquefied under pressure is
supplied in cylinders available for convenient use. The experimental
results of the action of SO₂ on various bacteria are somewhat
discrepant. It probably is fairly efficient for some diseases, but not
in tuberculosis.
326
Formaldehyde or Formic Aldehyde (CH₂O) is produced by
the slow and incomplete oxidation of methyl alcohol (CH₃OH)
under access of air. A saturated solution in water containing 40
per cent. of the formaldehyde gas is known as formalin. The
simple evaporation or heating of formalin is liable to produce the
polymeric paraform which is solid and inert. To prevent the
formation of paraform when formalin is evaporated, Trillat adds to
it a solution of calcium chloride (CaCl₂), the mixture being known
as formochloral. It is stated that when the air of a room is charged
with less than one per cent. of the vapour, rapid and complete
disinfection of surfaces occurs, and that it possesses a certain
amount of penetrating power into loose fabrics. No damage is
done to textile fabrics; and disinfection by this means possesses
the advantage over disinfection by sulphurous acid or chlorine that
the room can be entered without serious discomfort soon after the
disinfection is carried out. In solution formalin is undoubtedly a
powerful disinfectant, and in the gaseous condition it is at least
equal in value to SO₂, probably better. Formaldehyde is used as
a disinfectant.
(a) By evaporating a 60 per cent. solution of CH₂O in methyl
alcohol (trade name holzine) over pieces of glowing coke placed
under an asbestos plate (Opperman-Rosenberg apparatus). (b) By
subliming tabloids of paraform by the heat of a lamp. A
methylated spirit lamp is employed, and the moisture from the
combustion in this causes the transformation of a considerable
proportion of paraform into CH₂O vapour. It is doubtful if the
quantity of the latter evolved is sufficient for efficiency. (c) In
Trillat’s apparatus formalin (i.e. the 40 per cent. solution in water of
CH₂O) with CaCl₂ solution is heated in an autoclave worked at a
pressure of 40 lbs., provided with a pressure gauge and thermometer.
In all these methods the room must be carefully sealed, as the
tendency for the disinfectant to escape is greater than with SO₂ or
Cl. (d) The best method is to spray a solution of formalin 4 oz. to
one gallon of water on all the surfaces of the room (see page 333).
This is equal to a strength of 1 in 40 of formalin, or 1 in 100 of
formic aldehyde.
C. Other Disinfectants.
Tar Acids.—When coal tar is treated by acids and alkalies in
succession, it becomes separated into (1) hydrocarbons, (2) phenols
or tar acids, carbolic, cresylic, etc., (3) aniline and other basic
substances. The hydrocarbons are known in commerce as
“neutral tar oils.” They are brown and syrupy, turning milky
with water, and feebly disinfectant. The two most important “tar
acids” are phenol or carbolic acid (C₆H₅OH) and methyl-phenol,
also called cresol or cresylic acid (C₆H₄(CH₃)OH). The higher
members of this same group yield milky emulsions with water, and
are less poisonous than phenol. Various mixtures of them are used
as disinfectants, and sold as creolin, Jeye’s and Lawes’ fluids.
Izal belongs to the same series.
327
Carbolic Acid (phenol) did not kill anthrax spores until a 3
per cent. strength of its solution was used for 7 days (Koch), but
sporeless anthrax bacilli were destroyed in a few minutes by a 1 to
2 per cent. solution. The disinfecting power of carbolic acid is
greatly increased by adding mineral acids. Carbolic acid and lysol
are superior to creolin for disinfecting stools. A 5 per cent.
solution of carbolic acid destroys tubercle bacilli in sputum in 24
hours. Carbolic acid powders are in common use. In my opinion
quicklime is more valuable.
Cresol is obtained from “crude carbolic acid” by fractional
distillation at a temperature between 185° and 205° C. A one-half
per cent. solution has equal disinfecting power to a 2 or 3 or
sometimes a 5 per cent. of phenol (carbolic acid).
Creolin consists of cresol emulsified in a solution of hard
soap. Behring classifies the comparative germicidal power of
phenol, cresol, and creolin on bacteria in broth as 1, 4, and 10
respectively. When albumen is present, creolin loses a part of its
disinfectant power.
Lysol contains 50 per cent. of cresol, dissolved by means of
neutral potash soap. It is completely soluble in water and does
not turn milky as creolin does when water is added. It is more
effective than creolin, and still more than HgCl₂ in albuminous
liquids.
Soap has, owing to its alkalinity, disinfectant as well as
cleansing action. A temperature of 55°-75° C. greatly aids its
action. Antiseptic soaps possess no special value as germicides, but
carbolic soap is a useful insecticide.
Lime in a one-tenth per cent. solution destroys typhoid and
cholera microbes.
Mercuric Chloride (HgCl₂, corrosive sublimate) was found
by Koch to destroy anthrax bacilli in a dilution of 1 in 20,000.
Others have obtained less favourable results, but it is certainly a
powerful germicide. The germicidal effect is greatly diminished by
contact with organic matter, an insoluble albuminate of mercury
being produced. For this reason HgCl₂ is not the best disinfectant
for fæces unless mixed with acid, as in the following solution:
HgCl₂ ½ oz., HCl 1 oz., aniline blue 5 grains to three gallons of
water. This gives a solution of 1 in 960. The colouring is added
to avoid accidental poisoning. HgCl₂ is not a good disinfectant
for linen. Stains are apt to be fixed by it, and if linen soaked in it
is subsequently washed with soap, without first carefully washing
out the HgCl₂, it is darkened in colour. It attacks metals, and
must not therefore be placed in metal receptacles.
Chloride of Zinc in a solution containing 25 grains to the
fluid drachm is known as “Sir William Burnett’s solution.” It is
a good deodorant, but an inefficient disinfectant.
Chinosol (C₉H₆NKSO₄) belongs to the quinoline group.
It is an almost inodorous powder, very soluble in water, noncorrosive,
and does not stain. A solution of 1 in 1200 forms an
efficient germicide.
328
DISINFECTION BY PHYSICAL MEANS.
Natural processes tend to the destruction of pathogenic microbes
after their elimination from the patient. Of these desiccation, sunlight,
and fresh air are the most potent. Heat and cold have a
similar effect. Filtration or mechanical separation deprives a
contaminated liquid of its microbes.
Desiccation attenuates the virulence of and finally kills most
microbes. In laboratory experiments the vibrio of Asiatic cholera
when dried dies in from three hours to two days, according to the
degree of desiccation. The bacilli of enteric fever, tuberculosis, and
diphtheria only die after drying for a few weeks or even months.
The anthrax bacillus may retain its vitality for several years in a
desiccated state. Clearly, therefore, desiccation has no administrative
value in the prevention of disease, and on the contrary it aids
the dissemination of the microbes of tuberculosis, small-pox, scarlet
fever, etc.
Direct Sunlight kills a large proportion of the sporeless
pathogenic microbes. Diffuse is less energetic in its action than
direct sunlight. The bacillus of diphtheria is destroyed by a half to
one hour’s exposure to sunlight. As to tubercle bacilli, see page 316.
Downes and Blunt showed that diffused sunlight retards the putrefaction
of organic infusions, and that direct sunlight inhibits
putrefaction. Sunlight cannot, however, be trusted as an efficient
disinfectant. It only secures surface disinfection, and could not be
relied upon for pillows, mattresses, etc. M. Ward’s experiments
showed that the actinic rays of the sun are germicidal, independent
of the heat.
Fresh Air, like sunlight, should be employed as a valuable
auxiliary, not as an agent to be depended upon apart from systematic
disinfection. The experiments of Downes and Blunt showed that
light and oxygen together accomplished what neither alone could do.
The presence of air in anthrax increases danger. The anthrax
bacillus does not form spores in an animal suffering from this
disease, and does not do so post mortem, unless the animal is
dissected. Hence the importance of keeping the skin unbroken in
this disease, only examining a drop of blood to establish the
diagnosis.
Filtration is a means of separating microbes from the gases or
liquids containing them. For the filtration of water see page 96.
The carbolic sheet outside a sick-room is supposed to filter the air
leaving the room from microbes. It is probably useless except as a
reminder to the nurse to change her dress and adopt other precautions
on leaving the sick-room.
Settlement of dust also acts as an aerial disinfectant. If a
room be locked up, its air next day is almost free from particles,
and all that is then required is disinfection of the surfaces of the room
and of the articles in it. Whatever method of disinfection is
employed, it is not disinfection of the air, but of the surfaces of a
room which is the end in view.
329
Washing is the most efficacious means of removing infection.
It is a mechanical means of removing the particular matter of which
the contagium consists from the person or article to which it
adheres into the water, which subsequently enters the drain, in the
same way as do urine and fæces. Washing is an absolutely efficient
means of purifying articles that can be completely submitted to it.
A consideration of the physical laws governing the spread
of infection will make this clear. The contagia are passive.
When contained in a liquid they cannot escape from it under
ordinary circumstances. Thus foul smelling gases may escape
from sewage, but bacteria do not escape, except rarely during
bubbling, or from dried portions of the invert of the sewer.
Barring rare accidents “microbes submerged are imprisoned.”
Contagia are harmless until they become dust. Hence the danger
associated with the use of pocket handkerchiefs in such diseases as
influenza and phthisis; and the importance of keeping all infectious
discharges wet, until they can be finally disposed of.
DISINFECTION BY HEAT.
Heat may be applied in various ways: (1) Prolonged boiling
in water of materials which are not spoilt by this means. (2)
Destruction by fire of infected articles. (3) Dry hot air. (4)
Steam.
Boiling kills most pathogenic microbes. The cholera vibrio
is killed in four minutes at a temperature of 52° C. (126° F.); the
typhoid bacillus at 59°.4 C. (138°.8 F.) in ten minutes. If boiling
be continued for five minutes, the spores of pathogenic microbes
are killed. The addition of one to two per cent. of washing soda
to the water hastens this effect. For infected linen nothing beyond
this is required.
Destruction by Fire is to be recommended for comparatively
worthless articles, such as toys, straw from beds, rags, old clothing
and bedding.
Dry Hot Air has been largely used in the past in ovens, for
the disinfection of bulky bedding. It is now entirely superseded
by steam. Its disadvantages are that (a) heat penetrates very
slowly into the interior of bedding. Disinfection in test experiments
was not accomplished in the interior of small bundles of clothes in
three or four hours. (b) Scorching of articles often occurs. The
sole advantage of this method is that bound books and leather
goods are less liable to be damaged by it than by steam. If no
other apparatus is available a baker’s oven will serve to kill the
non-sporiferous microbes of cholera, enteric fever, and diphtheria,
as well as animal vermin. If, however, we accept the proper test
proposed by Buchanan of the efficacy of disinfection, the “destruction
of the most stable known infective matter,” dry heat is
unsatisfactory.
Steam may be employed as a disinfectant either (a) superheated,
or (b) saturated, i.e. close to the temperature at which condensation
occurs. This temperature depends upon the pressure under which330
the water has been boiled. At ordinary atmospheric pressure it is
100° C. (212° F.). The temperature of boiling is raised by subjecting
the water to pressure. Consequently boiling water and the steam
produced from it may be at any temperature. Thus steam may be
- (a) Under pressure, with a temperature above 212° F.
- (b) Not under pressure, at a temperature of 212° F.
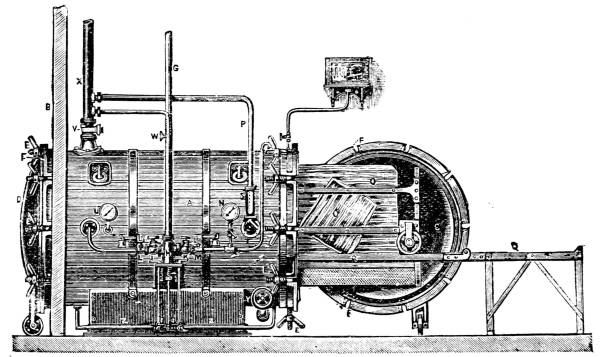
Fig. 57.
Equifex Saturated Steam Disinfector.
A—Disinfection chamber. B—Partition wall separating infected from disinfected side. C—Door
on disinfected side. D—Door on infected side. EE—Safety-locking bolt for securing door.
FF—Stiffening rings on doors. G—Steam inlet from boiler. H—Steam separator for arresting water
condensed in G. I—Valve controlling admission of steam to disinfecting chamber. K—Valve
controlling admission of steam to coils. LM—Safety valves regulating steam pressure in chamber
and coils respectively. NU—Pressure guages indicating steam pressure in chambers and coils
respectively. OO—Objects after disinfection. P—Wheeled carriage and cradle for OO. Q—Hinged
rails on which P runs. R—Exhaust pipe for steam and air on first admission. S—Thermometer
showing rise of temperature (to control complete air evacuation). T—Valve for closing exhaust
pipe R when air is completely evacuated. V—Sluice valve to cause sudden escape of steam.
W—Cock to admit steam to ejector. X—Exhaust pipe fitted with ejector for escape of steam before
and of air during drying. Y—Valve for admission of air for drying under suction of ejector. [In
some types of this machine this valve is placed on the lower part of C.]
Steam when admitted into a disinfecting stove comes into
contact with cold objects. If the steam is saturated, immediate
condensation to 1 ∕ 1600 part of its original volume occurs. Its latent
heat is at the same time evolved. The condensation causes
enormous shrinkage in bulk. More steam is thus insucked into
the partial vacuum produced, and this is repeated, until in every
part of the mattress or other material undergoing disinfection
equality of temperature is reached, when condensation of steam will
cease, and disinfection is complete. If the steam is superheated and
no condensation allowed, disinfection occurs by the relatively slow
method occurring with dry heat. In practice at the early stage
cooling causes some conversion of superheated into saturated
steam, though subsequently the much slower process of disinfection331
by conduction of heat goes on. Hence superheated steam is a less
efficient disinfecting agent than saturated steam.
Superheating is produced in disinfecting stores in two ways:
(1) By a jacket around the stove, which is kept at about double the
pressure and about 20° to 30° F. hotter than the interior of the
stove; as in the older patterns of the Washington Lyon stove.
(2) By having a jacket containing a solution of calcium chloride,
which is heated by a furnace under the stove. This solution
is kept at a constant strength by an automatic supply from a
cistern. The temperature of the boiling water is thus raised
without pressure to 225° F. This is the principle of Thresh’s stove.
The object of superheating steam is to assist in rapidly drying
materials; but this object can equally well be secured by
periodically allowing the sudden escape of the steam confined
under pressure, in pressure disinfectors. This last method is the
best, as it can not only be utilised at the last stage of the disinfection
for drying the articles; but at the earlier stage for sweeping the
air out of the stove, and thus removing what, owing to its low
conductivity for heat is one of the most serious obstacles to rapid
and efficient disinfection.
In the above description it has been assumed that the steam,
whether saturated or superheated, is confined, except when the
exhaust is employed for drying purposes. Steam may also be
employed as current steam. Current steam disinfectors are
initially cheap, but more steam, and therefore more fuel, is required
in their use; and unless pressure is used by impeding the escape of
the steam a temperature of only 212° F. can be secured. Accepting
Buchanan’s dictum, a stove supplying saturated steam under
pressure at a temperature in the interior of the stove of 234° F. is
to be preferred. This temperature with saturated steam destroys
the spores of the most resistant known microbe (that of symptomatic
anthrax). With superheated steam or hot air stoves on the same
basis a temperature of 280° F. would be required, which is damaging
to most textiles, except horsehair.
THE PRACTICE OF DISINFECTION.
The Management of the Sick-room and Patient requires
careful and conscientious attention to detail. Certain details are
given on page 319. All unnecessary furniture, carpets, and hangings
should be removed as soon as the nature of the illness is known;
but unless these articles have been contained in close trunks or
drawers, and not opened since before the onset of the illness, they
must be disinfected. Food left over from the patient’s meals must
be burnt, if solid, in the patient’s room; if liquid, emptied down
the water-closet. Dry sweeping of the floors is to be avoided, only
wet brushing or cloths being used. Volatile aerial disinfectants
during the sickness are valueless.
The Treatment of Discharges from the Patient is the most
important point in the management of infection. The stools should
be received into a bed-pan containing a 5 per cent. solution of332
carbolic acid, a 3 per cent. solution of cresol or lysol, or a 5 per
cent. solution of chloride of lime. Milk of lime (20 per cent.
strength) is very reliable, when added like the preceding solutions
in bulk equal to that of the stool to be disinfected. The urine and
vomit, if any, should be treated in exactly the same way. The
infection of enteric fever is often spread by undisinfected urine.
Discharges from the throat, nose and mouth of patients
should be received into a solution of
- lysol 5 oz. to 1 gallon of water, or
- carbolic acid 7 oz. to 1 gallon of water,
The efficacy of the carbolic acid solution is increased by adding
2 oz. of NaCl, or 12-14 oz. of NaCl to each gallon. Pocket-handkerchiefs
must be avoided, linen rags being employed instead,
and placed at once in one of the above solutions or burnt.
The skin may scatter infection, especially in small-pox and
scarlet fever. Frequent baths and inunction with vaseline or oil
are useful.
The disinfection of hands is most important for all attendants
on the infectious sick. A solution of corrosive sublimate 1-1000,
or one of the above solutions may be used for this purpose; but
this is to be supplemented by the free use of the nail-brush and soap
and water. The treatment of linen has been described (page 329).
Woollen articles of underclothing, and blankets can be
disinfected by steam, which shrinks them less than boiling water.
The ordinary laundry processes appear, however, to suffice to rid
them of infection, without boiling.
Bedding, curtains, and carpets should be disinfected by steam.
Certain precautions are required in removing these to the disinfecting
station. Surface disinfection of the room must have been first
effected (see below); and the infected bedding should be encased in
canvas bags or sheets. When a steam disinfector is inaccessible,
the mattress and pillows should be taken to pieces, the covers
washed, and their contents disinfected by spraying with formalin
solution (1 in 40) or HgCl₂ solution (1 in 1,000), and subsequently
exposing to sun and air. For disinfection of suits of clothes,
current steam may be improvised as follows:—Over two bricks at
the bottom of the kitchen “copper” thin floor-boards are placed,
above the level of 2 or 3 inches of water previously placed at the
bottom of the copper. The cover of the copper is put on, and by
means of a brisk fire steam is kept streaming through the clothes.
This is continued for an hour, and the clothes then hung out to dry.
Furniture, when wooden, can be washed. If upholstered it
can be disinfected by spraying (see p. 333), and then beating and
dusting in the open air.
Furs, Boots, and Shoes are spoilt by steam. For the first,
spraying freely with formalin (1 in 40), or exposure over a formalin
lamp (page 326) is recommended. Boots and shoes should be filled
and washed with a solution of HgCl₂, chinosol, or formalin.

Fig. 58.
Equifex Spray Disinfector in Use.
The sick-room can only be efficiently disinfected after the
patient has left it. The aim is surface disinfection. Aerial disin333fection
is sufficiently effected by open windows. Four chief
methods of surface disinfection are practised. (a) Fumigation by
SO₂, formalin, cresol, or other vapours (see page 326). (b) Spraying
the ceiling, walls, floor, and furniture with a disinfectant solution
is probably the most convenient method of disinfection. It is more
effectual than fumigation, less laborious than rubbing down walls,
etc., by bread or wet cloth, and less likely to damage wall-papers
than brushing a disinfectant solution on them. Solutions of HgCl₂
1 in 1,000, or chinosol 1 in 1,200, or formalin 1 in 40 are efficient.
A special spray apparatus (Fig. 58) is usually employed. A practical
point is to spray the wall from below upwards, to prevent the solution
running down the wall and producing streaks of discolouration.
(c) Washing ceiling and walls with the disinfectant solution may
be substituted. A one per cent. solution of hypochlorite of lime
is largely used for this purpose, applied by a long-handled334
whitewash brush. (d) Attrition of walls, etc., by means of bread
or dough sterilises them by mechanically removing microbes. The
bread is cut into pieces suitable for grasping in the hand, the cut
surface being applied to the wall. The crumbs must afterwards be
burnt in the room. This is the official method in Germany.
Floors may be treated like walls and ceiling after the patient
has left the room. During his occupancy of the room, tea-leaves
or sawdust thoroughly impregnated with lysol or cresol should be
sprinkled on the floor before it is swept, or washing substituted for
sweeping. Scrubbing with soap and water constitutes the best
disinfectant for floors and all other washable surfaces.
Books are difficult of disinfection. Steam damages leather.
The penetrating power of dry heat is doubtful. Cheap books
should be burnt. Abel discovered virulent diphtheria bacilli on
toys six months after the patient, to whom they belonged, had
diphtheria. Formalin and phenol vapours have been used to
disinfect books in closed chambers, the books being stood on end.
Letters can be rendered safe by steam disinfection.
Corpses of infectious patients should be placed in the coffin and
buried as early as possible. A thick layer of sawdust saturated with
lysol or cresol should be placed at the bottom of the coffin, and the
corpse enveloped in cotton wool. Cremation is better than burial.
335
Vital Statistics is the science of numbers applied to the life-history
of communities. Its significance is similar to that of the
more recently coined word—Demography—though the latter does
not necessarily confine itself strictly to study of life by statistical
means. Another term has been frequently used in recent years—“Vital
and Mortal Statistics.” The continued use of the word
“mortal” in this connection is undesirable and objectionable. The
term “Vital Statistics” is comprehensive and complete, as death
is but the last act of life.
Of the problems of life with which the science of Vital
Statistics is concerned, population, births, marriages, sickness, and
deaths, possess the chief importance; and in the following sketch of
the subject I shall concern myself chiefly with these. The subject
naturally divides itself into two sections: the sources of information,
and the information derived from these sources, and both of these will
require consideration.
The importance of numerical standards of comparison in
science increases with every increase of knowledge. The value of
experience, founded on an accumulation of individual facts, varies
greatly according to the character of the observer. As Dr. Guy
has put it: “The sometimes of the cautious is the often of the
sanguine, the always of the empiric, and the never of the sceptic;
while the numbers 1, 10, 100, and 100,000 have but one meaning for
all mankind.” Hence the importance of an exact numerical statement
of facts. The sneering statement that statistics cannot be
made to prove anything can only be made by one ignorant of
science. In fact, nothing can be proved without their aid, though
they may be so ignorantly or unscrupulously manipulated as to
appear to prove what is untrue. Instances of fallacious use of
figures will be given as we proceed.
An accurate statement of population forms the natural basis
of all vital statistics. Thus the comparison of the number of deaths
in one with the number of deaths in a second community has
no significance unless we know also the number living out of which
these deaths occurred. Even then our knowledge would be
defective, without further particulars as to the proportion in each
population living at different ages, and the number dying at the
corresponding ages. For other purposes we should require to know336
the number married and unmarried, the number engaged in different
industries, and so on; in order that the influence of marital conditions,
of occupation, etc., on the prospects of life may be
calculated. The first desideratum of accurate vital statistics is a
census enumeration of the population at such intervals as will not
cause the intervening estimates of population to be very wide of the
mark. In this country a decennial census is taken, the last
occurring in 1901. In the intervals the population of the entire
country, and of each town or district is estimated. Various methods
of estimating the population have been adopted. (1) If a strict
record of emigration and immigration is kept, then in a country in
which a complete registration of births and deaths is enforced, the
population can be easily ascertained by balancing the natural
increase by excess of births over deaths, and the increase or decrease
due to migration. This is done in New Zealand, but is impracticable
in England, as no complete account of migration can be kept.
(2) The increase of inhabited houses in a district being known
year by year, the increase of population may be estimated on the
assumption that the number of persons per house is the same as at
the last census. This may not be strictly accurate. In 1901 it was
found that in England and Wales the average number of persons
per house was fractionally less than in 1891.
(3) It may be assumed that the annual increase during the
present decennium will be 1 ∕ 10 of the increase during the last
decennium 1891-1901. If so, the population, e.g. in 1905, is the
enumerated population in 1901 plus 4¼; times the annual increase
occurring during 1881-91. (The fourth is required because the
census is taken early in April, and the population is estimated to
the middle of the year). This method is fallacious, because it
makes no allowances for the steadily increasing numbers who year
by year attain marriageable age and become parents. It assumes,
in other words, simple interest, when compound interest is in
operation.
(4) The Registrar-General’s method, the one generally adopted,
assumes that the same rate of increase will hold good as in the preceding
intercensal period, i.e. that the population increases in
geometrical progression, and not in arithmetical progression as
under (3).
The application of this method will be best understood by an
example. If the census population of a town is 32,000 in 1891, and
36,000 in 1901, what is the mean population in 1905?
(a) Find the rate of increase in 1891-1901.
- If P = population at census 1891,
- and if P1 = population at census 1901,
- and if R = rate of increase of population, then
- P1 = P + Rn in the nth year.
- log P1 = log P + 10 log R.
- 1 ∕ 10(log P1 - log P) = log R.
- (4·556303 - 4·505150) ∕ 10 = ·0051153 = log R.
337
(b) Apply this to the increase in the next 4¼ years.
- Here P1905 = P1901 R(17 ∕ 4)
- log P1905 = 4·556303 + (17 ∕ 4)(·0051153)
- = 4·578043.
By consulting the table of logs, the population corresponding to this
number will be found to be 37,848 = population at the middle of 1905.
Estimates made by the last-named “official” method are liable
to error, even for the entire country, and still more when applied
to special districts. Thus the decennial rate of increase of the
population of England and Wales in the 100 years has varied from
15·8 per cent. in 1821-31 to 11·6 per cent. in 1891-1901. The anomalies
are even greater when the official method is applied to great towns. In
one decennium such a town may, owing to brisk trade, have a rapid
increase of working population with many children, and in the next
decennium in consequence of emigration or transmigration there
may be little or no increase. The declining birth-rate, which is
having a greater effect on the number of population than the
declining death-rate, is another cause of disturbance which increases
the difficulty in forming a correct estimate of the population in intercensal
periods. A quinquennial census is highly desirable, in order
to avoid the doubts necessarily associated with estimates of
population in the later years of a decennium, and with the birth and
death-rates which are based on these estimates.
The Registration of Births and Deaths.—Civil registration
of births and deaths began in 1837, but was not compulsory till 1870.
It will be going beyond the scope of this chapter to give details of
the enactments as to registration. It suffices to state that it is the
duty of the practitioner to give a certificate stating the cause of
death of his patient to the best of his knowledge and belief. There
is no registration of still-births in this country. Many deaths are
registered of which the cause of death is not medically certified, and
the value of our national vital statistics is considerably diminished
on this account. Much improvement is desirable in the medical
certification of causes of death. Every medical student ought to
receive instruction on this subject before the completion of his
studies. Names of symptoms as dropsy, hæmorrhage, convulsions;
and obscure names, as abdominal disease, should be avoided. If
the patient has recently suffered from injury, or recently passed
through childbirth, or had a specific febrile disease, this must not
be omitted from the certificate.
The Registration and Notification of Sickness forms another
valuable source of information. Various attempts have been made
to secure a general registration of disabling sickness, but with only
partial success. District and workhouse medical officers appointed
since February, 1879, are required to furnish the medical officer of
health with returns of pauper sickness and deaths. This source of
information might with advantage be more fully utilised by medical
officers of health. Sec. 29 of the Factory and Workshops Act,338
1895, requires that every medical practitioner attending on or called
in to visit a patient whom he believes to be suffering from lead,
phosphorus, or arsenical poisoning, or anthrax, contracted in any
factory or workshop, shall send to the Chief Inspector of Factories
at the Home Office, London, a notice stating the name and full
postal address of the patient, and the disease from which he is
suffering; a fee of 2s. 6d. being payable for each notification, and a
fine not exceeding 40s. being incurred for failure to notify.
The Compulsory Notification of Infectious Diseases is
enforced by the Act of 1889, which now applies to the whole
country. The list of diseases to be notified is as follows:
“Small-pox, cholera, diphtheria, membranous croup, erysipelas, the disease
known as scarlatina or scarlet fever, and the fevers known by any of the following
names: typhus, typhoid, enteric, relapsing, continued, or puerperal, and also any
infectious disease to which the Act has been applied by the Local Authority in
manner provided by the Act.”
It is the duty of the medical practitioner to ascertain whether in
his own district, such diseases as whooping cough and measles
have been added to the schedule of notifiable diseases. It is the
duty of (a) the head of the family to which the patient belongs;
in his default, of (b), the nearest relatives in the house; in their
default, of (c), every person in attendance upon the patient; and in
default of any such person, of (d) the occupier of the building, as
soon as they become aware that the patient is suffering from an
infectious disease to which this Act applies, to send notice thereof
to the Medical Officer of the District. (e) The more formal duty
of sending to the Medical Officer of Health a certificate stating
the name of the patient, the situation of the building, and the
infectious disease from which in his opinion the patient is suffering,
is imposed on every medical practitioner attending on, or called
in, to visit the patient, on becoming aware that the patient is
suffering from an infectious disease to which this Act applies. He
is entitled to a fee of 2s. 6d. if the case occurs in his private practice,
and of 1s. if the case occurs in his practice as medical officer
of any public body or institution. He is subject to a fine not
exceeding 40s. if convicted of failure to notify. The value of
returns of infectious diseases as enabling preventive measures to
be taken is increased by interchange of notification returns of
different districts. This is now undertaken weekly for a large
number of districts by the Local Government Board, and the
Registrar-General publishes quarterly summaries of such returns,
as well as weekly returns of infectious diseases for the metropolis.
Marriages are usually stated in proportion to the total
population, or the number per thousand of population; but a
more accurate method would be to base the marriage-rate for
comparative purposes on the number of unmarried persons living
at marriageable ages. In England the marriage-rate is always
higher in large towns than in rural districts. Thus in 1900 the
marriage-rate in London was 17·6 as compared with an average
marriage-rate in 1891-95 of 15·2 per thousand of the estimated
population in England and Wales. The higher marriage-rate in
towns is chiefly owing to the fact that higher wages and greater339
scope for remunerative work attract young country people of
marriageable ages to towns.
Births are usually reckoned as a rate per thousand of
population. Clearly, however, if one population had a larger
proportion than another of women of child-bearing years this
method of comparison would not be free from possible error.
Even were the proportion of women of child-bearing ages equal,
the comparison might be fallacious if in one population the proportion
of single women was much higher than in the other.
Illegitimate births do not materially vitiate this conclusion, as
such births do not constitute more than 4 per cent. of the total
births, and this number is not excessive in the districts in which
there is the greatest excess of single women, viz. in districts
in which a large number of domestic servants are employed.
The only strictly accurate method is to subdivide the births into
legitimate and illegitimate, stating the former per 1,000 married
women of child-bearing years, and the latter per 1,000 unmarried
women of child-bearing years. I append an example of the
relative accuracy of the three methods above indicated12:—
|
BIRTH-RATE |
PER 1,000
INHABITANTS. |
PER 1,000 WOMEN
AGED 15-45. |
PER 1,000 MARRIED
WOMEN AGED 15-45 YEARS. |
| Kensington |
21.8 |
61.6 |
215.4 |
| Whitechapel |
39.9 |
172.1 |
328.3 |
| Percentage excess of birth-rate in
Whitechapel over that in Kensington |
83% |
179% |
53% |
Thus, according to the ordinary method (A) of stating the
legitimate birth-rate, it is 83% higher in Whitechapel than in
Kensington, whereas it is really only 53% higher. Similarly a
statement of the illegitimate birth-rate in the two districts “per
1,000 inhabitants,” shows an excess of only 6% in Whitechapel,
while a statement “per 1,000 unmarried women aged 15-45
years” shows the real excess of 144%. Both in this and other
civilised countries there has been in the last 25 years a steady
decline in the birth-rate. In England the maximum birth-rate was
36·3 per 1,000 of population in 1876, and the minimum 29·3 in 1899.
This diminution is only caused to a minor degree by postponement
of marriage to more mature years, and by a larger proportion of
celibacy. Nor is there any reasonable ground for the view that
a diminished power of either sex to produce children has been
produced by alcohol, syphilis, tobacco, or other causes. The main
cause of the diminution of the birth-rate is340 “the deliberate and
voluntary avoidance of child-bearing on, the part of a steadily
increasing number of married persons.”
Deaths are calculated in proportion to every 1,000 of the
population, the unit of time being a year. This unit is preserved
even when death-rates for shorter periods, e.g. a week, are stated.
Thus the death-rates for the 33 great towns published weekly in
the chief newspapers are annual death-rates; they represent the
number who would die per 1,000 of the population, supposing
the same proportion of deaths to population held good throughout
the year. The best plan to obtain the weekly annual death-rate
is as follows: the correct number of weeks in a year being 52·17747,
if the population of a town be 143,956, and the number of deaths in
a given week are 35, then the death-rate is 12·687. Thus:—
143,956 ∕ 52·17747 = 2758. 1,000 ∕ 2,758 = 0·3625. This is the factor by which the weekly
number of deaths must be multiplied.
35 × 0·3625 = 12·6875 or 12·7.
The above is the crude death-rate. Various corrections are required,
which must now be considered. The most important of these are
for public institutions, for visitors, and for age and sex. A public
institution, e.g. a workhouse, infirmary, or asylum, in a given
district may consist almost entirely of persons belonging to another
district. The rule is to relegate to the district to which they belong
all deaths of inmates of an institution, i.e. subtract all deaths of
outsiders occurring in inside institutions, and add all deaths of
inhabitants occurring in outside institutions. The population as
well as the deaths of these institutions should be excluded, in so far
as they are derived from the outside district, in order to make the
net death-rate approximately correct.
Theoretically the correction ought to be extended so as to
apply to visitors who do not die in public institutions. In practice,
however, this cannot be effected, until a central “clearing house”
is established. The exclusion of deaths of visitors from the district
in which they occur is easy; their inclusion in the returns of the
district from which they come is more difficult to secure. For
the present, they should be included in the death-rate of the
district in which they occur.
Death-rate according to Age and Sex.—To obtain a true
conception of the death-rate in a community, it is necessary to
state the number of deaths in each sex in proportion to the number
living at different ages. The importance of this is shown by the
following extract from the Registrar-General’s report for 1899.
England and Wales.—Deaths to 1,000 living at each of 12 groups
of ages.
|
ALL
AGES. |
AGED
0- |
5- |
10- |
15- |
20- |
25- |
35- |
45- |
55- |
65- |
75- |
85 AND
UPWARDS. |
| Males |
19·5 |
60·4 |
3·8 |
2·2 |
3·6 |
5·3 |
7·1 |
12·3 |
20·0 |
37·2 |
69·8 |
152·6 |
300·3 |
| Females |
17·3 |
50·7 |
3·9 |
2·3 |
3·3 |
4·3 |
6·1 |
10·0 |
15·4 |
29·8 |
61·5 |
142·6 |
272·0 |
Thus at ages over 5 and under 45 for males, and under 55 for
females, the death-rate is lower than is the total death-rate for all341
ages. For females at all ages except from 5 to 15, the death-rate
is lower than for males. From the above statement it will be
clear that a considerable excess of women (as in a residential
district with a large number of domestic servants) or a considerable
excess of either sex at the ages of 15 to 45 (as in most large
towns) in proportion to the number living at other ages, would
produce a lower total or crude death-rate, which does not imply
any truly more healthy condition than that of another district,
which is less favourably constituted so far as the proportion of
the sexes and the numbers living at different ages are concerned.
By a means of correction now to be described this source of error
can be eliminated. The method of obtaining the factor for correction
can be best understood by an example. The annual death-rate
of England and Wales in 1881-90 was 19.15, and the death-rate
at each age-group is given in the following table:
| AGES. |
MEAN ANNUAL DEATH-RATE IN ENGLAND AND WALES
1881-90, PER 1,000 LIVING AT EACH GROUP OF AGES. |
POPULATIONOF HUDDERSFIELD IN 1891 |
CALCULATED NUMBER OF DEATHSIN HUDDERSFIELD. |
| Under 5 |
Males.
61.59 |
Females.
51.95 |
Males.
4,551 |
Females.
4,785 |
Males.
280 |
Females.
249 |
| 5 |
5·35 |
5·27 |
4,691 |
5,081 |
25 |
27 |
| 10 |
2·96 |
3·11 |
5,113 |
5,165 |
15 |
16 |
| 15 |
4·33 |
4·42 |
4,905 |
5,549 |
21 |
25 |
| 20 |
5·73 |
5·54 |
4,541 |
5,461 |
26 |
30 |
| 25 |
7·78 |
7·41 |
7,466 |
8,834 |
58 |
65 |
| 35 |
12·41 |
10·61 |
5,576 |
6,265 |
69 |
66 |
| 45 |
19·36 |
15·09 |
3,944 |
4,649 |
76 |
70 |
| 55 |
34·69 |
28·45 |
2,393 |
3,017 |
83 |
86 |
| 65 |
70·39 |
60·36 |
1,128 |
1,590 |
79 |
96 |
| 75 and upwards |
162.62 |
147.98 |
250 |
466 |
41 |
69 |
|
44,558 |
50,862 |
773 |
799 |
| Totals |
\————/ |
\———/ |
|
95,420 |
1,572 |
The population of Huddersfield at each of the corresponding
periods as given by the census of 1891, is also
shown in this table, and in the last column the number
of male and female deaths that would occur by applying
the death-rates for England and Wales to the population
of Huddersfield are shewn. The total number of deaths thus
calculated is 1572 in a population of 95,420, and the total death-rate
= 16·47 per 1000. This is the standard death-rate, i.e., the
death-rate at all ages calculated on the hypothesis that the rates at
each of 12 age-periods in Huddersfield were the same as in England
and Wales during the ten years of the last intercensal period, viz.
19·15 in 1881-90.13 But the standard death-rate of Huddersfield342
would have been 19·15 instead of 16·47, were it not for the fact that
the distribution of age and sex in the Huddersfield population is
more favourable than in the country as a whole. Hence it must be
increased in the ratio of 19·15: 16·47, i.e., multiplied by the factor
19·15 ∕ 16·47 = 1·1627. When the crude or recorded death-rate for 1900
of 16·78 is multiplied by this factor we obtain the corrected death-rate
of 16·78 × 1·1627 = 19·51 per 1000, which is the correct figure to
compare with the death-rate of 18.31 for England and Wales in that
year. If the death-rate of England and Wales be stated as 1000,
then 1000 × 1951 ∕ 1831 = 1066, is the comparative mortality figure for
Huddersfield. Similarly in the year 1900 the comparative mortality
figure of London was 1093, of Croydon 831, of Norwich 919, while
that of Liverpool was 1539, of Salford 1541. In all the towns
except Plymouth and Norwich the corrected death-rate is higher
than the crude or recorded death-rate. This implies that, in all
except these two towns, the factor of correction is greater than
unity.
This is a convenient point for briefly discussing the relationship
between the birth-rate and death-rate. The opinion is commonly
held that a high birth-rate is a direct cause of a high death-rate,
owing to the great mortality amongst infants. The table on
page 340 shows that the death-rate at ages under five is three times as
high as at all ages together, and it is therefore natural to suppose
that a high birth-rate by producing an excessive proportion of
persons of tender years will cause a high general death-rate. This
might be so, if the birth-rate were to remain high for only five years.
But if the high birth-rate continued longer, the proportion of the
total population at ages of low mortality would be increased, and
the general death-rate would be lowered. We have already seen
that in nearly all the great towns, in which the birth-rate is higher
than in rural districts, the age distribution of the population is more
favourable to a low death-rate than in rural districts; and their
higher crude death-rate is made still higher than that of rural districts
when the necessary factor of correction is applied.
The Infantile Mortality should be stated in terms of the
infantile population. This is more accurately assumed to be equal
to the number of births in the given year, than estimated from the
number stated to be under one year of age at the last census. The
number of deaths under one year of age per 1000 births was 163 for
England and Wales in 1899, being lowest in the agricultural counties
and highest in manufacturing counties. In the 33 great towns it
averaged 172 in the year 1900, ranging from 132 in Croydon,
Huddersfield and Halifax to 236 per 1000 births in Preston. Of
1000 male children born in England and Wales in 1881-90, the
number surviving at the age of three months was 921, at the age of
six months 889, twelve months 839, while the number of female
children surviving one year of 1000 born was 869. In towns a
smaller number survive. Of the conditions causing this high343
infantile mortality, ignorance and inexperience on the part of parents
bear a considerable part, especially as influencing the food and
mode of feeding. The death-rates at other age-groups beyond
infancy are given in the table on page 340. Season influences the
death-rate. The third quarter of the year has the lowest death-rate,
unless the amount of Epidemic Diarrhœa has been excessive. In
the first quarter of the year, the highest death-rate usually occurs.
Mild winters and cool summers both lower the mortality. The
seasonal incidence of infectious diseases need only be mentioned
in passing.
Density of Population has important bearings on the death-rate.
Thus the urban districts in 1899 had a death-rate of 19·2 and
the rural of 16·3 per 1000 of population. Farr found that the death-rate
increased with the density of populations, not in direct
proportion, but in proportion to the 6th roots of the contrasted
populations. This rule does not now hold generally good. It is
only after the density has reached a certain degree of intensity that
it begins to exert an appreciable effect. Even then it is what is
implied in aggregation rather than the aggregation itself that is
pernicious. In particular, poverty is usually greater in densely
populated districts than elsewhere, with its accompaniments of
deficient food and clothing and bad housing. Hence the excess of
phthisis in tenemented houses, especially in houses with only three
rooms. I have shown that the true density that should be considered
is the number of persons to each room, not the number of persons
on a given area (“The Vital Statistics of the Peabody Buildings,”
Roy. Statist. Soc., Feb., 1891).
Occupation and Mortality.—To obtain correct statistics
showing the influence of occupation on vitality, one must know the
number and age of those engaged in each industry, and the
corresponding number of deaths. A statement of the mean age at
death of those engaged in different occupations would be most
fallacious (page 344). The best plan is to restrict the statistics to
men aged 25-65, and calculate for these death-rates in a standard
population, after the fashion already described (page 340). By
this means a “comparative mortality figure” can be obtained.
For all males it is 1000, for farmers 563, teachers 603, lawyers 821,
doctors 966, butchers 1096, plumbers 1120, brewers 1427, innkeepers
1659, potters 1706, file-makers 1810. Speaking generally, the
occupations are most unhealthy in which there is most exposure to
dust, to the breathing of foul air, and to excessive indulgence in
alcoholic drinks (for further details see the author’s Elements of
Vital Statistics, page 169 et seq.).
Deaths from Various Causes.—These may be stated in
proportion to total deaths from all causes, or in terms of the
population. The first plan must be adopted only when it is desired
to ascertain the proportional share of a given cause of death in the
total mortality. In 1899, in England and Wales the diseases
named in the first column of the table (page 344), were the most
prolific causes of deaths.
344
England and Wales, 1899.
Deaths from Various Causes to 10,000 Deaths from all Causes.
| Bronchitis |
880 |
| Phthisis |
729 |
| Pneumonia |
685 |
| Old age |
541 |
| Diarrhœa, Dysentery |
511 |
| Cancer |
452 |
| Apoplexy |
327 |
| Influenza |
213 |
| Whooping cough |
174 |
| Measles |
172 |
| Diphtheria |
160 |
| Enteric fever |
108 |
| Scarlet fever |
64 |
| Small-pox |
3 |
The diseases in the second column are given in order to
indicate their proportional share of the total number of deaths.
The proper plan of stating the death-rate from a given disease
is in terms of the population, or better still subdivided into death-rates
from the disease for different age-groups as in the table on
page 340, if the number of deaths is not too small to admit of this.
The importance of stating the death-rate for different age-groups is
greatest for such diseases as diarrhœa, whooping cough, and measles,
in which most of the deaths occur at ages under five. In the
following table are given the death-rates from the causes of death
which are most important, either from their magnitude, or because
of their preventible character:—
England and Wales, 1899.—Death-rate per 1,000 Persons living.
| Small-pox |
·005 |
| Measles |
·32 |
| Scarlet fever |
·12 |
| Influenza |
·39 |
| Whooping cough |
·32 |
| Diphtheria |
·29 |
| Enteric fever |
·20 |
| Typhus fever |
·001 |
| Cholera |
·04 |
| Diarrhœa, Dysentery |
·94 |
| Intemperance |
·0914 |
| Cancer |
·83 |
| Phthisis |
1·34 |
| Other tubercular diseases |
·58 |
| Premature birth |
·58 |
| Old age |
·99 |
| Apoplexy |
·60 |
| Convulsions |
·57 |
| Valvular disease of heart |
·38 |
| Bronchitis |
1·61 |
| Pneumonia |
1·26 |
| Gastro-enteritis |
·61 |
| Bright’s disease |
·29 |
| Accidents |
·59 |
| Ill defined and not specified causes |
·73 |
| ———— |
| All causes |
18·33 |
Determination of Longevity. We have hitherto considered
only death-rates, i.e. the number dying each year out of each 1,000 of
population. The mean duration of life involves another aspect of
the same problem. Although nothing is more uncertain than the
duration of individual life, the duration of life for the entire
community is subject to so little variation that annuities and life
assurance can be made the subject of exact calculations. Of the
tests employed to measure the duration of human life the most
commonly employed is the mean age at death.15
sum of ages at death.
Mean age at death = ——————————
number of deaths.
This is a fair method
of stating the average longevity of a particular group of persons, if
the group is sufficiently large to avoid the possible error caused by
paucity of data (page 349). But it would be entirely unsafe to
assume that by this means a safe standard of comparison between345
two groups can be formed. Thus in 1890 it was stated that the
mean age at death of workmen was 29-30 years, of the well-to-do
classes 55-60 years. This statement throws no light on the relative
vitality of the two classes under comparison. The well-to-do
classes consist largely of those whose working days are past; and
it is as untrustworthy to compare their mean age at death with that
of workmen, as it would be to base any conclusion on the fact that
mean age at death of bishops is much higher than that of curates.
The mean age at death is lowest in countries with a high birth-rate.
Hence it would be very fallacious to compare the mean age at
death in England and France.
The probable duration of life (vie probable) is a term sometimes
employed to denote the age at which any number of children
born into the world will be reduced to one half. In practice it can
only be ascertained from a life-table.
The true mean duration of life or expectation of life can only
be ascertained from a Life Table, and this must therefore be briefly
described. This is the true biometer, of equal importance in all
inquiries connected with human life with the barometer or thermometer
and similar instruments employed in physical research.
The Life Table represents “a generation of individuals passing
through time.” The data required for its construction are the
number and ages of the living, and the number and ages of the
dying, i.e. the data required for ascertaining the death-rate for each
year of life. Theoretically the best plan for forming a Life Table
would be to observe a million children, all born on the same day,
through life, entering in a column (headed lx) the number who
remain alive at the end of each successive year until all have died;
and in a second column (headed dx) the number dying before the
completion of each year of life. This method is impracticable,
and were it otherwise, the experience would be obsolete before it
could be utilised. The method employed in constructing the
national Life Tables for England is, without tracing the history
of individuals through life, to assume that the population being
given by the census returns and the death-rate for each age for a
given decennium being known, that the same death-rate will
continue during the remainder of the lives of the population
included in the census returns.
The total mean number living and the total number dying for
a given age-period are known. The mean chance (px) of living
one year during this age-period is found by the fraction
Population - ½ Deaths
——————————— = px
Population + ½ Deaths
It is usual to start with a million or 100,000 children at birth,
and to make a separate table for the proportionate number of males
and females at birth. Thus in Brighton in 1881-90 these were in
the proportion of 51,195 and 48,805. Starting with 51,195 male
infants at birth, and multiplying this number by ·84608, the
probability of surviving for one year, we obtain 51,195 × ·84608 =346
43,315. For the second year of life, the probability of surviving
was ·93398; hence the number of survivors is
43,315 × ·93398 = 40,452, and so on.
The general arrangement is shewn in the following example of
a Life Table, which only gives the data at or near the two extremes
of life, the intermediate figures having been omitted from considerations
of space.
Brighton Life Table.—Males.
(Based on the mortality of the 10 years 1881-90.)
AGE.
x |
DYING IN EACH YEAR OF LIFE.
dx |
BORN AND SURVIVING AT EACH AGE.
lx |
SUM OF THE NUMBER LIVING, OR YEARS OF
LIFE LIVED AT EACH AGE, x + 1, AND UPWARDS, TO THE LAST
AGE IN THE TABLE.
Σlx+1 |
MEAN AFTER LIFE-TIME (EXPECTATION OF
LIFE) AT EACH AGE.
exº |
| 0 |
7,880 |
51,195 |
2,206,174 |
43·59 |
| 1 |
2,863 |
43,315 |
2,162,859 |
50·43 |
| 2 |
996 |
40,452 |
2,122,407 |
52·96 |
| 3 |
733 |
39,456 |
2,082,951 |
53·29 |
| 4 |
440 |
38,723 |
2,044,228 |
53·29 |
| — |
— |
— |
— |
— |
| — |
— |
— |
— |
— |
| 97 |
12 |
29 |
43 |
1·60 |
| 98 |
7 |
17 |
26 |
1·53 |
| 99 |
4 |
10 |
16 |
1·48 |
The 43,315 males surviving to the end of the first year of life
out of 51,195 born will each have lived a complete year in the first
year, or among them 43,315 years. Similarly the 40,452 males will
live among them 40,452 further complete years, and so on, until all
the males started with become extinct at the age of 105. Evidently,
therefore, the total number of complete years lived by the 51,195
males started with at birth will be
43,315 + 40,452 + 39,456 + 38,723 + ... + 10 + 6 + 4 + 3 + 2 + 1
= 2,206,174 years, this sum being obtained by adding together the
numbers living at each age beyond (i.e. below on this table) the age
in question right down to its last item. This number of years is
lived by 51,195 males. Hence the number of complete years lived
by, i.e. the expectation of life of, each male
= 2,206,174 ∕ 51,195 = 43·09 years.
This is the curtate expectation of life. It deals only with the complete
years of life, not taking into account that portion of life-time lived
by each person in the year of his death, which may be assumed to
be on an average half a year. Hence the complete expectation of life
according to the above table is 43·59 years.
In the following table the expectation of life (complete) for
various towns and for England is given:—
347
Life Table.—Expectation of Life at Birth.
|
Male. |
Female. |
| English Life Table, |
1838-54 (Farr) |
39·91 |
41·85 |
| „ |
1871-80 (Ogle) |
41·35 |
44·62 |
| „ |
1881-90 (Tatham) |
43·66 |
47·18 |
| London, 1881-90 (Murphy) |
40·66 |
44·91 |
| Brighton, 1881-90 (Newsholme) |
43·59 |
49·25 |
| Manchester City, 1881-90 (Tatham) |
34·71 |
38·44 |
| Glasgow, 1881-90 (Chambers) |
35·18 |
37·70 |
Formulæ of varying degrees of accuracy have been devised
for giving in the absence of a Life Table an approximation to the
expectation of life.
Willich’s Formula is as follows:—If x = expectation of life,
and a = present age, then x = 2 ∕ 3 (80-a). Thus, at the age of 50 years
the expectation of life, according to this formula, is 20 years. By
the English life-table for 1881-90 it was 18.82 for males, and
20·56 for females. Farr’s formula is based on the birth and
death-rates. If b = birth-rate and d = death-rate per unit of population,
then
Expectation of life = (2 ∕ 3 × 1 ∕ d) + (1 ∕ 3 × 1 ∕ b).
Thus b for England and Wales, 1889-98 = 30·3 ∕ 1,000 = ·0303.
and d for England and Wales, 1889-98 = 18·4 ∕ 1,000 = ·0184.
(2 ∕ 3 × 1 ∕ ·0303) + (1 ∕ 3 × 1 ∕ ·0184) = 47.2 years, as compared with the
expectation of life for 1881-90 shown in the above table.
| In a life-table the number out of which one dies annually |
 |
are |
| the mean age at death |
identical |
| and the expectation of life |
in value |
when the whole duration of life from birth to death is included
in the calculation. This is only true for a stationary or life-table
population, in which the number dying is assumed to be regularly
replaced by a corresponding number of persons of the same age.
Life Capital.—The life-tables now in use are those based on
the experience of 1881-90. The gain in any subsequent year, as in
1900, may be ascertained as follows: the mean population and the
death-rate for each age-group as 0-5, 5-10, etc., are calculated.
Then the mean death-rate of the same community for 1881-90 is
applied to this population. By this means the “calculated number”
of deaths in 1900 is obtained. The difference between these numbers
and the “actual number” obtained from the death-registers, gives
the gain or loss during the year. Next multiply these differences
by the mean expectation of life for the corresponding groups of
years. By adding the gains thus ascertained and subtracting any
losses, we obtain the net gain in “life-capital” (Tatham) during
the year 1900.
348
Tests of the Health of a Community. 1. The general
death-rate is the test most commonly applied, and generally trusted.
It has its limitations in this respect. It may usually be trusted
in comparing a town or district for a single year with preceding
years, as the age and sex distribution of a given population only
changes slowly. But when comparison with other towns or
districts is made, the possibility that erroneous conclusions may
be drawn becomes considerable. (a) Before the death-rates of two
districts can be compared, either this comparison must be made
by means of death-rates for age-groups (0-5, 5-10, ... 65-75, etc.)
or the factors of correction, the method of obtaining which is
described on page 341, must be applied. (b) It must be ensured
that in the two compared districts, an equal amount of correction
has been made for deaths occurring in public institutions and
among visitors (page 340). (c) Even when the above precautions
are taken, it is conceivable that a town with a death-rate of 15 per
1,000 may really be as healthy as another with a death-rate of 12
per 1,000, though a statistical justification of this statement is a
difficult task.
Social conditions quite irrespective of the sanitary condition or
the natural salubrity of a district have an important influence on
the death-rate. Poverty and all that it connotes, necessarily involves
a higher death-rate than occurs among the well-to-do. Furthermore,
the domestic servants employed by the latter frequently die in
districts other than those in which they are employed, without any
possibility of the requisite correction being made.
2. The zymotic death-rate is frequently quoted as a test of
sanitary condition. This is a death-rate based on the deaths from
the “seven chief zymotic diseases,” small-pox, measles, whooping-cough,
diphtheria, scarlet fever, fever (chiefly enteric), and diarrhœa.
This death-rate should be entirely discarded, the death-rate from
each infectious disease being separately stated. A high death-rate
from enteric fever would be a much more serious reflection on the
health of a town than a high death-rate for whooping-cough.
The death-rate from each of these diseases in London and in
England in 1899 was as follows:—
DEATH-RATE IN 1899 PER 1,000 LIVING.
|
England and Wales. |
London. |
| Small-pox |
·005 |
nil |
| Measles |
·32 |
·47 |
| Scarlet fever |
·12 |
·08 |
| Diphtheria |
·29 |
·43 |
| Typhus |
·001 |
nil |
| Enteric fever |
·20 |
·18 |
| Whooping cough |
·32 |
·38 |
| Diarrhœa |
·94 |
·92 |
A statement of the death-rate from each of these diseases for a series
of years is a much more trustworthy test than a similar statement
for a single year, in which accidental causes may have caused a
temporary increase, or than a statement of the average result for a
series of years, which tends to conceal the epidemic variations of the349
disease in question. The danger of such averages has been well
exposed by Chadwick in the remark that “a mean between the
condition of Dives and Lazarus tends to make it appear that after
all Lazarus has not so much to complain of.”
3. The infantile mortality (page 342) is a delicate test of mixed
sanitary and social conditions, and stress may always be laid on it
from these standpoints. The importance of comparing death-rates
at other age-groups has already been explained.
4. The most delicate and exact method, if all the data are
accurate and complete, is to construct a Life Table, and ascertain
the expectation of life in comparison with that of other communities.
The preceding statistical tests of the salubrity of a community,
and any others that may be available, should all, when practicable,
be utilised; and it should always be remembered that these tests,
especially the general death-rate, are most trustworthy when
contrasting the experience of a community with its past experience,
and least trustworthy when contrasting its experience with that of
others; owing to the difficulty in the latter case of ensuring the
avoidance of error arising from non ceteris paribus.
Statistical Fallacies.—If “fallacies” be regarded as
synonymous with “errors,” clearly they may occur at every step.
They may be classified as errors of data, and errors of methods.
The most important errors of data are erroneous estimates of
population, and erroneous returns of deaths, especially in the
direction of exclusion of certain deaths (page 340). Death-rates
for short periods are relatively untrustworthy. The erroneous use
of the mean age at death as a test of longevity has been mentioned
(page 344). These are in part also errors of methods, and numerous
mixed examples are given below.
Errors from Paucity of Data frequently arise, the “fallacy
of small numbers,” a too hasty generalization, being the most
common fault in medical writings, especially in therapeutics. The
degree of approximation to the truth of a varying number of
observations is estimated by means of Poisson’s formula.
- μ = total number of cases recorded in two groups.
- m = number in one group.
- n = number in the other group, so that μ = m + n.
The extent of variation in the proportion of each group to the whole will
vary within the proportions represented by—
m ∕ μ + 2√(2mn ∕ μ3), and n ∕ μ - 2√(2mn ∕ μ3)
The larger the number of the total observations (μ), the less will be the
value of 2√(2mn ∕ μ3), and the less will be the limits of error in the simple
proportion m ∕ μ.
Thus, of 147 cases of enteric fever, 17 died, a fatality of 11·4 per cent. The
possible error is determined by the second half of the above formula—
= 2√(2 × 17 × 130 ∕ 1473) = 2√(4,420 ∕ 3,176,523) = ·0746.
350
i.e. the possibility of error = ·0746 to unity or 7·46 per cent. In other
words, in a second series of cases of enteric fever under the same
conditions as the above, the fatality may vary from 3·94 to 18·86
per cent., a vague result which indicates that the first series cannot
be regarded as establishing more than a primá facie case in favour
of any special method of treatment that may have been adopted.
Non ceteris paribus.—The necessity that data to be compared
shall be collected on a uniform plan, and be of a strictly comparable
nature, is very frequently ignored. The conclusion that the
administration of a given antiseptic is a valuable means of treating
enteric fever is not demonstrated by the fact that the fatality in the
series of cases thus treated is 7 per cent., while in another series
treated without antiseptics it is 14 per cent., unless it is shown that
the age and other previous conditions of the patients in the two
cases were not widely different, and unless the series are sufficiently
long to avoid the fallacy due to paucity of data.
Errors from the Composition of Rates.—If the death-rate
of A having a population of 10,000 is 10 per 1000, and of B
having a population of 20,000 is 15 per 1000, the combined death-rate
is not (10 + 15) ∕ 2 = 12·5. To obtain the correct combined death-rate,
the number of deaths in A (=100) and in B (=300) must first
be ascertained, and the death-rate on a population of 30,000 in which
400 deaths occurred will then be found to be 13·3 per 1000.
Errors from Stating Deaths in proportion to Total
Deaths.—There is nothing erroneous per se in stating the proportion
of deaths at one age as a ratio of the total deaths at all ages,
or the deaths from one cause as a ratio of the total deaths from all
causes. It is a useful and in fact the only method practicable when
it is required to give the proportion of one of these to the other.
But beyond this, such a ratio cannot be trusted. For instance, the
proportion of fatal accidents among male infants is 12·2, and among
female infants 25·1 per cent. of the total fatal accidents in the male
and female sex respectively. But it would be erroneous, if it were
concluded from these figures that female are more subject to fatal
accidents than male infants. The only conclusion that they justify
is that at higher ages females are much less subject to fatal
accidents than males. In actual facts, for every 1000 infants
born, only 2·9 female as against 3·1 males die under one year of
age as the result of accident.
Again, suppose the case of two towns, A and B. A with
a population of 10,000 has 150 annual deaths, of which 20 are
caused by cancer; the general death-rate therefore being 15, and
the death-rate from cancer 2·0 per 1000, while the deaths from
cancer form 2 ∕ 15 of the total deaths. B, with the same population
as A, has 300 deaths, its death-rate being 30 per 1000, and 40
deaths from cancer, its cancer death-rate being 4·0 per 1000; while
the proportion of the deaths from cancer to the total deaths is
2 ∕ 15 as before. It is useful to know in regard to each of these
individual communities that cancer causes 2 ∕ 15 of its total mortality,351
but no comparison between the two is practicable on this basis.
The only proper comparison is between the death-rate from cancer
per 1000 of population in A and B, which shows that it is twice
as high in B as in A. A still more accurate method is to ascertain
the number of deaths from cancer, and the number living at different
age-groups, thus avoiding any errors due to variations in age and
sex distribution of population.
Errors as to Averages.—The most common of these results
from paucity of data (page 349). Note that the results obtained
from an average cannot be applied to a particular case. The
mean duration or expectation of life, obtained from a life-table,
expresses with almost mathematical certainty, the number of years
of life of the members of a community taken one with another, but is
often not accurate when applied to a single individual.
In Army statistics errors have arisen by failure to comprehend
what is meant by the average strength of a force. The statistics
must comprise the lives of a given number of persons as well
as the deaths occurring among them for an entire year, or allowance
must be made in this respect when required.
Hospital statistics for similar reasons are frequently fallacious.
Thus death-rates have been frequently given per 100 occupied
beds, which are most misleading, as the frequency of succession
of patients as well as the nature of the patients’ complaints will
vary greatly in different hospitals. The only proper method of
stating hospital-returns is on the basis of the aggregate annual
number of cases treated to a termination. The cases should be
further subdivided according to age and sex and disease. Average
death-rates for epidemic diseases when used to compare one community
with another may give rise to erroneous conclusions. This is
inseparable from the nature of such diseases. During the period
under comparison, one town may happen to have, say, three
epidemics, and the other four; possibly if two or three additional
years had been added to the series, the place of the two towns
would have been reversed as regards their average death-rate
from the disease in question. The proper plan is to give the
death-rates from the epidemic disease for every year recorded, to
draw a curve of these death-rates for the two towns on the same
scale, and to compare the height, the variations of height, and
the trend of the curve in each instance.
352
INDEX.
- A
- Acarus Scabiei, 276
- Achorion Schönleinii, 276
- Acne, 161
- Aerated Waters, 46
- Ague (see Malaria)
- Air—Bacteria in, 109, 121
- Composition, Properties of, 100
- Contagia and, 109, 114
- Degree of Moisture of, 119, 128
- Gaseous and Other Impurities of, 111
- Examination of, 126
- Moisture of, 234
- In Soil, 221
- Suspended Impurities of, 105
- Temperature of, 119, 128
- of Mountains, 228
- Purification of, 129
- Air-space, amount required, 142
- Alcohol, 55
- Amines process, 191
- Ammonia—in Air, 101
- in Water, 86
- Amyloids, 7
- Amylaceous Foods, 19
- Anopheles (see Mosquito)
- Anthrax, 274, 306
- Anti-cyclones, 233
- Antidiphtheritic Serum, 268, 299
- Anti-syphonage, 174
- Antill Trap, 180
- Apjohn’s Formula, 242
- Arnott’s Valve, 149
- Arrowroot, 19
- Arsenic—in Beer, 62
- in Wall Papers, 214
- in Clothing, 272
- Artesian Wells, 71
- Artificial Ventilation, 152
- Relative Value, 154
- Ascaris, 278
- Ash-pits, 199
- Aspect, 201
- Atavism, 246
- B
- Bacteria, 273
- Bacterial Methods, Sewage Treatment, 192
- Ballard’s Researches, 303
- Barley, 18
- Barometers, 237
- Barometric Pressure, 231
- Baths, 262
- Beaufort’s Scale, 243
- Beef, 10
- Beer, 61
- Bell-trap, 180
- Benham’s Gas Burners, 157
- Berkefeld Filter, 98
- Beverages, 46
- Bilharzia Hæmatobia, 277
- Birds, Flesh of, 11
- Birth-rate, 339
- Black Death, 1
- Bonds, English, Flemish, 205
- Borax and Boric Acid, 44
- Boyle’s Mica Valve, 149
- Bowels, Attention to, 248
- Brandy, 63
- Bread, brown and white, 17, 41
- Bricks, 205
- Broad Irrigation, 192
- Burnett’s Solution, 327
- Butter, 14
- Buys Ballot’s Law, 232
- C
- Caffeine, 48
- Calorigen Stove, 157
- Carbohydrates, 6
- Carbolic Acid as Disinfectant, 327
- Carbon Filters, 97
- Carbonic Acid—in Air, 102, 111
- estimation of, 126
- Carbonic Oxide, 112
- Calorie, 32
- Carpets, 218
- Cement, 213
- Cereal Foods, 15
- Cesspools, 183
- Cheese, 13
- Chicory, 50
- Chigoe, 277
- Chlorides in Water, 81
353
- Chlorine as Disinfectant, 324
- Cholera, 92, 225, 302
- Cisterns, 76
- Clarke’s Process, 94
- Soap Test, 79
- Climate, 227
- Closets, 168
- Clothing, 265
- Coal Gas, 115
- Cocoa, 51
- Cod Liver Oil, 14
- Coffee, 50
- Cols, 233
- Comparative Mortality Figures, 342
- Concrete, 207
- Condiments, 45
- Condy’s Fluid, 325
- Conservancy Methods, 194
- Constipation, 248
- Constitution, 245
- Consumption (see Tuberculosis)
- Container, 169
- Cooking, 339
- Cooper’s Ventilators, 149
- Corrosive Sublimate as Disinfectant, 327
- Cotton, 270
- Cream, 15
- Cyclones, 232
- Cysticercus Cellulosæ, 23, 279
- D
- D-Trap, 169
- Damp, Cause of, 211
- Damp-proof Course, 210
- Death-rate, 2
- Crude, 340
- Corrected, 342
- Zymotic, 297, 348
- Decomposed Meat, 24
- Demography, 335
- Density of Population, 343
- Destructors, 200
- Dew Point, 241
- Diarrhœa, Epidemic, 302
- Diet, 29
- Dietaries, 33
- Diffusion, 129
- Diphtheria, 225, 298
- Diseased Animals, Meat of, 23
- Disinfection, 324
- Distillation, 94
- Drainage, 166
- of Soil, 226
- Drains, 177
- Examination of, 180
- Drugs, 54
- Dust in Air, 105
- Dust-bins, 199
- Dyspepsia, 28, 248
- E
- Earth-closets, 195
- Eggs, 11
- Ellison’s Bricks, 151
- Endemic Diseases, 289
- Enteric Fever, 91, 224, 300
- Epidemic Diarrhœa, 26, 225, 302
- Epidemic Diseases, 289
- Erysipelas, 110, 305
- Excreta, 165
- Exercise, 249
- Expectation of Life, 346
- F
- Farcy (see Glanders)
- Farr’s Formula, 347
- Fats, 6
- Favus, 276
- Fermented Drinks, 55
- Filaria, 278
- Filters, 97
- Filtration, 95
- Fire Grates, 159
- Fish, 11
- Dangers of, 26
- Flax, 270
- Fleas, 277
- Flesh Food, 9
- Floors, 217
- Flour, 16
- Flukes (see Trematoda)
- Fomites, 299
- Food, 4
- Diseases due to, 23
- Heat value of, 32
- Preparation and Preservation of, 38, 43
- Relation of, to Mechanical Work, 36
- Variations from Climate, Age, &c., 29
- Vegetable and Animal, 31
- Forests and Climate, 229
- Formalin., 44
- and Formaldehyde as disinfectants, 326
- Fortin’s Barometer, 238
- Fruits, 21
- Fuel, 161
- Fungi, 20
- G
- Gaertner’s Bacillus, 25
- Galton’s Stove, 155
- Gas Stoves, 157, 161
- Gelatin, 9
- Gin, 63
- Glanders, 304
354
- Glaisher’s Tables, 241
- Glucose, 21
- Gluten, 15
- Gout, 28, 59
- Goux System, 194
- Graveyards, Air of, 118
- Green Vegetables, 20, 42
- Ground-water, 222
- Gully-trap, 167, 180
- H
- Habits, 247
- Haffkine’s Cholera Vaccine, 228
- Hardness of Water, 79, 90
- Heat, kinds of, 159
- Hendon Disease, 27
- Heredity, 245
- Hermite Process, 191
- Hopper-closets, 170
- Hot Water Supplies, 163
- Pipes, 162
- House Construction, 209
- Drainage, 165
- Materials of Construction of, 205
- Position of, 201
- Refuse, 198
- Slops, 165
- Humidity, absolute and relative 235
- Measurement of, 240
- Hydatids, 279
- Hydrocarbons, 6
- Hydrophobia, 305
- Hygrometers, 101, 240
- I
- Idiosyncrasy, 246
- Illuminants, 115
- Immunity, 287
- Incubation Periods, 287
- Infantile Mortality, 342
- Infective Diseases, 284
- of Animals, 24
- Infectious Diseases, Hospitals for, 320
- Notification of, 338
- Influenza, 309
- Insects and Disease, 281
- Intermittent Downward Filtration, 192
- Iron in Water, 83
- Isobars, 231
- Isolation, 319
- Isothermal Lines, 230
- J
- Jenner’s Discoveries, 293
- K
- Koch’s Postulates, 285
- L
- Latrines, 174
- Laveran, 282
- Lead in Water, 82, 90
- Workers in, 108
- Lice (see Pediculi)
- Liebig’s Extract of Meat, 10
- Life Capital, 347
- Tables, 345
- Linen, 270
- Lysol as Disinfectant, 327
- M
- Macaroni, 17
- Main Sewers, 183
- Maize, 18
- Malaria, 91, 224, 307
- Mallein, 304
- Malt, 18, 62
- Malted Bread, 17
- Margarine, 15
- McKinnell’s Ventilator, 149
- Metabolism, 6
- Measles, 297
- Meat, 9
- Diseased, 24
- Tinned, 25
- Parasites in, 23
- Meat Poisoning, 25
- Metchnikoff’s Theory, 289
- Meteorological Observations, 237
- Metallo-keramic Joint, 177
- Miasm, 285
- Microbes (see Bacteria)
- Microscoporon Audouini, 275
- Milk, 12
- and Disease, 26, 296, 299, 301
- Boric Acid in, 44
- Souring of, 274
- and Tuberculosis, 311
- Mineral Waters, 47
- Mortar, 206
- Mosquito, 282, 308
- Moule’s Closet, 195
- Mountain Climate, 228
- Mutton, 10
- N
- Natural Ventilation, 147
- Relative value, 154
355
- Nematoda, 278
- Nitrogen, 274
- Nitrates in Water, 81
- Nitrification, 192, 220
- Nitrogenous Foods, 5
- Notification of Consumption, 314
- Infectious Diseases, 318, 338
- Trade Diseases, 337
- O
- Oatmeal, 17
- Obesity, 28
- Occupation and Mortality, 343
- Offensive Trades, 120
- Oidium albicans, 275
- Oils, 46
- Onion, 20
- Over-feeding, 28
- Oxygen in Air, 100
- Oxygen Absorbed, 86
- Oxyuris, 278
- Oysters and Disease, 291
- Ozone, 101
- P
- Pan-closets, 169
- Pandemics, 289
- Parasites, 276
- in Meat, 23
- Pasteur-Chamberland Filter, 98
- Pasteurization of Milk, 13, 312
- Pediculi, 277
- Personal Hygiene, 245, 257, 260
- Pettenkofer’s Method, 126
- Phagocytosis, 287
- Phosphorus, in Food, 5
- Workers with, 108
- Phthisis, 225, 310
- Piles, 248
- Plaster, 212
- Plague, 281, 305
- Pneumatic System, Liernur, 183
- Poisson’s Formula, 349
- Population, Density of, 343
- Estimation of, 336
- Pork, 11
- Porter-Clarke Process, 95
- Portland Cement, 206
- Potato, 20
- Privy or Midden System, 196
- Proof Spirit, 64
- Proteids, 5
- Propulsion, Ventilation by, 153
- Puerperal Fever, 306
- Q
- Quarantine, 320
- R
- Rabies (see Hydrophobia)
- Rainfall, 235
- Rain Gauge, 242
- Water, 67
- Pipes, 166
- Rats and Plague, 306
- Registration of Births and Deaths, 337
- Relapsing Fever, 28, 300
- Respiration, 102, 113
- Rest, 257
- Rheumatic Fever, 225, 306
- Rickets, 28
- Ringworm, 275
- Rivers, 72
- Roasting, 38
- Roofs, 208
- Rum, 63
- Rye, 18
- S
- Saccharomycetes, 275
- Sago, 19
- Salts as Food, 7
- Saprophytes, 286
- Scarlatina or Scarlet Fever, 296
- Schools, Closure of, 322
- Scott Moncrieff Process, 192
- Scrofula, 316
- Scurvy, 2, 27
- Seasonal Incidence of Diseases, 289
- Sea Relation to Climate, 229
- Sebaceous Glands, 260
- Semolina, 17
- Septic-tank, 193
- Sewage—Disposal of, 190
- Dry and Wet Methods of Removal of, 197
- Sewers, 183, 184
- Air of, 117
- Problems as to flow in, 187
- Shell-fish and Disease, 291
- Sheringham’s Valve, 151
- Shone System, 186
- Sinks, 167
- Silk, 270
- Sleep, 258
- Slop-closets, 174
- Small-pox, 2, 292
- Smoke Nuisance, 160
- Test, 181
- Soap, 261
- Soil, 219
- Diseases in Relation to, 224
- Drainage of, 226
- Soil-pipe, 174
- Spices, 46
- Spirits, 63
- Springs, 68
356
- Starch, Detection of, 19
- Starvation, 27
- Statistical Fallacies, 349
- Steam as Disinfectant, 329
- Stoves, 157, 161
- Streets, Width of, 203
- Sugar, 21
- Sulphuretted Hydrogen, 112
- Sulphurous Acid (Disinfectant), 325
- Summer Diarrhœa, 225
- Sunshine Recorders, 243
- Sweat Glands, 260
- Sweating Sickness, 1
- Sylvester’s Method, 150
- Synoptic Map, 231
- Syphonic Closet, 172
- T
- Tannin, 49
- Tape-worms, 279
- Tapioca, 19
- Tea, 30, 47
- Temperature of Rooms, 158
- Tetanus, 304
- Thermometers, Maximum and Minimum, 239
- Thermometers, Dry and Wet Bulb, 240
- Thrush, 275
- Tight Lacing, 267
- Tinea, 276
- Tobacco, 54
- Tobin’s Tubes, 152
- Toxins, 286
- Trades and Disease, 107, 118
- Offensive, 120
- Traps, 179
- Trematoda, 277
- Trichina Spiralis, 23
- Trichophyton, 275
- Trough-closets, 174
- Tuberculin Test, 311
- Tuberculosis, 309
- Prevention of, 314
- Tuberculous Meat, 24
- Typhoid Fever (see Enteric Fever)
- Typhus Fever, 300
- V
- V-shaped Depressions, 233
- Vaccination, 293
- Valve-closets, 170
- Variola (see Small-pox)
- Veal, 10
- Vegetables, 20
- Ventilation, 132
- Methods of, 146
- Problems as to, 137
- By Propulsion, 153
- Ventilation by Introduction of Warmed Air, 155
- Of Mines, 156
- Of Drains, 178
- Vermicelli, 17
- Vernier, 238
- Vinegar, 46
- Vital Statistics, 335
- W
- Wall-paper, 214
- Walls of House, 210
- Wanklyn’s Ammonia Process, 86
- Warming of Houses, 159
- Wash-out and Wash-down Closets, 172
- Waste-pipes, 166
- Water, 65
- As food, 8
- Analysis of, 84
- Classification of supplies of, 73
- Constant and Intermittent Services, 76
- Enteric Fever and, 72
- Hardness of, 79
- Impurities of, 78, 89, 90
- Sources of, 66
- Storage and Delivery of, 74
- in Soil, 222
- Purification of, 94
- Water-closets, 168
- Water-gas, Carburetted, 115
- Water-test of Drains, 182
- Weather Forecasting, 234
- Weissman on Heredity, 246
- Wells, 69
- Whiskey, 63
- Whooping Cough, 298
- Widal Reaction, 301
- Willich’s Formula, 347
- Windows, 216
- Winds, 131, 230, 243
- Wines, 62
- Wool, 269
- Wool-sorters’ Disease, 274
- Work, Amount done, 252
- Relation of Food to, 36
- Wort, 62
- Writers’ Palsy, 252
- Y
- Yellow Fever, 305
- Z
- Zygophyta, 275